What is Bordetella pertussis?
Bordetella pertussis is a pathogenic Gram-negative aerobic bacterium that is the causative agent of the disease pertussis or whooping cough.
- B. pertussis is the type species of Bordetella that consists of minute mesophilic coccobacillus that facilitates the colonization of animal tissues.
- B. pertussis is one of the eight species belonging to the genus Bordetella and one of the seven pathogenic species, most of which are associated with respiratory infections in humans and other animals.
- These are obligate pathogens of humans colonizing the ciliary epithelial cells of the respiratory tract. These are rarely present in the environment except in the form of aerosols and droplets, which is also the mode of transmission of the disease.
- It is an important pathogenic species of the genus Bordetella characterized by different virulence factors that enable infections.
- B. pertussis is a fastidious, slow-growing organism causing infections only in humans. The disease in humans is the result of excellent colonization and mesophilic growth.
- The infection in humans by B. pertussis is similar to infections caused by B. parapertussis in sheep as well as humans.
- Most Bordetella species are isolated from warm-blooded animals like humans which are mostly present in the respiratory tract of the host. However, recently these microorganisms can be isolated from other parts like human blood and human wound cultures.
- The bacterium was first isolated by Bergey, Harrison, Breed, Hammer, and Huntoon in 1923 and it was named Haemophilus pertussis. It was later renamed Bordetella pertussis in 1952 by Moreno-Lopez.
- The species name ‘pertussis’ is taken from two Latin words; ‘per’ meaning very and ‘tussis’ meaning cough, representing the severe cough caused by the bacterium.
- Pertussis is a severe respiratory infection that affects both the upper and lower respiratory tract of humans and is particularly severe in infants of developing countries.
- There have been major improvements in the reduction of pertussis by the development and improvement of vaccines over the years. However, the disease continues to be an issue in various countries with more health systems.
- B. pertussis is a Biosafety Level 2 pathogen that should be handled by following specific guidelines and regulations.
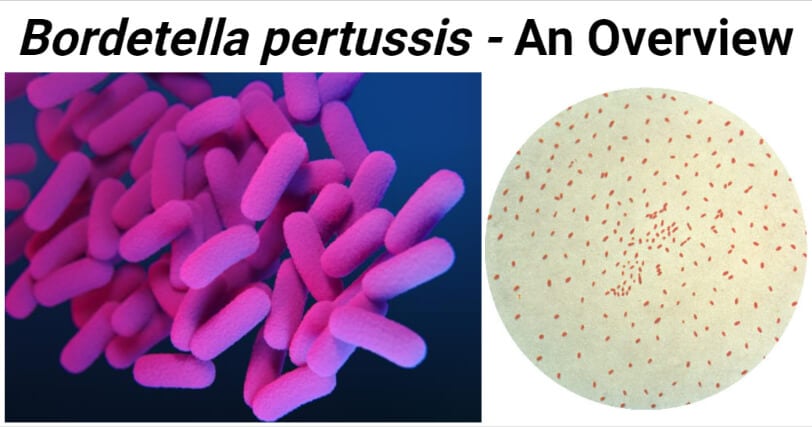
Figure: Left- Medical illustration of drug-resistant, Bordetella pertussis bacteria. Image Source: CDC-Antibiotic Resistance Coordination and Strategy Unit. Right-A photomicrograph of a culture specimen processed using the Gram staining method, and reveals the presence of Gram-negative, Bordetella pertussis bacteria. Image Source: CDC.
Classification of Bordetella pertussis
- The genus Bordetella consists of minute coccobacilli that are obligate pathogens of humans and animals characterized by the 16S rRNA gene sequences. Before the designation of the bacteria into a separate genus, the species were placed in the Haemophilus pertussis.
- The genus belongs to the family Alcaligenaceae of the Betaproteobacteria that is closely associated with Achromobacter and Alcaligens of the family.
- The members of the family Alcaligenaceae are characterized by their DNA-DNA hybridization and 16S rRNA gene sequence analyses. Besides, these are differentiated by ubiquinone-8 as the major isoprenoid quinine.
- The differentiation of Bordetella from other genera is the occurrence of Bordetella from warm-blooded animals, whereas other genera occur mostly in soil, water, and hospital environments.
- The genus consists of seven different species based on the analysis of phenotypic characteristics, DNA base composition, and nucleic acid hybridization.
- B. pertussis is the type species of Bordetella along with two other species B. parapertussis and B. bronchiseptica that are respiratory pathogens of mammals with an economic impact on both human health as well as agriculture.
- These species were even considered the subspecies of the same species which was later differentiated as different species based on the genotypic evaluation.
- These bacteria are grouped as B. bronchoseptica clusters as they have similar genome size and pathogenic niche.
The following is the taxonomical classification of B. pertussis;
| Domain | Bacteria |
| Phylum | Proteobacteria |
| Class | Betaproteobacteria |
| Order | Burkholderiales |
| Family | Alcaligenaceae |
| Genus | Bordetella |
| Species | B. pertussis |
Habitat of Bordetella pertussis
- The primary habitat of B. pertussis is the ciliary epithelium present in the respiratory tract of human beings. The occurrence, however, has been found in the respiratory tract of animals.
- It is a strict aerobic pathogen that doesn’t occur as an inhabitant of the animal body but is transmitted from other infected individuals.
