The innate immune system provides the first line of host defense against microbes, before adaptive immune responses have had sufficient time to develop. The mechanisms of innate immunity exist before exposure to microbes.
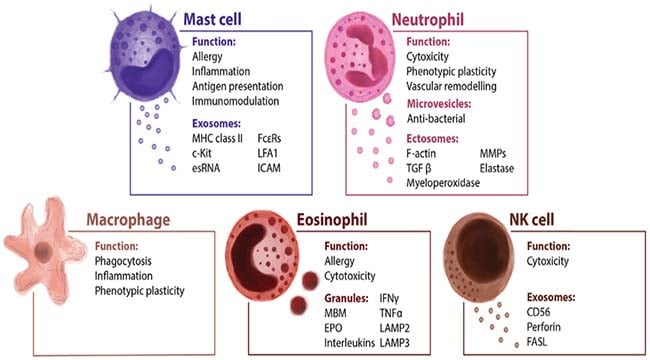
- The cellular components of the innate immune system include epithelial barriers and leukocytes (neutrophils, macrophages, NK cells, lymphocytes with invariant antigen receptors, and mast cells).
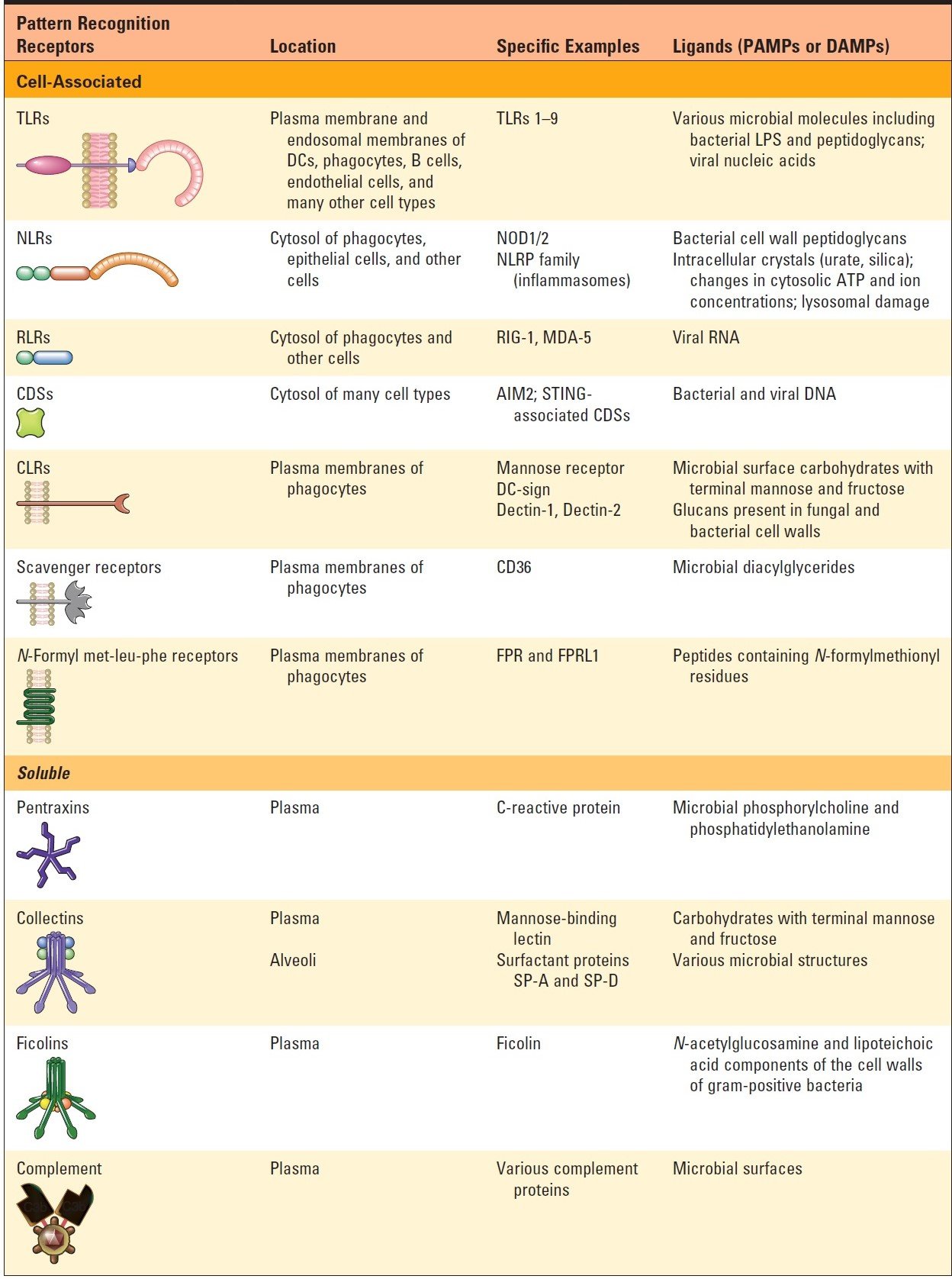
- The innate immune system uses cell-associated pattern recognition receptors, present on plasma and endosomal membranes and in the cytosol, to recognize structures called pathogen-associated molecular patterns (PAMPs), which are shared by microbes, are not present on mammalian cells, and are often essential for survival of the microbes, thus limiting the capacity of microbes to evade detection by mutating or losing expression of these molecules.
