- Carbohydrates are molecules that contain carbon, hydrogen, and oxygen.
- There are twice as many hydrogen atoms as carbon or oxygen atoms.
- The general formula for a carbohydrate can be written as Cx(H2O)y.
- They act as the source of energy (e.g. glucose), as a store of energy (e.g. starch and glycogen) and as structural units (e.g. cellulose in plants and chitins in insects).
- Most carbohydrates are polymers.
- Polymers are large, complex molecules composed of long chains of monomers.
- Monomers are small, basic molecular units.
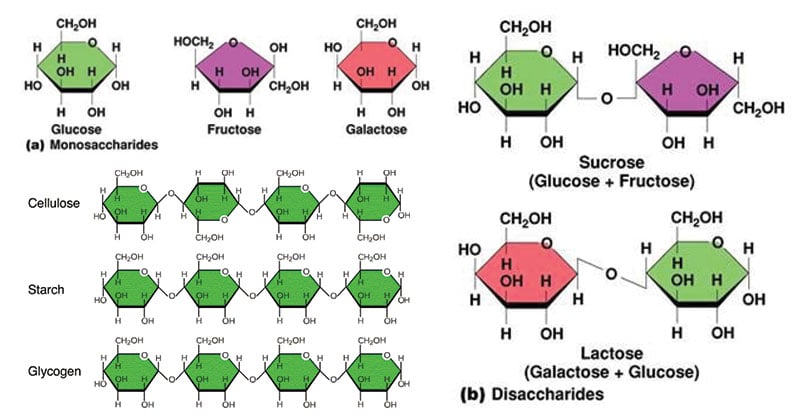
- Carbohydrates can be divided into three groups: monosaccharides, disaccharides, and polysaccharides.
Monosaccharides – Structure, Properties, and Examples
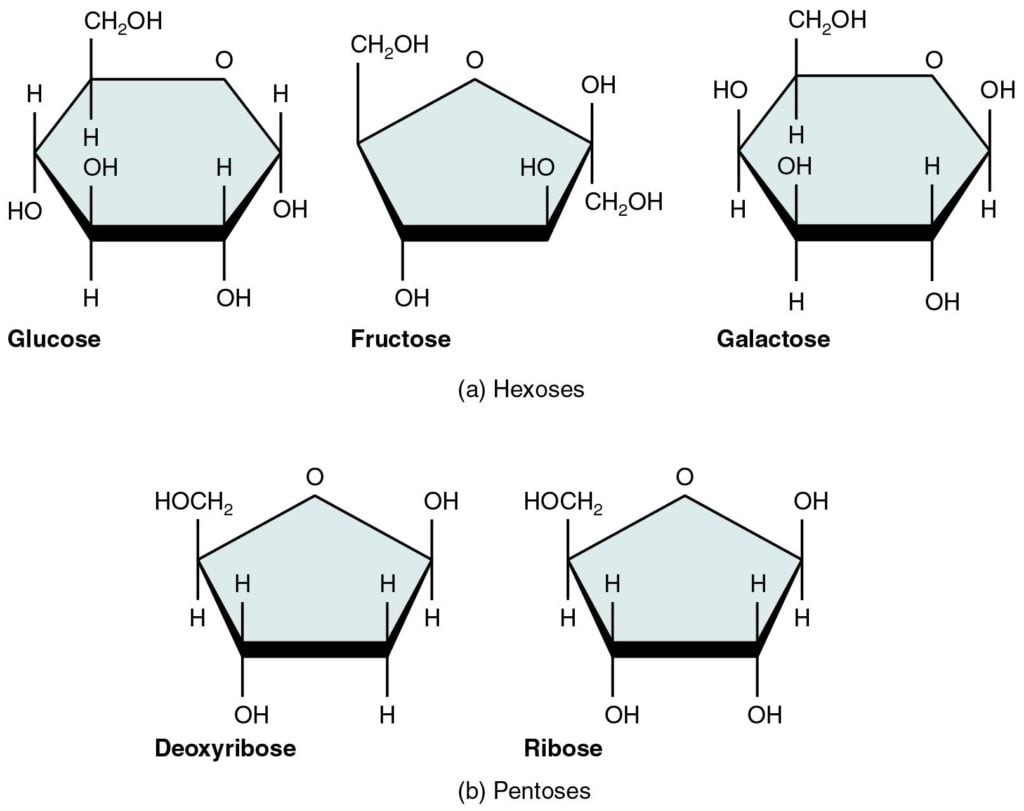
- Monosaccharides are simple sugars in which there are one oxygen atom and two hydrogen atoms for each carbon atom present in the molecule.