- B. pertussis has no known reservoir other than humans, and the bacteria are assumed to transmit directly from one infected individual to another.
- The bacteria does, however, survive in the air forming aerosols or droplets through which it transmits from one host to the other.
- The ability of the bacteria to survive in the human by evading the immune system of the host and with excellent colonization characteristics. All of these attributed to the diverse and highly efficient virulence factors of the bacteria.
- Like most pathogenic bacteria, B. pertussis also expresses sets of gene products in response to changes in the varied environment.
- The long-term survival within the host and in the environment depends on the synthesis of necessary factors at different concentrations and times as a response to the directed stimuli.
- B. pertussis has adapted as the strict pathogen of the human body regulated by different products that act both as antimicrobial agents against other pathogens and as the virulence factors.
Morphology of Bordetella pertussis
- The morphology of Bordetella species is quite distinct from other pathogenic species which allows easier differentiation based on their morphological characteristics.
- The cells of B. pertussis are Gram-negative minute coccobacilli ranging in size between 0.2-0.5 µm × 0.5-2.0 µm.
- The cells are occasionally filamentous that can elongate several µm in length, usually observed in clinical samples.
- B. pertussis is non-motile with no flagella which are used to differentiate the bacteria from other species like B. bronchoseptica and B. avium.
- The cells are either encapsulated or surrounded by a sheath of slime. The capsule is usually observed in freshly isolated species whereas the slime formation occurs in vitro in the form of biofilm.
- Both the capsule and slime sheath are composed of polysaccharides encoded by genes, but the conditions of their expression are not known.
- The surface of the cell consists of fine filamentous appendages that can be observed in the form of filaments and membranous vesicles in the culture supernate. The fimbriae are 3-5 mm in width and 110-250 nm in length.
- As Bordetella species are Gram-negative, thus they contain an outer membrane and an inner membrane, in between which is the cell wall.
- The outer membrane is composed of lipopolysaccharides linked by long sugar units and anchored by lipid units.
- The lipopolysaccharide of B. pertussis is different from that of other Gram-negative bacteria with different phosphate composition than the lipid A in other bacteria.
- The lipopolysaccharide of B. pertussis acts as endotoxins that are toxic to the host and thus, acts as a virulence factor.
- The cell wall is composed of a single layer of peptidoglycan consisting of glucose derivatives; N-acetyl glucosamine and N-acetylmuramic acid with pentapeptide linkages.
- The cell membrane is a typical bacteria cell membrane with the lipid bilayer and protein particles embedded in the lipid pool.
- The chromosome of B. pertussis is circular with 4124236 bp and 3456 protein-coding gene sequences. The average G+C content of B. pertussis is 67.3%.
Cultural Characteristics of Bordetella pertussis
- Bordetella pertussis is the most fastidious species among all Bordetella species which requires about 3-6 days for the development of colonies on artificial media.
- The medium of choice for the selective isolation of B. pertussis is the Bordet-Gengou medium with glycerol. The medium is composed of a potato-extract medium without peptone containing 50% blood.
- The charcoal horse blood agar is a better medium for the selective cultivation of B. pertussis as it has a longer shelf-life and is superior in its ability to support B. pertussis growth.
- For routine analysis, sheep or horse blood agar is used, but the blood content is reduced to 15-20%, which facilitates the observation of hemolysis.
- Commercial media for B. pertussis include Stainer-Scholte broth and cyclodextrin solid medium.
- B. pertussis is an obligate aerobe with the most efficient growth at the temperature range of 30-37°C.
- The metabolism is mostly based on the oxidation of amino acids as these bacteria do not usually utilize carbohydrates.
- The growth of B. pertussis on artificial media is difficult due to the susceptibility of the bacteria to various compounds like unsaturated fatty acids, colloidal sulfur, and sulfides.
- The selective medium used for the cultivation of B. pertussis contains adsorbents for the inhibitors.
- The variation during growth is a common feature of all the species of the genus Bordetella where cultures usually exhibit different colony types even in the same agar surface.
- Variation in colony morphology on a repeated subculture with subsequent loss of antigens and changes in growth requirements is more common in B. pertussis than other Bordetella species.
- The change in morphology is attributed to the combined processes of serotype variation, antigenic modulation, and phase variation.
- Different liquid media may be used for the bulk growth of B. pertussis that is free of blood or tissue extracts.
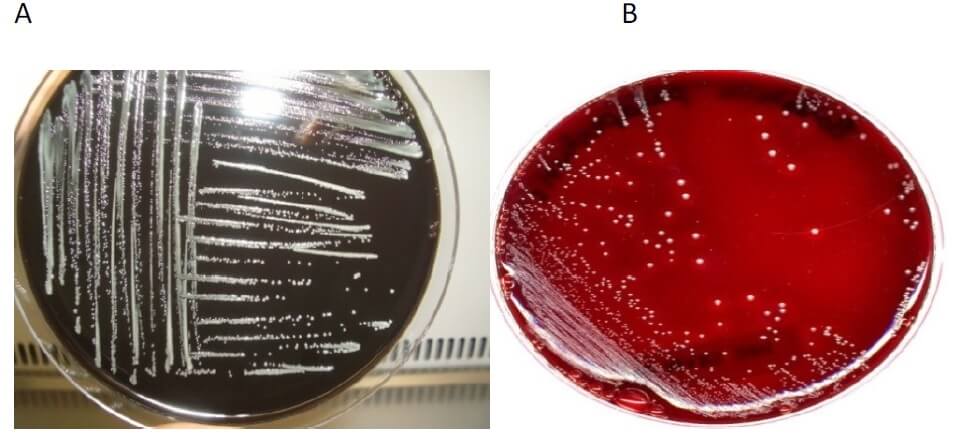
Figure: A- Bordetella pertussis on Reagan Lowe (RL) medium. B- Bordetella pertussis on Bordet Gengou (BG) medium. Image Source: WHO.