- In addition, these receptors recognize molecules made by the host but whose expression or location indicates cellular damage; these are called DAMPs (Damage-associated molecular pattern molecules).
- Toll-like receptors (TLRs), present on the cell surface and in endosomes, are the most important family of pattern recognition receptors, recognizing a wide variety of ligands, including bacterial cell wall components and microbial nucleic acids.
- Cytosolic pattern recognition receptors exist that recognize microbial molecules.
- These receptors include the RIG-I-like receptors (RLRs), which recognize viral RNA, CDSs which recognize microbial DNA, and NOD-like receptors (NLRs), which recognize bacterial cell wall constituents and also serve as recognition components of many inflammasomes.
- Pattern recognition receptors, including TLRs, NLRs, and RLRs, signal to activate the transcription factors NF-κB and AP-1, which stimulate expression of cytokines, costimulators, and other molecules involved in inflammation, and the IRF transcription factors, which stimulate expression of the antiviral type I interferon genes.
- The inflammasome, a specialized caspase-1 containing enzyme complex that forms in response to a wide variety of PAMPs and DAMPs, includes recognition structures, which are often NLR family proteins, an adaptor, and the enzyme caspase-1, the main function of which is to produce active forms of the inflammatory cytokines IL-1 and IL-18.
- Soluble pattern recognition and effector molecules are found in the plasma, including pentraxins (e.g., CRP), collectins (e.g., MBL), and ficolins.
- These molecules bind microbial ligands and enhance clearance by complement-dependent and complement-independent mechanisms.
- Innate lymphoid cells are cells with lymphocyte morphology and functions similar to T lymphocytes, but do not express clonally distributed T cell antigen receptors.
- Three helper subsets of ILCs secrete the same cytokines as Th1, Th2, and Th17 helper T cells.
Complement System
- The complement system includes several plasma proteins that become activated in sequence by proteolytic cleavage to generate fragments of the C3 and C5 proteins, which promote inflammation, or opsonize and promote phagocytosis of microbes.
- Complement activation also generates membrane pores that kill some types of bacteria.
- The complement system is activated on microbial surfaces and not on normal host cells, because microbes lack regulatory proteins that inhibit complement.
- In innate immune responses, complement is activated mainly spontaneously on microbial cell surfaces and by mannose-binding lectin to initiate the alternative and lectin pathways, respectively.
- The two major effector functions of innate immunity are to induce inflammation, which involves the delivery of microbe-killing leukocytes and soluble effector molecules from blood into tissues, and to block viral infection of cells by the antiviral actions of type 1 interferons.
- Both types of effector mechanism are induced by the PAMPs and DAMPs.
Cytokines
- Several cytokines produced mainly by macrophages, DCs, and other innate immune cells mediate inflammation.
- TNF and IL-1 activate endothelial cells, stimulate chemokine production, and increase neutrophil production in the bone marrow.
- IL-1 and TNF both induce IL-6 production, and all three cytokines mediate systemic effects, including fever and acute-phase protein synthesis by the liver.
- IL-12 and IL-18 stimulate production of the macrophage-activating cytokine IFN-γ by NK cells and T cells.
- These cytokines function in innate immune responses to different classes of microbes, and some (IL-1, IL-6, IL-12, IL-18) modify adaptive immune responses that follow the innate immune response.
NK Cells
- NK cells are one type of innate lymphoid cells that have cytotoxic functions and secrete FIN-γ, similar to CTLs.
- NK cells defend against intracellular microbes by killing infected cells and providing a source of the macrophage-activating cytokine IFN-γ.
- NK cell recognition of infected cells is regulated by a combination of activating and inhibitory receptors.
- Inhibitory receptors recognize class I MHC molecules, because of which NK cells do not kill normal host cells but do kill cells in which class I MHC expression is reduced, such as virus-infected cells.
Neutrophils
- Neutrophils and monocytes (the precursors of tissue macrophages) migrate from blood into inflammatory sites during innate immune responses because of the effects of cytokines and chemokines produced by PAMP- and DAMP-stimulated tissue cells.
- Neutrophils and macrophages phagocytose microbes and kill them by producing ROS, nitric oxide, and enzymes in phagolysosomes.
Macrophages
- Macrophages also produce cytokines that stimulate inflammation and promote tissue repair at sites of infection.
- Phagocytes recognize and respond to microbial products by several different types of receptors, including TLRs, C-type lectins, scavenger receptors, and N-formyl met-leu-phe receptors.
- Molecules produced during innate immune responses stimulate adaptive immunity and influence the nature of adaptive immune responses.
Dendritic Cells (DCs)
- DCs activated by microbes produce cytokines and costimulators that enhance T cell activation and differentiation into effector T cells.
- Complement fragments generated by the alternative pathway provide second signals for B cell activation and
antibody production. - Innate immune responses are regulated by negative feedback mechanisms that limit potential damage to tissues.
- IL-10 is a cytokine that is produced by and inhibits activation of macrophages and DCs.
- Inflammatory cytokine secretion is regulated by autophagy gene products.
- Negative signaling pathways block the activating signals generated by pattern recognition receptors and inflammatory cytokines.

A great note on innate immunity. It will be perfect if you can also introduce innate lymphoid cells (i.e. ILC1, ILC2 and ILC3).
Wonderful man.. U r so young nd dynamic.. I’m a Passionate Virologist nd teaching in vet school, Vet Microbiology.. Ur notes are so useful..
Thank you veru much