Transcriptomics is the field of science that deals with the study of the transcriptome, which is the collection of all RNA transcripts produced within specific cells of an organism.
Transcriptomics includes the study of everything related to RNA, including its sequences, structure, functions, locations, transcription, translation, expression levels, and degradation. High-throughput methods, such as microarray analysis and RNA sequencing, are used to study these RNA transcripts.

Transcriptomics is important because it includes the study of all mRNA transcripts in a cell, which reflects actively expressed genes at any given time. By studying RNA transcripts, transcriptomics provides information about how genetic information is expressed, how proteins are produced, and how the cell functions. This makes transcriptomics important for understanding how genetic information leads to cellular activities.
The field of transcriptomics has evolved along with other fields of omics including genomics, proteomics, and metabolomics, becoming an important part of modern biological research. New technologies in transcriptomics have made it easier to study RNA transcripts and their functions in different biological processes.
Historical Development of Transcriptomics
- Before the development of transcriptomics, individual RNA transcripts were studied using labor-intensive methods such as northern blotting and Sanger sequencing. Although these techniques were popular in the 1990s, they could only be used to study a tiny subsection of transcriptome and were time-consuming.
- The term “transcriptome” was first used in the 1990s. Initial attempts to study whole transcriptomes began in 1991 with the publication of a partial human transcriptome from the human brain.
- The early 1990s saw the development of Expressed Sequence Tag (EST) sequencing using Sanger sequencing.
- By the mid-1990s, techniques like serial analysis of gene expression (SAGE) and microarrays developed, allowing the study of thousands of transcripts simultaneously.
- RNA-Seq, developed in the mid-2000s, further advanced the field by allowing high-throughput sequencing of entire transcripts. Next-generation sequencing (NGS) platforms like Illumina transformed the field of transcriptomics.
- These advancements have made it possible to study transcriptomes of different tissues, diseases, and even single cells, contributing to our understanding of gene regulation and cellular processes.
Types of Transcriptomics
There are different types of transcriptomics. The major types are:
1. Bulk Transcriptomics
- Bulk transcriptomics studies gene expression in bulk samples containing thousands of cells and provides a collective gene expression profile of the entire population of cells. This method helps us to understand the gene expression patterns in a large group of cells.
- It can be used to study thousands of genes simultaneously and provides quantitative data on gene expression levels. However, it has certain limitations such as the inability to study cell diversity and identify rare cell types.
- While bulk transcriptomics includes several methods, RNA sequencing remains the primary method to study bulk transcriptomes.
2. Single-Cell Transcriptomics
- Single-cell transcriptomics is used to study the gene expression of individual cells.
- In contrast to bulk transcriptomics which studies gene expression in bulk samples, single-cell transcriptomics measures gene expression at the individual cell level and is useful to study the cellular diversity.
- Multicellular organisms contain different types of cells, each with different transcriptional profiles. So, the ability to study gene expression levels of individual cells is important to understand the different cell types and biological processes.
- Single-cell transcriptomics is used to study cellular diversity and rare cell populations that may be overlooked in bulk transcriptomics. It also allows the discovery of novel cell types.
3. Spatial Transcriptomics
- Spatial transcriptomics measures gene expression in a sample while preserving spatial information. It is important to study the level of gene expression along with their spatial distribution to understand the function and structural organization of genes.
- Bulk transcriptomics and single-cell transcriptomics do not provide spatial information as they destroy the structural organization of cells.
- Spatial transcriptomics technologies preserve spatial information and provide valuable information about the organization, interactions, and functions of cells.
- Spatial transcriptomics can be divided into imaging-based and sequencing-based technologies. Both methods are used to study transcriptomics while preserving spatial information.

4. Meta Transcriptomics
- Meta transcriptomics studies RNA transcripts and the gene expression of an entire community of organisms including microorganisms, plants, animals, and other eukaryotes.
- Meta transcriptomics initially focused on the study of microbial communities only. However, it is not limited to microbes and includes other organisms as well.
- This method is useful in studying the functions and interactions of organisms within their natural environment.
- Meta transcriptomics is commonly used in environmental studies and the study of complex ecosystems such as microbiomes in gut, soil, and water samples.
Methods Used in Transcriptomics
There are different methods for transcriptome analysis. The most widely used ones are:
1. Expressed Sequence Tag (EST) Sequencing
- ESTs are short nucleotide sequences produced from a single RNA transcript.