The following are some cultural characteristics of B. pertussis on different culture media:
a. Bordet-Gengou test
- The colonies of B. pertussis on Bordet-Gengou are small and pinpoint as the colony diameter rarely exceeds 3mm. The size of the colonies might increase up to 4 mm after 5-6 days in some strains.
- Bordetella pertussis is a fastidious organism that can be cultivated on an artificial medium after about 3-6 days of incubation.
- The medium is a selective medium as it contains compounds that support the growth of bacteria against different inhibitors that might be present in general media.
- The colonies exhibit a characteristic narrow zone of hemolysis visible around the colonies. The type of hemolysis observed in β-hemolysis.
- The colonies are convex and glistening with an entire edge, that usually dries out after prolonged incubation.
- After subsequent subcultures, variation in colony morphology might be observed. Colonies of B. pertussis that are flat, non-hemolytic can be observed in some cases.
b. Charcoal Blood Agar (Regan-Lowe Agar)
- Charcoal blood agar supplemented with horse blood is used for the enrichment of Bordetella species and its selective isolation. The medium contains cephalexin that inhibits the growth of normal flora of the nasopharynx.
- The colonies on the agar appear small, grey, shiny, shiny, and smooth. These have a pearl-like luster that appears like a mercury droplet.
- The size of the colonies is small and lies between 1-2 mm and might exceed up to 3 mm after 5-6 days of incubation.
Biochemical Characteristics of Bordetella pertussis
The biochemical characteristics of B. pertussis can be tabulated as follows:
| S.N | Biochemical Characteristics | B. pertussis |
| 1. | Capsule | Capsulated |
| 2. | Shape | Coccobacilli |
| 3. | Gram Staining | Gram-Negative |
| 4. | Catalase | Positive (+) |
| 5. | Oxidase | Positive (+) |
| 6. | Citrate | Negative (+) |
| 7. | Methyl Red (MR) | Negative (-) |
| 8. | Voges Proskauer (VR) | Negative (+) |
| 9. | OF (Oxidative-Fermentative) | Oxidative |
| 10. | Coagulase | Negative (-) |
| 11. | DNase | Negative (-) |
| 12. | Urease | Negative (-) |
| 13. | Gas | Negative (-) |
| 14. | H2S | Negative (-) |
| 15. | Hemolysis | β-hemolytic |
| 16. | Motility | Non-motile |
| 17. | Nitrate Reduction | Negative (-) |
| 18. | Gelatin Hydrolysis | Negative (-) |
| 19. | Pigment Production | Positive (+) (Brown) |
| 20. | Indole | Negative (-) |
| 21. | TSIA (Triple Sugar Iron Agar) | Alkali/Alkali (Red/ Red) |
| 22. | Spore | Non-sporing |
Fermentation
| S.N | Substrate | P. pertussis |
| 1. | Adonitol | Negative (-) |
| 2. | Arabinose | Negative (-) |
| 3. | Cellobiose | Negative (-) |
| 4. | Dulcitol | Negative (-) |
| 5. | Fructose | Negative (-) |
| 6. | Galactose | Negative (-) |
| 7. | Glucose | Negative (-) |
| 8. | Glycerol | Negative (-) |
| 9. | Glycogen | Negative (-) |
| 10. | Hippurate | Negative (-) |
| 11. | Inulin | Negative (-) |
| 12. | Inositol | Negative (-) |
| 13. | Lactose | Negative (-) |
| 14. | Malonate | Negative (-) |
| 15. | Maltose | Negative (-) |
| 16. | Mannitol | Negative (-) |
| 17. | Mannose | Negative (-) |
| 18. | Pyruvate | Negative (-) |
| 19. | Raffinose | Negative (-) |
| 20. | Rhamnose | Negative (-) |
| 21. | Ribose | Negative (-) |
| 22. | Salicin | Negative (-) |
| 23. | Sorbitol | Negative (-) |
| 24. | Starch | Negative (-) |
| 25. | Sucrose | Negative (-) |
| 26. | Trehalose | Negative (-) |
| 27 | Xylose | Negative (-) |
Enzymatic Reactions
| S.N | Enzymes | P. pertussis |
| 1. | Acetoin | Negative (-) |
| 2. | Acetate Utilization | Negative (-) |
| 3. | Alkaline phosphatase | Negative (-) |
| 4. | Esculin Hydrolysis | Negative (-) |
| 5. | Lecithinase | Negative (-) |
| 6. | Lipase C14 activity | Negative (-) |
| 7. | Lysine Decarboxylase | Negative (-) |
| 8. | Ester C8 lipase activity | Positive (+) |
| 9. | Naphthol-AS-B1 phosphohydrolase | Positive (+) |
| 10. | Trypsin | Negative (-) |
Virulence Factors of Bordetella pertussis
Bordetella pertussis is an obligate pathogen that has adapted to the environments in the human body by means of various virulence-related factors. These factors include structures, products as well as genes that regulate the progression of disease in the host. It produces an array of toxins and adhesins that are important in the colonization of hosts while protecting them against the host immune system.