- They have general formula as (CH2O)n.
- Monosaccharides are reducing sugars.
- The test for reducing sugar is called Benedict’s test.
- They are sugars, which taste sweet, are soluble in water and are insoluble in non-polar solvents.
- They exist in straight chains or in the ring or cyclic forms.
- They are classified according to the number of carbon atoms in each molecule as trioses (3C), tetroses (4C), pentoses (5C), hexoses (6C), heptoses (7) and so on.
- The names of all sugars end with -ose.
- Examples: Glyceraldehyde (triose), Erythrose (tetrose), Ribose (pentose), Glucose (hexose), Fructose (hexose), Galactose (hexose), Sedoheptulose (heptose), etc.
- They are used as a source of energy in respiration.
- They are important building blocks for large molecules.
Disaccharides – Structure, Properties, and Examples
- Disaccharides are made up of two monosaccharides joined together by a condensation reaction.
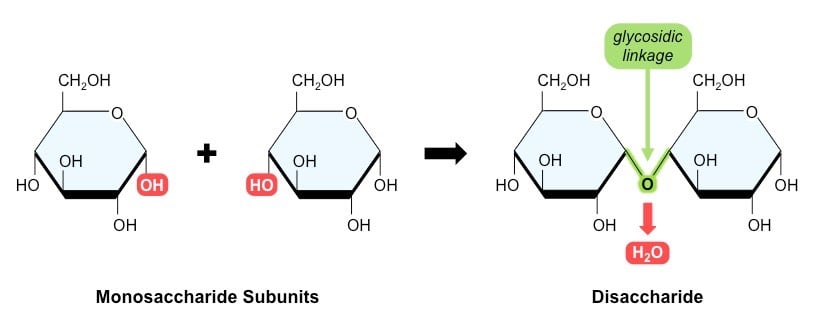
- The condensation reaction is the joining of two molecules with the formation of a new chemical bond and a water molecule is released when the bond is formed.
- A glycosidic bond is formed between two monosaccharides. If carbon 1 on one monosaccharide joins to carbon 4 on another monosaccharide, it is called a 1,4-glycosidic bond.
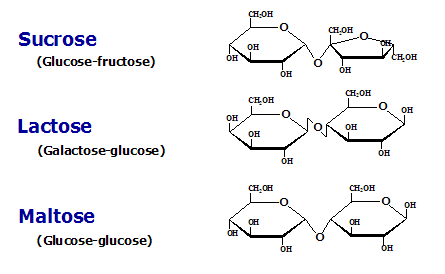
- Examples: Maltose is formed from two α-glucose molecules joined together by a glycosidic bond. Sucrose is formed from a condensation reaction between a glucose molecule and a fructose molecule. Lactose is formed from glucose and a galactose molecule.
- Sucrose is a non-reducing sugar.
- Disaccharides can be split apart into two monosaccharides by breaking the glycosidic bond by adding water molecules, which is known as hydrolysis reaction. The water provides a hydroxyl group (-OH) and hydrogen (-H), which helps the glycosidic bond to break.
- Sucrose is the transport sugar and Lactose is the sugar found in milk which an important constituent of the diet of young mammals.
Polysaccharides – Structure, Properties, and Examples
- Polysaccharides are polymers formed by combining many monosaccharide molecules (more than two) by condensation reactions.
- Molecules with 3-10 sugar units are known as oligosaccharides while molecules containing 11 or more monosaccharides are true polysaccharides.
- Polysaccharides do not taste sweet.
- Because their molecules are so enormous, the majority of polysaccharides do not dissolve in water.
- Polysaccharides made solely from one kind of monosaccharides are called homopolysaccharides (Starch) while those made of more than one monomer are called heteropolysaccharides (Hyaluronic acid).
