Staphylococcus capitis is a Gram-positive, coagulase-negative coccus, present as a part of the human normal flora mostly localized in areas around the scalp and face that have been lately associated with bacteria in neonates.
- Staphylococcus capitis is a coagulase-negative Staphylococci with documented potential for both human disease and nosocomial spread.
- It is non-motile, non-spore-forming facultatively anaerobic even though the best growth is observed under aerobic conditions.
- The species name ‘capitis’ is derived from the Latin name ‘caput’ meaning head, indicating the part of the human body where cutaneous populations of this species are usually the largest and most frequent.
- S. capitis is further divided into two subspecies on the basis of various characteristics; S. capitis subsp. capitis and S. capitis subsp. ureolyticus.
- It was first discovered or isolated by Kloos and Schleifer in 1975 from human skin; subspecies S. capitis subsp. capitis was also identified by Kloos and Schleifer in 1975 but subspecies S. capitis subsp. ureolyticus was discovered by Bannerman and Kloos in 1991.
- Staphylococcus capitis has been implicated in biofilm-related infections such as endocarditis, urinary tract infection, and catheter-related bacteremia. S. capitis is likely to be transmitted mostly via humans.
- S. capitis is particularly important in neonatal intensive care units where it causes up to 20% of cases of neonatal sepsis.
- Both S. capitis subsp. capitis and S. capitis subsp. ureolyticus are present on the human body as a part of the normal flora, mostly around the head.
Classification of Staphylococcus capitis
- Classification of Staphylococcal species is primarily based on the cell wall composition, G + C content of DNA, DNA–DNA hybridization, 16S rRNA sequence similarities, and the genome sizes.
- The members of the same species demonstrate relative DNA-binding values of generally 70 percent or greater, whereas organisms representing different species demonstrate relative DNA-binding values of less than 70 percent.
- The further classification of subspecies of S. capitis is based on their urease activity, ability to produce acid from maltose in anaerobic conditions, fatty acid profile, larger colony size, and DNA sequence differentiation.
The following is the taxonomical classification of S. capitis:
| Domain | Bacteria |
| Phylum | Firmicutes |
| Class | Bacilli |
| Order | Bacillales |
| Family | Staphylococcaceae |
| Genus | Staphylococcus |
| Species | S. capitis |
| Subspecies | S. capitis subsp. capitis |
| Subspecies | S. capitis subsp. ureolyticus |
Habitat of Staphylococcus capitis
- Humans are the primary host of S. capitis subsp. capitis whereas primates are the primary host of S. capitis subsp. ureolyticus.
- Even though other coagulase-negative Staphylococci are the predominant species present on the scalp of preadolescent children, these are replaced by S. capitis after puberty.
- The increase in populations of S. capitis on the scalp after puberty is associated with an increase in sebaceous gland activity at puberty.
- S. capitis subsp. capitis has a strong preference and S. capitis subsp. ureolyticus has a moderate preference for the human head.
- Besides, S. capitis subsp. ureolyticus is also found in the external auditory meatus of adults, but it doesn’t show any urease activity as there is only a minimal amount of urea.
- Despite their strong preference for the head, S. capitis is sometimes found in other areas of the body like arms and legs.
Morphology of Staphylococcus capitis
- S. capitis subsp. capitis is non-motile, non-spore-forming, Gram-stain-positive cocci, with an average size of 0.8–1.2 μm in diameter.
- The cells occur in pairs and are often seen in tetrads as a result of their division in more than one plane to form irregular grapelike clusters.
- The cells are facultatively anaerobic, and the strains of S. capitis are variably encapsulated.
- The cell wall is composed of peptidoglycan and teichoic acid in the ratio of approximately 60–30 percent respectively of the dry weight.
- The peptidoglycan is the main structural polymer in the wall, and it plays a vital role in maintaining the spherical shape of the cell.
- The cell membrane is a lipid-protein bilayer where phospholipids, glycolipids, menaquinones, and carotenoids make up the major lipid components of the membrane, and proteins like adenosine triphosphatase, polyprenolphosphokinase, various oxidases and dehydrogenases, and several penicillin-binding proteins serve as cell membrane-associated proteins.
- Few cell wall-associated proteins of S. capitis have been characterized and shown to promote adherence to extracellular matrix proteins and soluble plasma components.
- Some of the cell-wall associated proteins like SdrF, SdrG, and SdrH as members of the Sdr family of proteins are found in S. capitis that promote the attachment and adhesion of the organism to the host surface.