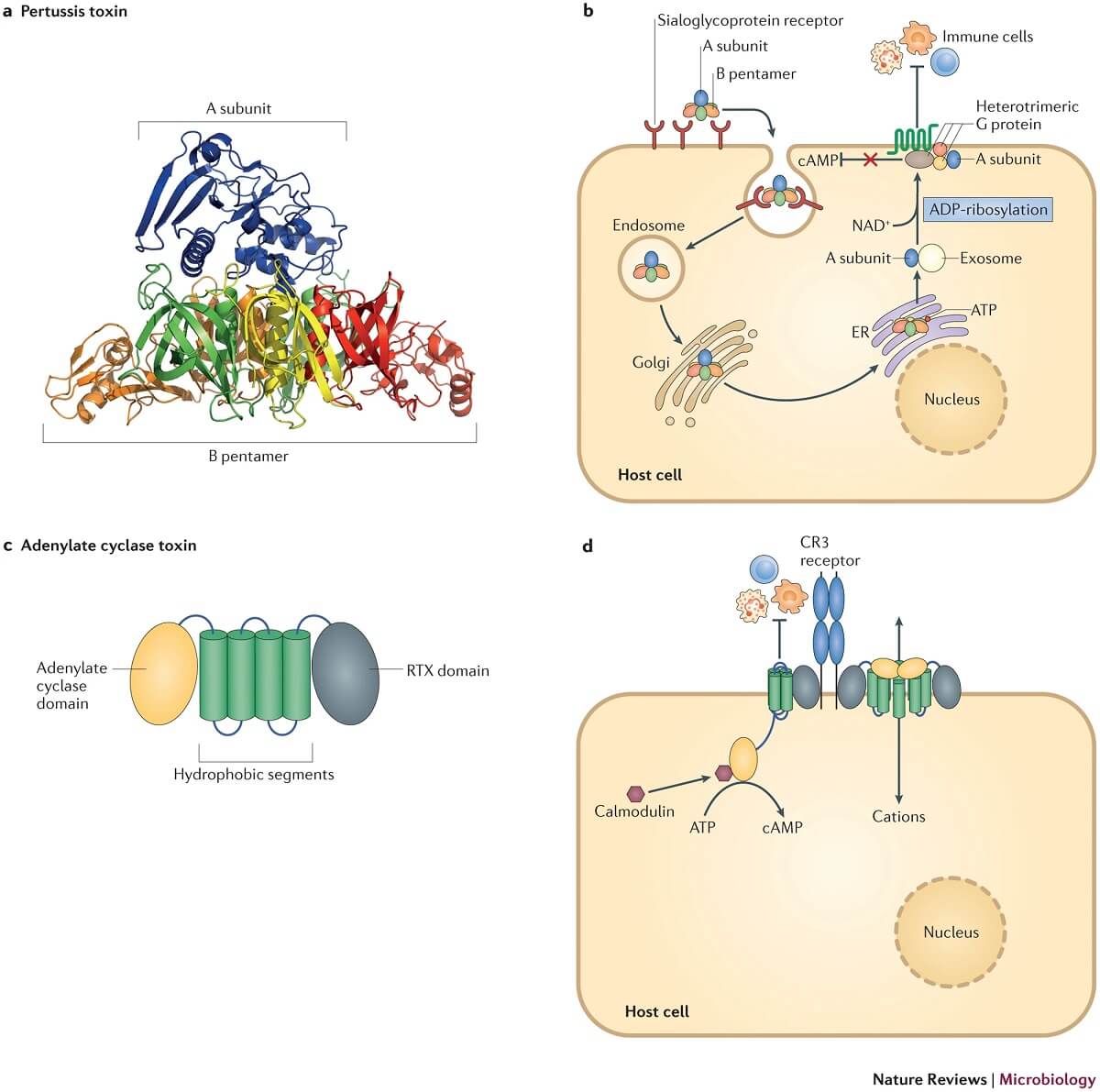
Figure: Toxin-mediated virulence of Bordetella spp. Image Source: Nature Reviews Microbiology.
The following are the common virulence factors associated with infections caused by B. pertussis and its pathogenesis;
a. Filamentous haemagglutinin
- Filamentous haemagglutinin is a cell-surface-associated protein that is secreted extracellularly into the surrounding environment.
- The protein plays a significant role in the initial attachment of B. pertussis to the ciliary epithelium of the respiratory tract in humans. It also acts as a bridge adhesion that facilitates the binding to other microorganisms.
- The protein has three different binding sites and two main immune dominant regions that are highly immunogenic.
- The haemagglutinin is secreted in the form of its precursor FhaB which requires a special protease that is essential for the processing of the precursor into the mature protein.
- The binding of B. pertussis to the ciliated epithelium is mediated by the carbohydrate-binding domain of the protein that has a unique affinity for glycolipids and ciliated cells.
- The protein also has the heparin-binding activity, which allows the attachment of the bacteria to non-ciliated cells.
- Filamentous haemagglutinin also binds to phagocytic cells like macrophages which helps the bacteria escape the host immune system.