- The process of EST sequencing begins by converting RNA into complementary DNA (cDNA) using reverse transcription, followed by sequencing the cDNA.
- The Sanger sequencing method was the primary sequencing technique used for ESTs until the development of high-throughput methods.
- In recent years, high-throughput and large data output have made RNA-Seq the dominant method and replaced conventional EST sequencing. Although modern high-throughput methods have largely replaced them, EST libraries played an important role in early microarray designs.
2. Serial Analysis of Gene Expression (SAGE)
- SAGE was one of the first methods used for whole transcriptome analysis using sequencing.
- SAGE is a technique that identifies and quantifies transcripts present in a biological sample by generating short sequence tags from mRNA.
- The main principle of SAGE is to convert mRNA into short, unique sequence tags that correspond to specific transcripts. These tags are then concatenated and sequenced, which identifies the gene expression levels.
- SAGE involves isolating mRNA from the biological sample which is converted into cDNA using reverse transcription. Restriction enzymes cleave the cDNA into small fragments, which are then ligated to oligonucleotide adapters. These fragments are further cleaved to produce short tags, which are then ligated together to form ditags. The ditags are concatenated into long chains, cloned into a vector for amplification, and sequenced. The resulting sequences are analyzed to identify and quantify the original mRNA transcripts.
- SAGE provides quantitative transcriptome analysis and is useful in discovering novel genes. However, there are several limitations of this technique including the difficulties in identifying unknown genes due to the short length of tags, specificity issues due to overlapping sequences, and potential incompatibilities with some restriction enzymes.
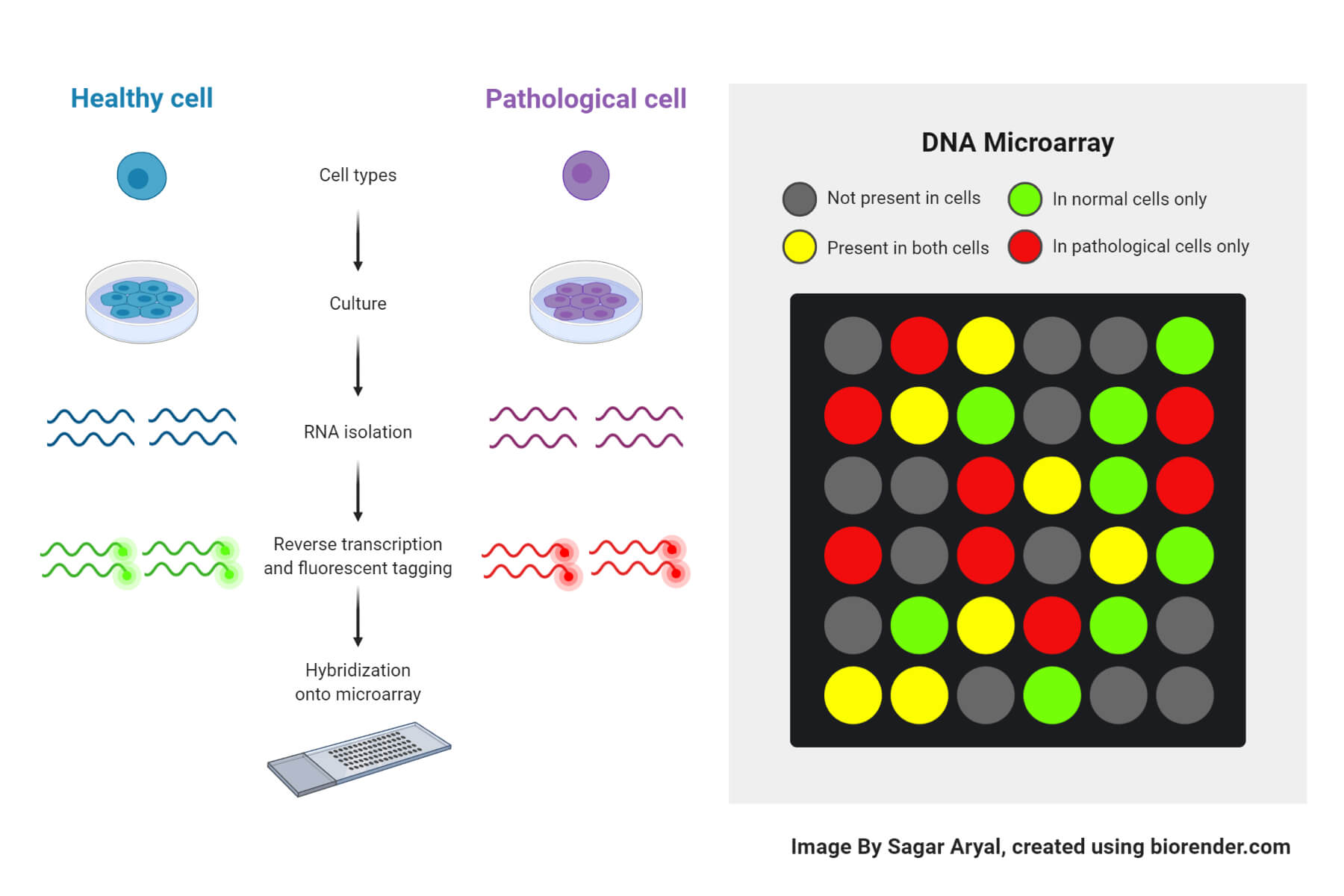
3. Microarrays
- A microarray consists of short nucleotide oligomers, called DNA probes, which are arranged on a solid surface like a glass slide that quantifies gene expression using hybridization.