Cultural Characteristics of Staphylococcus capitis
Species of Staphylococcus can be grown on a different set of agar and liquid media, which helps in the identification and selective growth of the organism. These organisms are capable of growing in multiple sets of culture media like nutrient agar, Mannitol Salt Agar, P agar, and thioglycollate medium. Depending on various cultural characteristics like growth patterns and colony morphology, species-level identification can be made.
The following are the colony morphology of S. capitis on different media:
1. Nutrient agar (NA)
- Circular, cream-colored to white colonies of S. capitis are observed on NA. The colonies are mostly 1 mm in diameter with an entire margin.
- The colonies have raised elevation and a dense center with transparent borders.
2. Mannitol Salt Agar (MSA)
- Yellow colonies of the size 1-3 mm are seen which are surrounded by yellow zones indicating the release of acid as a result of mannitol utilization.
3. P agar
- S. capitis subsp. capitis: White or greyish colonies of the size 1-3mm in diameter that are smooth, slightly convex, glistening, and opaque are seen on P agar. The color might change to yellow or yellow-orange after storage at 1-4°C.
- S. capitis subsp. ureolyticus: Raised, opaque, glistening colonies are seen on P agar. Some strains develop yellow pigmentation after late incubation. Colonies may be either smooth or rough and have either slightly irregular or entire edges. The colony diameter is about 4.3–7.1 mm.
4. Blood agar (BA)
- Wrinkled, medium-sized (1-4 mm in diameter), β-hemolytic, opaque, rough white colonies are observed. Colony pleiomorphism is common on blood agar.
- Prominent hemolysis can be seen after 48 hours of incubation.
- S. capitis subsp. ureolyticus doesn’t show hemolysis.
Biochemical Characteristics of Staphylococcus capitis
The biochemical characteristics of S. capitis can be tabulated as follows:
| S.N | Biochemical Characteristics | Staphylococcus capitis |
| 1. | Capsule | No capsule |
| 2. | Shape | Cocci |
| 3. | Catalase | Positive (+) |
| 4. | Oxidase | Negative (-) |
| 5. | Citrate | Negative (-) |
| 6. | Methyl Red (MR) | Negative (-) |
| 7. | Voges Proskauer (VP) | Negative (-) |
| 8. | Urease | Negative (-) |
| 9. | Coagulase | Negative (-) |
| 10. | DNase | Positive (+) |
| 11. | Clumping factor | Negative (-) |
| 12. | Gas | Positive (+) |
| 11. | H2S | Positive (+) |
| 12. | Hemolysis | Positive (+) for S. capitis subsp. capitis and Negative (-) for S. capitis subsp. ureolyticus. |
| 13. | Motility | Negative (-) |
| 14. | Nitrate Reduction | Positive (+) for S. capitis subsp. capitis, variable for S. capitis subsp. ureolyticus. |
| 15. | Gelatin Hydrolysis | Negative (-) |
| 16. | Pigment Production | Variable |
| 17. | Novobiocin resistance | Susceptible |
| 18. | Lysozyme resistance | Resistant |
| 19. | Bile esculin test | Negative (-) |
| 20. | Growth on 1.5% NaCl | Variable |
Fermentation
| S.N | Substrate | Staphylococcus capitis |
| 1. | Mannitol | Positive (+) |
| 2. | Glucose | Positive (+) May produce only d-lactate or both l- and d-lactate from glucose anaerobically. |
| 3. | Fructose | Positive (+) |
| 4. | Galactose | Negative (-) |
| 5. | Lactose | Variable |
| 6. | Maltose | Negative (-) for S capitis subsp. capitis and Positive (+) for S. capitis subsp. ureolyticus. |
| 7. | Mannose | Positive (+) |
| 8. | Raffinose | Negative (-) |
| 9. | Ribose | Negative (-) |
| 10. | Sucrose | Positive (+) |
| 11. | Starch | Negative (-) |
| 12. | Trehalose | Negative (-) |
| 13. | Xylose | Negative (-) |
| 14. | Salicin | Negative (-) |
| 15. | Glycerol | Positive (+) |
| 16. | Dulcitol | Negative (-) |
| 17. | Cellobiose | Negative (-) |
| 18. | Rhamnose | Negative (-) |
| 19. | Arabinose | Negative (-) |
| 20. | Inulin | Negative (-) |
| 21. | Sorbitol | Negative (-) |
| 22. | Pyruvate | Negative (-) |
Enzymatic Reactions
| S.N | Enzymes | Staphylococcus capitis |
| 1. | Hyaluronidase | Variable |
| 2. | Acetoin | Variable |
| 3. | Alkaline Phosphatase | Variable |
| 4. | Ornithine Decarboxylase | Negative (-) |
| 5. | Pyrrolidonyl aminopeptidase | Positive (+) |
| 6. | β-galactosidase | Negative (-) |
S. capitis subsp. ureolyticus can be distinguished from S. capitis subsp. capitis by the production of urease, the ability to produce acid from maltose, and fatty acid composition.