- It also binds to a serum protein which is the regulator of the complement activation. The binding, thus inhibits the classical complement pathway ceasing the formation of the membrane attack complex.
b. Fimbriae
- Fimbriae are filamentous, cell surface-associated polymeric protrusions that extend from the bacterial cell surface and facilitate the colonization of the respiratory tract.
- B. pertussis produces serologically distinct fimbriae that are composed of two major subunits Fim2 and Fim3, respectively.
- The major subunit forms the long filamentous structure characteristic of the fimbriae while the minor subunit is present on the tip of the structure.
- The major subunits bind to chondroitin sulfate, heparin sulfate, and dextran sulfate that are present in the respiratory tract.
- The minor subunit enables the binding of the bacteria to cells via their cell-surface integrin VLA-5, which then significantly enhances the haemagglutinin-mediated attachment.
- Fimbriae are involved in the initial attachment of the bacteria to the laryngeal mucosa and are also essential for the formation of biofilm during infections.
c. Pertactin
- Pertactin is an outer membrane protein that is also involved in bacterial adherence to the host cell surface.
- The mechanism employed by B. pertussis during adherence is not yet known, and no receptor involved in the process have been discovered yet.
- However, it is known that the amino acid sequence of the bacteria reveals an RGD motif which is a known integrin-binding moiety in other bacterial adhesins like filamentous haemagglutinin.
- There have been studies that have indicated that the protein also helps in providing resistance to clearance of bacteria from the lungs.
- Even though the exact role of the pertactin in pathogenicity is not known, it is considered an important protective immunogen that has even been used in the preparation of vaccines.
d. Tracheal cytotoxin
- A tracheal cytotoxin is a part of the peptidoglycan component of the bacterial cell wall which damages the ciliated respiratory epithelial cells.
- The destruction of cilia and the epithelial cells result in ciliostasis and forces the infected individual to cough relentlessly to remove mucus.
- It is a disaccharide that is similar to the monomeric subunit of the bacteria peptidoglycan produced during the growth of the cell envelope.
- The release of tracheal cytotoxin results in an exaggerated host response in the form of the release of inflammatory mediators like IL-1 and cytokine-inducible NO synthase.
- Tracheal cytotoxin also has a toxic effect on other cells, reducing neutrophil function at low concentrations and inducing toxic activity at higher concentrations.
e. Dermonecrotic toxin
- Dermonecrotic toxin is a heat-labile toxin that is inactivated at the temperature of 56°C and causes necrotic skin lesions when injected subcutaneously.
- The toxin catalyzes the polyamination or deamidation of small Rho family GTPases. The enzymes are essential in the reorganization of the actin skeleton and cell motility.
- Even though this toxin is the first toxin of Bordetella species to be defined, not much is known about the toxin due to the difficulty in the purification process.
- It has a potent vasoconstrictive activity which can cause death or weight loss due to ischemic lesion or necrosis of the skin.
- The necrotizing effect of the toxin is due to the specific constrictive effect on the vascular smooth muscle. The effect on the muscle of the respiratory tract can induce a local inflammatory reaction, which accounts for some of the pathology of the disease.
f. Pertussis toxin
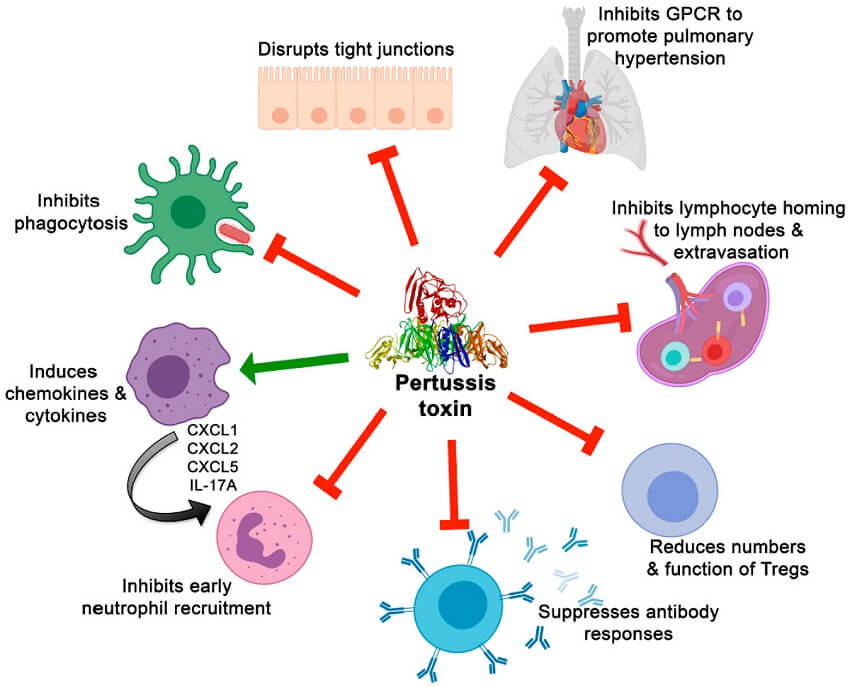
Figure: Effects of PT on immune cells and responses and on other aspects of pertussis pathogenesis. Image Source: MDPI Toxins.
- Pertussis toxin is an important virulence factor and a protective antigen exclusive to B. pertussis.
- It is an endotoxin that is transported across the bacterial outer membrane by a type IV secretion system.
- The toxin is an AB5 type toxin consisting of one active subunit and five binding B oligomers.