- At first, RNA is isolated from the target samples which is reverse transcribed into cDNA and labeled with fluorescent dyes. The labeled cDNA is hybridized to the microarray. After washing, the array is scanned with a laser. Probes that hybridize to transcribed RNA emit light, and the intensity of this light is used to measure gene expression levels.
- Despite its ability to study gene expression and the transcriptome, the results from microarrays can vary significantly due to differences in laboratory procedures, laser scanners, fluorescent readouts, and analytical methods. Therefore, microarray-based mRNA expression analysis should be used as a preliminary step and should be validated using other methods including RT-PCR.
- Microarrays are significantly cheaper than RNA sequencing. The data generated by microarrays are smaller and easier to manage. However, this method may not effectively detect genes with very low expression levels and new transcripts.
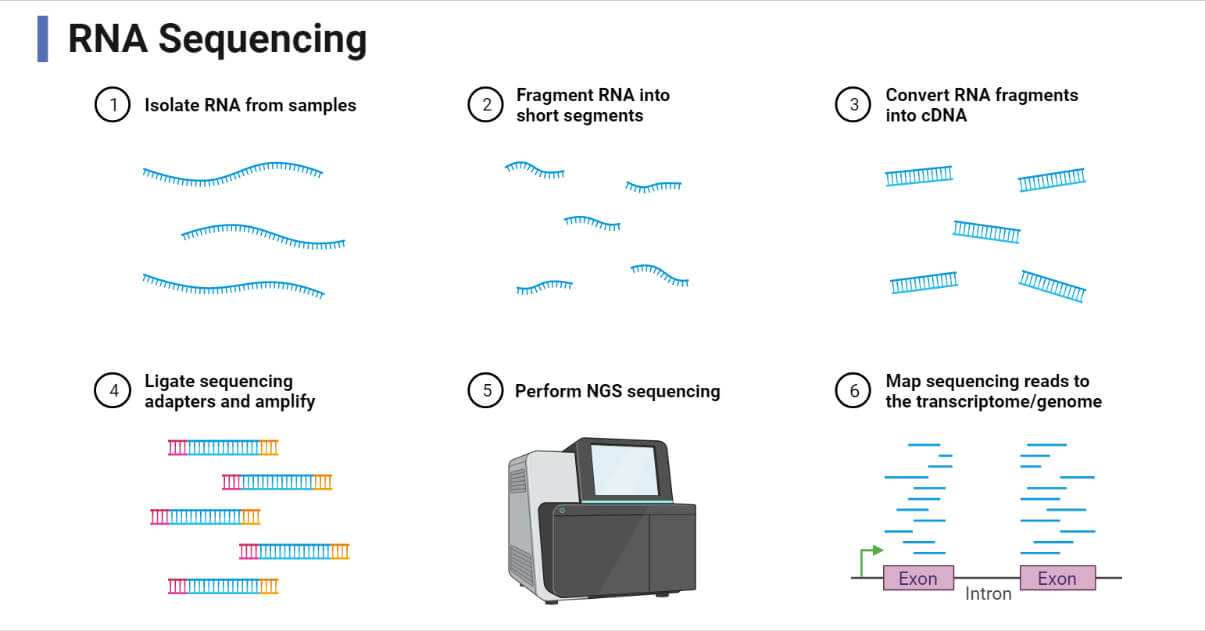
RNA Sequencing (RNA-Seq)
- RNA-Seq combines high-throughput sequencing with computational methods to quantify RNA transcripts. This novel technology is based on next-generation sequencing (NGS) platforms which are highly automated.