Virulence factors of Staphylococcus capitis
Coagulase-negative Staphylococci, generally have a lower virulence potential compared to the most virulent staphylococcal species, aureus; however, S. capitis does encode for several factors predicted to be important for biofilm production, persistence, and immune evasion. Besides, S. capitis is also associated with an endopeptidase that functions as a bacteriocin. These virulence factors are mostly involved in protecting the organism against the host immune system and developing resistance against different groups of microbial agents.
1. Biofilm formation
- Biofilm is a multicellular composite of the bacterial cell and extracellular matrix that together provide a barrier to the organism against various immune cells.
- Biofilm formation is regulated by a group of genes that encode for proteins that support the binding of the organism to a surface as well as to each other.
- Following attachment, biofilm accumulation in staphylococci typically occurs by the production of a poly-N-acetlyglucosamine (PNAG) homopolymer, which facilitates intercellular adhesion between the species.
- PNAG production is dependent on the ica locus, which has been recently identified in the genome of S. capitis.
- The plasmin-sensitive protein Pls of S. capitis further promotes cell-cell interaction and allows the process of accumulation of cells to form a biofilm. Pls is considered to share a similar domain structure and sequence homology with the accumulation-associated protein (Aap) of S. epidermidis.
- Proteins, carbohydrates, teichoic acids, and DNA have all been shown to compose staphylococcal biofilms, thus a variety of biofilm-associated genes and operons involved in the four stages of biofilm growth, namely adherence (primary attachment), accumulation, maturation, and dispersal, have been identified in S. capitis as well.
- Biofilm development allows for the deep-sited cells to become more resistant to administered antibiotics and the body’s natural mechanisms, interfering with attempts by the host immune system to clear the infection.
2. Poly-γ-glutamic acid
- Another gene coding for the cap operon has also been observed during the genome analysis of S. capitis, which mediates the production of a second exopolysaccharide, poly-γ-glutamic acid (PGA).
- PGA has an essential role in the pathogenesis of the disease, more specifically, resistance to host antimicrobial peptides and reduced susceptibility to phagocytosis.
3. Endopeptidase ALE-1
- Endopeptidase ALE-1 is 25-kDa, zinc-containing metallopeptidase encoded by a plasmid that also contains the life gene that is responsible for immunity.
- Endopeptidase is synthesized as a preproenzyme, and the N-terminal leader sequence is removed during secretion, while 15 tandem repeats of propeptides, also at the N-terminus, are released by extracellular cysteine peptidase, yielding a fully active enzyme.
- It makes up the Class IIIa staphylococcins comprising of large, heat-labile proteins.
- Endopeptidase has antimicrobial property that hydrolyses glycine-glycine bonds present in the peptidoglycan of different microorganisms.
4. Phenol-soluble Modulins and Exoproteins
- Phenol-soluble modulins (PSM) are secreted amphipathic peptides that appear to have multiple functions in the pathogenicity of S. capitis.
- PSMs are pro-inflammatory and possess cytolytic properties that contribute to biofilm development and even have antimicrobial activity.
- S. capitis also encodes for a suite of exoproteins that likely contribute to infection, which include proteases such as ClpP, which is involved in biofilm formation, and SepA, which degrades host antimicrobial peptides, as well as hemolysins, lipases, and esterases.
- These proteins likely facilitate immune evasion, host colonization, and persistence.
Pathogenesis of Staphylococcus capitis
As S. capitis is a commensal, it rarely causes diseases; however, it is involved in different hospital-acquired infections in people with a compromised immune system. Various proteins, surface-associated adhesins, and extracellular proteins work together as virulence factors to cause such infections. The exact mechanism of infection in the case of S. capitis is not yet fully understood, but it is assumed to similar to other coagulase-negative Staphylococcal species like S. epidermidis and S. lugdunensis. The pathogenesis of infections caused by S. capitis can be described as follows:
1. Attachment/ Adhesin/ Colonization
- Attachment to the host cell surface is the first step in the pathogenesis of S. capitis.
- Cell-surface adhesins like SdrF, SdrG, and SdrH act as fibrinogen-binding molecules that allow attachment to the fibrinogen present on the host cell.
- In a surgical wound, the bacterium utilizes these adhesion mechanisms to adhere to the deeper tissues and the implanted device.