- The A subunit in the cytosol of the host cell catalyzes the transfer of released ADP-ribose from the hydrolyzed cellular NAD+ to a family of G proteins.
- These proteins are involved in signal transduction pathways within the host cell, and the binding of ADP-ribose disturbs the cell signaling machinery of epithelial and immune system cells.
- The B oligomers of the toxin facilitate the attachment of the toxin to the host cell, required for the entry of A subunit of the toxin into the cytosol.
- The toxin also upregulates the function of the macrophage integrin CR3 and thus, promotes attachment and intake of the bacteria with the help of filamentous haemagglutinin.
- The most important targets of the toxin are the phagocytic cells where it limits the migration of the cells to the site of infection in the respiratory tract and reduces their phagocytic potential.
g. Adenylate cyclase toxin
- Adenylate cyclase toxin (ACT) is an immunomodulatory toxin that functions as both a hemolysin as well as a cytotoxin.
- The toxin is a polypeptide with two domains where one of the domains mediates the binding of the toxin to target cells and translocation of the toxin to the cytosol.
- The toxin mediates adherence to the host cell by binding to the αmβ2 integrin present on myeloid phagocytic and natural killer cells.
- Adenylate cyclase is activated by calmodulin and catalyzes the conversion of intracellular ATP to cAMP that inhibits superoxide generation, immune effector cell chemotaxis, and phagocytosis.
- ACT is a bi-functional virulence factor involved in the early stage of infection and can elicit a protective immune response.
- In the eukaryotic cell, ACT causes unregulated synthesis of cAMP from ATP, resulting in interference with normal cell functioning.
- ACT also induces apoptosis in target cells, which are primarily phagocytic cells.
h. Lipopolysaccharides/ Endotoxin
- The lipopolysaccharide of B. pertussis is smaller than that of other gram-negative bacteria and is often termed lipooligosaccharide.
- In B. pertussis, lipopolysaccharide is involved in whooping cough syndrome by inducing NO production in infected tracheal cells which results in damage of the respiratory ciliated cells.
- The lipopolysaccharide induces endotoxic activity which may act synergistically with the tracheal cytotoxin.
- The nitric oxide produced destroys iron-dependent enzymes and inhibits mitochondrial function and DNA synthesis in the host cells.
Pathogenesis of Bordetella pertussis
The exact mechanism of pathogenesis of B. pertussis in humans is not yet known, but studies have been conducted both in vitro and in vivo in animal models. Humans are the only reservoirs of B. pertussis, and thus the transmission of bacteria occurs only from one infected individual to another. The pathogenesis of B. pertussis is complex and multifactorial where the loss of a single toxin and adhesin might affect the ability of the bacteria to colonize the host surfaces.
Figure: Summary of infection of the respiratory tract by B. pertussis and its immunomodulatory effects. Image Source: https://doi.org/10.1016/j.coph.2006.12.004.
The overall pathogenesis of B. pertussis can be described in the following steps;
a. Entry and Colonization
- B. pertussis transmits from the infected individual to a healthy host in the form of droplets.
- The bacteria then binds to the epithelial cells of the upper respiratory tract. The primary target cells are the ciliated epithelium of the respiratory tract.
- The initial attachment of bacteria to ciliated cells results in loss of both ciliated and non-ciliated cells and increased secretion of mucus.
- The colonization of the host tissue occurs due to the synergistic action of pertussis toxin, filamentous haemagglutinin, and fimbriae.
- The carbohydrate-binding domain of the haemagglutinin has a higher affinity with the glycolipids present in the cilia.
- The attachment is further enhanced by fimbriae which attaches to the integrin protein found at the cilia tip.
- The bacteria also has different factors that work to escape from the immune response of the host by either binding to the phagocytic cells or by releasing toxins targeted at the phagocytic cells.
b. Toxin production
- The colonization of the respiratory tract by B. pertussis is followed by the production of different toxins which paralyze the attached cells as well as invades the immune cells.
- Toxins like tracheal cytotoxin, pertussis toxin, and adenylate cyclase toxin are released into the host cell which causes paralysis of the cell either by destructing the process of cytoskeleton formation.
- These toxins also induce the secretion of different cytokine stimulating compounds like IL-1 and NO synthase.
- Dermonecrotin toxin results in vasoconstrictive action on the smooth muscle of the respiratory tract, which induces local inflammation.
- The adenylate cyclase toxin causes an unregulated synthesis of cAMP into ATP which interferes with normal cell functioning.
c. Immune response
- The first cells of the innate immune system that respond to the initial colonization of B. pertussis are resident and infiltrating macrophages and immature dendritic cells.
- These cells are activated by the secretion of cytokines induced by the virulence factors of the bacteria.
- Even though most of the bacterial cells are destroyed within acidic compartments, but a large portion of the bacteria are capable of evading destruction and replicating within macrophages by residing in the non-acidic compartments.
- B. pertussis destroys macrophages by enhancing the activation of cytokines and inflammatory stimulatory compounds like IFN-γ and interleukin-17.
- B. pertussis cells are also taken up within subcellular structures in neutrophils that undergo lysosomal maturation.
- Neutrophil function is inhibited by adenylate cyclase toxin by decreasing phagocytosis, superoxide generation, and inhibiting chemotaxis.
- In addition to the innate immune system, humoral immunity is also activated by bacteria. The infection induces IgG and IgA, which is detectable in the serum after about five days of incubation.