- RNA-Seq allows simultaneous sequencing of thousands of different RNA molecules in a single run, unlike classical sequencing methods such as Sanger’s method.
- The RNA-Seq process begins with the extraction of mRNA and converting it into stable cDNA. The cDNA fragments are sequenced using high-throughput methods. The raw sequencing data are then processed to generate a counts table. This data is used to annotate gene expression levels. Various online tools are available for data processing.
- RNA-Seq can quantify a wide range of transcripts, including novel and low-abundance transcripts. However, RNA-Seq is much more expensive than microarrays, making it less accessible for large-scale studies. RNA-Seq also generates large amounts of data that require advanced bioinformatics skills and computational resources.
Applications of Transcriptomics
- Transcriptomics has applications in disease diagnosis and profiling. RNA-Seq methods help in identifying regulatory elements like transcriptional start sites which are important elements in human disease.
- Transcriptomics is useful in understanding the molecular mechanisms of different diseases and also provides insight into different potential therapeutic agents.
- Transcriptomics is used in pharmacogenomics to study the response of individuals to specific drugs and is useful in personalized medicine.
- Transcriptomics is also useful in understanding host-pathogen immune interactions and studying immune responses.
- Transcriptomics is also used in developmental biology to study gene expression during different developmental stages.
- Transcriptomics is also used to study noncoding RNAs, which play important roles in different cellular processes.
Video on Transcriptomics
References
- Bashiardes, S., Zilberman-Schapira, G., & Elinav, E. (2016). Use of Metatranscriptomics in Microbiome Research. Bioinformatics and biology insights, 10, 19–25. https://doi.org/10.4137/BBI.S34610
- Dong, Z., Chen, Y. Transcriptomics: Advances and approaches. Sci. China Life Sci. 56, 960–967 (2013). https://doi.org/10.1007/s11427-013-4557-2
- Exploring Transcriptomics: Techniques and Applications – DrOmics Labs
- Kanter, I., & Kalisky, T. (2015). Single cell transcriptomics: methods and applications. Frontiers in oncology, 5, 53. https://doi.org/10.3389/fonc.2015.00053
- Khodadadian, A., Darzi, S., Haghi-Daredeh, S., Sadat Eshaghi, F., Babakhanzadeh, E., Mirabutalebi, S. H., & Nazari, M. (2020). Genomics and Transcriptomics: The Powerful Technologies in Precision Medicine. International journal of general medicine, 13, 627–640. https://doi.org/10.2147/IJGM.S249970
- Kukurba, K. R., & Montgomery, S. B. (2015). RNA Sequencing and Analysis. Cold Spring Harbor protocols, 2015(11), 951–969. https://doi.org/10.1101/pdb.top084970
- Li, X., Wang, CY. From bulk, single-cell to spatial RNA sequencing. Int J Oral Sci 13, 36 (2021). https://doi.org/10.1038/s41368-021-00146-0
- Lowe, R., Shirley, N., Bleackley, M., Dolan, S., & Shafee, T. (2017). Transcriptomics technologies. PLoS computational biology, 13(5), e1005457. https://doi.org/10.1371/journal.pcbi.1005457
- Malone, J.H., Oliver, B. Microarrays, deep sequencing and the true measure of the transcriptome. BMC Biol 9, 34 (2011). https://doi.org/10.1186/1741-7007-9-34
- Supplitt, S., Karpinski, P., Sasiadek, M., & Laczmanska, I. (2021). Current Achievements and Applications of Transcriptomics in Personalized Cancer Medicine. International journal of molecular sciences, 22(3), 1422. https://doi.org/10.3390/ijms22031422
- Transcriptomics | Transcriptome analysis (illumina.com)
- Wang, Y., Liu, B., Zhao, G., Lee, Y., Buzdin, A., Mu, X., Zhao, J., Chen, H., & Li, X. (2023). Spatial transcriptomics: Technologies, applications and experimental considerations. Genomics, 115(5), 110671. https://doi.org/10.1016/j.ygeno.2023.110671
- What is transcriptomics? – PHG Foundation

Hii there Shilpi this side currently pursuing Masters in Molecular Biology and Biotechnology. In my research I have transcriptomic related work. Just now started going through your work and it really seems good and easy to understand. Hoping to learn the same in both theoretical and practical ways. Thank you
wonderful description
very good information