- In the case of medical devices like valve implants and catheters, the surface of the device becomes coated with host-derived plasma proteins, extracellular matrix proteins, and coagulation products (platelets and thrombin) immediately after adhesion by S. capitis.
- Wall teichoic acid further enhances the initial adhesion of S. capitis to medical devices by binding to adsorbed fibronectin.
- The initial attachment is immediately followed by an irreversible attachment that is strengthened by molecule specific adhesins.
- Besides, autolysin encoded by the atlL gene present in the organism is involved in cell separation, stress-induced autolysis which contributes to the process of biofilm formation.
- Poly-γ-glutamic acid, an exopolysaccharide, produced by the organism, provides a decreased susceptibility towards the immune cells and phagocytosis as the polysaccharide binds to the phagocytic cells.
2. Biofilm formation
- Attachment to the cell surface is then followed by the biofilm formation, which is the most important part of pathogenesis by S. capitis.
- Following attachment, biofilm accumulation in staphylococci typically occurs by the production of a poly-N-acetlyglucosamine (PNAG) homopolymer, which facilitates intercellular adhesion between the species.
- PNAG production is dependent on the ica locus, which has been recently identified in the genome of S. capitis.
- The plasmin-sensitive protein Pls of S. capitis further promotes cell-cell interaction and aids in the process of biofilm formation.
- It is further supported by phenol-soluble modulins and exoproteins which are involved in cytolytic activities and help increase the amount of extracellular matrix.
- The biofilm acts as a barrier against different immune cells and surrounding microorganisms.
- It also helps the organism adjust to changing environmental conditions like pH and temperature.
Clinical Manifestations of Staphylococcus capitis
- Even though S capitis has not yet evolved as a pathogenic organism, lately, it has been associated with different nosocomial infections which indicate the ability of the organism to act as an opportunistic pathogen.
- Most of these infections are related to different medical implant devices like artificial valves, catheters, and prosthetic joints.
- Although these are mild infections, if left untreated, this might lead to bacteremia and septic shock.
- Especially in neonates, bacteremia, and sepsis caused by S. capitis are quite common and quite severe.
- Besides, infections like endocarditis, myocarditis, and pericardial effusions might also occur frequently.
- Patients suffering from such infections might prostrate and have a high fever. Hypotension and shock may also occur, caused both by the septic state and by cardiac failure.
- Complications in peripheral organs mainly result from embolic lesions; these may include skin abscesses and retinal emboli, as well as abscesses in the brain and the spleen.
Lab Diagnosis of Staphylococcus capitis
Lab diagnosis of infections caused by S. capitis begins with the collection of samples, which in this case, are scabs, joint aspirates, and pus aspirated from deep sites. The first observation is a direct examination of these samples under the microscope. The diagnosis is mostly related to the identification of the organism; thus, it is mostly centered on the identification of the organism.
1. Cultural characteristics and Biochemical characteristics
- Culturing the organism on different selective media and observing the colony morphology on these media provides a basis for the identification of the organism.
- This also limits the scope of diagnosis and makes the process more feasible.
- Isolation of the organism from primary clinical specimens is achieved on selective culture media like blood agar supplemented with 5 percent sheep blood, following an incubation period of 18–24 h in the air at 35–37°C.
- The isolated colonies are then subjected to different biochemical tests which help in the species determination.
- Depending on the microscopic observation, colony morphology, and biochemical tests, S. capitis can be detected.
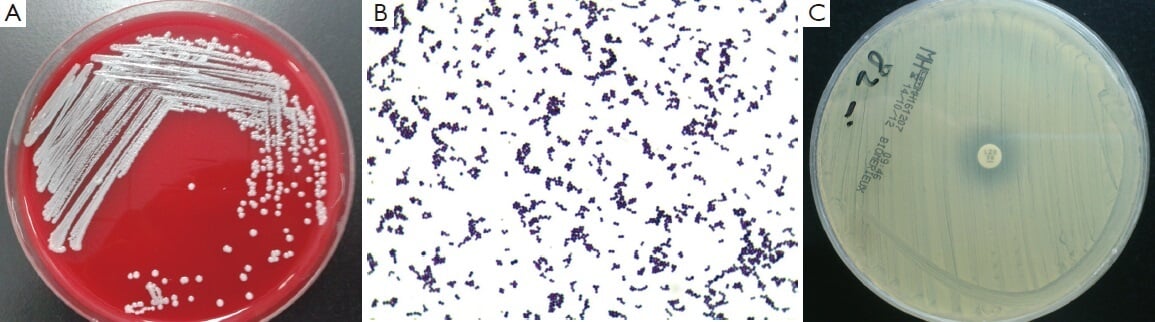
Figure: (A) Isolation of the S. capitis from the blood using blood agar; (B) Morphology of S. capitis under Microscope using Gram stain; (C) S. capitis shown resistance to LZD by K-B method. LZD, Linezolid. Image Source: Bing Gu.