Clinical Manifestation of Bordetella pertussis
- Bordetella pertussis is an obligate pathogen that causes respiratory infections in humans, especially in neonates and children.
- The infection caused by B. pertussis is called pertussis or whooping cough, which is characterized by severe infection of the respiratory tract.
- Pertussis is characterized by bronchopneumonia, paroxysmal coughing, and the distinctive whopping intake of air.
- The duration of illness in the case of pertussis typically ranges between 6 to 12 weeks, but sometimes, it might be longer. The incubation period for pertussis is typically 7 to 10 days.
- The infection occurs as a three-stage illness; catarrhal, paroxysmal, and convalescent.
- The catarrhal phase is a non-specific mild clinical condition that begins immediately after the incubation period for up to 2 weeks.
- During this phase, common symptoms like nasal congestion, rhinorrhea, lacrimation, malaise, and mild sore throat are observed.
- The phase is followed by a worsening cough, which marks the beginning of the paroxysmal stage of the disease.
- During the paroxysmal stage, the respiratory secretions of the patient are highly infectious, but the transmission is the most efficient during the first three weeks as the cough onset.
- The stage is characterized by a repetitive series of 5 to 10 forceful coughs during a single expiration followed by a sudden inspiratory effort resulting in a whoop.
- As the illness progresses, episodes of cough usually increase in frequency and severity at night.
- The final stage of the infection is the convalescent stage which lasts about 1 to 12 weeks characterized by decreasing frequency and severity of coughing episodes, whooping, and vomiting.
- The convalescent stage is the recovery period during which superimposed viral respiratory infections can trigger a recurrence of paroxysms.
- The clinical features of the disease differ with age, general health, and the immune status of the patient.
- Other common complications of pertussis include insomnia, apnea, weight loss, and urinary tract infections. The less common manifestation include pneumonia, otitis medua, and rare death.
Lab Diagnosis of Bordetella pertussis
The diagnosis of pertussis is difficult as the symptoms of the disease are similar to other mild infections like common cough or cough. The most important and useful markers that can be used as indicators of the disease are a mild increase in the leukocyte count and marked lymphocytosis. The clinical definition of the disease requires one or more typical symptoms that include inspiratory whoop and paroxysmal cough for about a week. Diagnosis of pertussis can be obtained either through the traditional method of culture and serological testing or by the use of genomic analysis to identify the organism accurately.
a. Sample collection
- The samples commonly used for the diagnosis of pertussis are nasopharyngeal aspirate or nasopharyngeal swabs.
- The sensitivity of diagnosis depends on the technique of swab collection and secretions. The swab should be introduced deeply into the nose to reach the nasopharynx.
- The swabs used should be made of dacron or calcium alginate, which should then be cultured on half-strength charcoal blood agar.
- In the case of secretion, these should be collected with a suction device with a mucus trap. The secretions are then plated on the Charcoal agar or Bordet-Gengou medium.
b. Microscopic, Cultural and Biochemical characteristics
- Culture is the gold standard for the diagnosis of pertussis; thus, the samples obtained from the patients are cultured on appropriate media to observe the cultural characteristics of the organism.
- Two distinct media can be used as the media of choice for the culture of B. pertussis, which are Bordet-Gengou and Regan-Lowe agars.
- To this agar, cephalexin is added to inhibit the growth of contaminating bacteria like Pseudomonas and Streptococcus.
- The identification of the species is made on the basis of cultural characteristics like colony morphology and culture conditions for growth.
- Biochemically, the bacteria can be distinguished on the basis of tests like oxidase, urease, citrate utilization, and nitrate reduction.
c. Direct fluorescent-antibody assay
- The direct fluorescent antibody assay is usually performed directly on the samples for the microscopic visualization of fluorescent antibodies directed towards B. pertussis cells.
- The assay is performed on nasopharyngeal samples, but the sensitivity and specificity of the method are relatively low.
- The assay should be supported by other methods like culture, PCR, and serological testing.
d. Serological Testing
- Serological diagnosis of B. pertussis is based on the identification and variation of IgG or IgA against different virulence factors of B. pertussis during the acute and convalescent phases of the disease.
- The antigens often targeted during the serological testing of B. pertussis are pertussis toxin, filamentous haemagglutinin, and pertactin.
- The ability to produce antibodies may be affected by the disease history of immunization and disease.
e. PCR analysis
- Diagnosis of pertussis by PCR assays is an established method for the detection and identification of the causative agent of pertussis.
- PCR methods have the advantage of being more specific and sensitive than the conventional method of culture and biochemical testing.
- There are various primers from different chromosomal regions of the bacteria that have been developed for the diagnosis of B. pertussis.
- Some common issues associated with the identification of B. pertussis by PCR methods are the possibility of false-positive results and the difficulty of the process.
Treatment of Bordetella pertussis infections
- The treatment of pertussis can be obtained either through the use of antibiotic therapy or by vaccine immunization.
- The early treatment of the disease with recommended antimicrobials is useful in the prevention of harmful complications and the reduction of the duration of the disease.
- The first choice for the treatment of pertussis has been the administration of oral erythromycin. The administration reduces the symptoms it used during the early course of illness.
- The dose of the antibiotics for children is 40-50 mg/kg given every 6 hours for 14 days.
- Azithromycin is a newer antibiotic for treatment which is used 10 mg/kg on day 1 and 5 mg/kg on day 2 to 5 as a single dose.
- Other recommended method of treatment of B. pertussis includes antibiotics like azithromycin erythromycin and trimethoprim-sulfamethoxazole.
- Even though erythromycin resistance, macrolide resistance, and fluoroquinolone resistance have been rarely found, the resistance is still rare.
Prevention of Bordetella pertussis infections
- The most important and efficient method of prevention pertussis is by the administration of vaccines that are either whole-cell vaccines or acellular vaccines.
- The first whole-cell vaccines were discovered in the 1920s, and effective vaccination started in the late 1940s.
- The whole-cell pertussis vaccines are produced from the smooth forms of the bacteria, which might result in some local and systemic side effects.
- Due to the adverse side effects associated with whole-cell pertussis component vaccines, a new acellular vaccine has been developed.
- Besides, other methods like the use of masks and while maintaining appropriate distance with the infected individuals.
References for Bordetella pertussis
- Topley W. W. C (2007). Topley and Wison’s Microbiology and Microbial Interactions; Bacteriology, 2 Vol. Tenth Edition. John Wiley and Sons Ltd.
- Bergey, D. H., Whitman, W. B., De, V. P., Garrity, G. M., & Jones, D. (2009). Bergey’s manual of systematic bacteriology: Vol. 2. New York: Springer.
- Coote JG. Environmental sensing mechanisms in Bordetella. Adv Microb Physiol. 2001; 44:141-81. doi: 10.1016/s0065-2911(01)44013-6. PMID: 11407112.
- Hulbert RR, Cotter PA. Laboratory Maintenance of Bordetella pertussis. Curr Protoc Microbiol. 2009 Nov;Chapter 4:Unit 4B.1. doi: 10.1002/9780471729259.mc04b01s15. PMID: 19885941.
- Carbonetti NH. Bordetella pertussis: new concepts in pathogenesis and treatment. Curr Opin Infect Dis. 2016 Jun;29(3):287-94. doi: 10.1097/QCO.0000000000000264. PMID: 26906206; PMCID: PMC4846492.
- Nieves DJ, Heininger U. Bordetella pertussis. Microbiol Spectr. 2016 Jun;4(3). doi: 10.1128/microbiolspec.EI10-0008-2015. PMID: 27337481.
- Hoppe, J E, and J Schwaderer. “Comparison of four charcoal media for the isolation of Bordetella pertussis.” Journal of clinical microbiology vol. 27,5 (1989): 1097-8. doi:10.1128/JCM.27.5.1097-1098.1989
- SUTHERLAND, I W. “The protective activity of components of Bordetella pertussis cell walls.” Immunology vol. 6,3 (1963): 246-54.
- Weiss A. (2006) The Genus Bordetella. In: Dworkin M., Falkow S., Rosenberg E., Schleifer KH., Stackebrandt E. (eds) The Prokaryotes. Springer, New York, NY. https://doi.org/10.1007/0-387-30745-1_27
- Smith AM, Guzmán CA, Walker MJ. The virulence factors of Bordetella pertussis: a matter of control. FEMS Microbiol Rev. 2001 May;25(3):309-33. doi: 10.1111/j.1574-6976.2001.tb00580.x. PMID: 11348687.
- Adam M. Smith, Carlos A. Guzmán, Mark J. Walker, The virulence factors of Bordetella pertussis: a matter of control, FEMS Microbiology Reviews, Volume 25, Issue 3, May 2001, Pages 309–333, https://doi.org/10.1111/j.1574-6976.2001.tb00580.x
- Finger H, von Koenig CHW. Bordetella. In: Baron S, editor. Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston; 1996. Chapter 31. Available from: https://www.ncbi.nlm.nih.gov/books/NBK7813/
- Dorji, D., Mooi, F., Yantorno, O. et al. Bordetella pertussis virulence factors in the continuing evolution of whooping cough vaccines for improved performance. Med Microbiol Immunol 207, 3–26 (2018). https://doi.org/10.1007/s00430-017-0524-z
- van den Berg, B M et al. “Role of Bordetella pertussis virulence factors in adherence to epithelial cell lines derived from the human respiratory tract.” Infection and immunity vol. 67,3 (1999): 1056-62. doi:10.1128/IAI.67.3.1056-1062.1999
- Kilgore, Paul E et al. “Pertussis: Microbiology, Disease, Treatment, and Prevention.” Clinical microbiology reviews vol. 29,3 (2016): 449-86. doi:10.1128/CMR.00083-15
- Melvin, Jeffrey A et al. “Bordetella pertussis pathogenesis: current and future challenges.” Nature reviews. Microbiology vol. 12,4 (2014): 274-88. doi:10.1038/nrmicro3235
- Solans, Luis, and Camille Locht. “The Role of Mucosal Immunity in Pertussis.” Frontiers in immunology vol. 9 3068. 14 Jan. 2019, doi:10.3389/fimmu.2018.03068
- Higgs, R., Higgins, S., Ross, P. et al. Immunity to the respiratory pathogen Bordetella pertussis. Mucosal Immunol 5, 485–500 (2012). https://doi.org/10.1038/mi.2012.54.

It is very most important notes I have no believe how can explain overall character of B pertussis full description really I like it thanks you so much heart ????
I am reading your all posts. its really awesome…..
This is my website – https://biologyideas.com/