2. Rapid identification kits
- Many clinical laboratories have started to employ different commercial identification kits or automated instruments that allow rapid determination of bacterial species.
- Microbial cellular fatty acid compositions are used for the identification of S. capitis.
- Some of the common automated systems for the Staphylococcal species identification include MicroScan Conventional Pos ID, Rapid Pos ID, and BBL Crystal Gram-Pos ID.
3. Molecular diagnosis
- Molecular methods of diagnosis of bacteria usually include tests that help in the identification of the organism at a molecular level.
- This method utilizes the unique set of nucleic acid sequences present in each organism which provides a more detailed and accurate identification.
- One of the most important molecular methods is Polymerase Chain Reaction (PCR) which helps in the amplification and detection of bacterial DNA.
- Besides, DNA sequencing is performed to determine the DNA sequence of the bacteria that can then be used for its identification.
- Ribotyping is yet another molecular method that involves rRNA restriction fragment polymorphism methods.
Treatment of Staphylococcus capitis
- It is generally assumed that S. capitis responds to nafcillin, cephalosporins, and vancomycin with or without rifampin.
- Cefalotin is the first choice for the treatment of endocarditis caused by S. capitis, but in the case of acute necrosis, daptomycin is used.
- The treatment is comparatively easy as the infections are mostly mild, and most members of S. capitis are susceptible to most antibiotics.
- But, the formation of biofilms increases the resistance of the organism against such drugs.
- New approaches are being adopted to find a new set of antimicrobial targeting different molecules.
Prevention of Staphylococcus capitis
The ability of the organism to form biofilm and cause sepsis in neonates indicates the need for preventive strategies to be adopted against S. capitis. The following are some preventive strategies that can be followed to avoid infections by S. capitis:
- Coating of biomaterials on the medical devices prevents the attachment of the organism to such devices.
- Periodic cleaning of wounds and regular dressing should also be employed to avoid infections in those areas.
- A clean hospital environment is imperative to prevent nosocomial infections.
References
- Topley WWC (2007). Topley and Wison’s Microbiology and Microbial Interactions; Bacteriology, 2 Vol. Tenth Edition. John Wiley and Sons Ltd.
- Bergey, D. H., Whitman, W. B., De, V. P., Garrity, G. M., & Jones, D. (2009). Bergey’s manual of systematic bacteriology: Vol. 3. New York: Springer.
- Soumya, K. R., Philip, S., Sugathan, S., Mathew, J., & Radhakrishnan, E. K. (2017). Virulence factors associated with Coagulase Negative Staphylococci isolated from human infections. 3 Biotech, 7(2), 140. https://doi.org/10.1007/s13205-017-0753-2
- Tevell, S., Hellmark, B., Nilsdotter-Augustinsson, Å. et al.Staphylococcus capitis isolated from prosthetic joint infections. Eur J Clin Microbiol Infect Dis 36, 115–122 (2017). https://doi.org/10.1007/s10096-016-2777-7
- Cui, B., Smooker, P. M., Rouch, D. A., Daley, A. J., & Deighton, M. A. (2013). Differences between two clinical Staphylococcus capitis subspecies as revealed by biofilm, antibiotic resistance, and pulsed-field gel electrophoresis profiling. Journal of clinical microbiology, 51(1), 9–14. https://doi.org/10.1128/JCM.05124-11
- Wirth, T., Bergot, M., Rasigade, J. et al.Niche specialization and spread of Staphylococcus capitis involved in neonatal sepsis. Nat Microbiol 5, 735–745 (2020). https://doi.org/10.1038/s41564-020-0676-2
- Paweł Mak. Chapter 13 – Staphylococcal Bacteriocins. Pet-To-Man Travelling Staphylococci. Academic Press. 2018. Pages 161-171. ISBN 9780128135471. https://doi.org/10.1016/B978-0-12-813547-1.00013-3.
- TAMMY L. BANNERMAN AND WESLEY E. KLOOS. Staphylococcus capitis subsp. ureolyticus subsp. nov. from Human Skin. INTERNATIONAL JOURNAL OF SYSTEMATIC BACTERIOLOGY, Jan. 1991, p. 144-147.
- Greco-Stewart et al. Biofilm formation by Staphylococcus capitis strains isolated from contaminated platelet concentrates. Journal of Medical Microbiology (2013), 62, 1051–1059.
- https://www.tgw1916.net/Staphylococcus/capitis.html