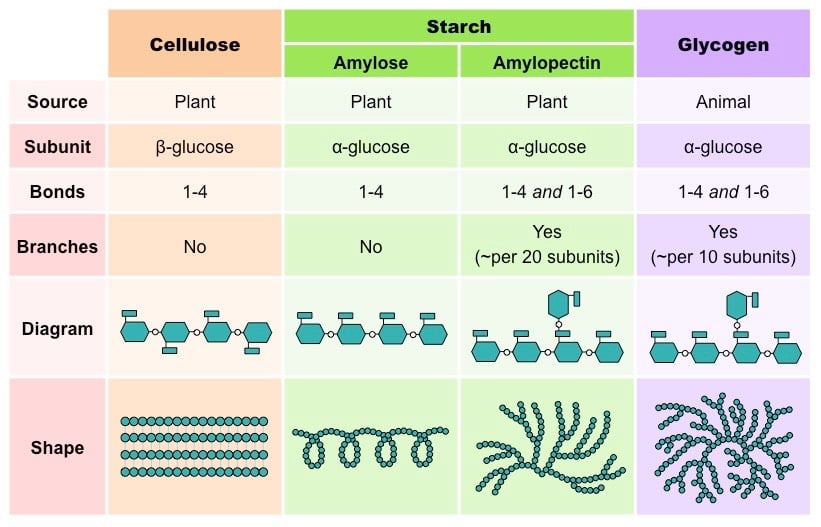
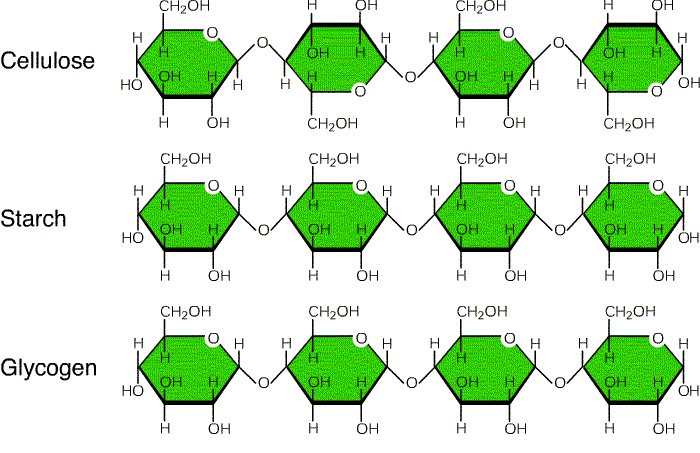
- Starch is made up of long chains of α-glucose (Amylose and Amylopectin). Glycogen is made of α-glucose linked together by glycosidic bonds. Cellulose is also made of many β-glucose molecules linked by glycosidic bonds between carbon 1 and carbon 4.
- Starch is the main energy storage materials in plants. Glycogen is the main energy storage materials in animals. Cellulose is the major component of cell walls in plants.
- The test for starch is called an Iodine test.
References
- Ann Fullick, Jo Locke and Paul Bircher. 2015. A Level Biology for OCR- A. Oxford University Press. (Buy this book)
- Ann Fullick. 2015. Edexcel AS/A level Biology 1. 2nd Edition. Pearson Education Limited.
- CGP. 2015. A-Level Biology Exam Board: AQA. Complete Revision and Practice. Original material by Richard Parsons.
- Glenn Toole and Susan Toole. 2015. AQA Biology for A-Level. 2nd Edition. Oxford University Press.
- Mary Jones, Richard Fosbery, Jennifer Gregory and Dennis Taylor. 2014. Cambridge International AS and A Level Biology Coursebook. 4th Edition. Cambridge University Press.
- Mary Jones. 2010. Cambridge International A/AS-Level Biology Revision Guide. Hodder Education.
- Sue Hocking, Frank Sochacki and Mark Winterbottom. 2015. OCR AS/A level Biology A. 2nd Edition. Pearson Education Limited.

This is cool.
This was super helpful. Very clear and concise and the background knowledge I need that my biochem professor explained but I needed additional help. Only note, I think the image you have of Maltose is incorrect. Maltose is formed by two alpha glucoses but the C1 on the second glucose molecule in the figure is in the beta form.
Thank you so much, we will correct the structure of Maltose and update soon.
Thanks genius
Love u dai
jai Gorkha
I appreciatiate your work it was very good
very use full for my exam
Great job
Well explained . I appreciate you so much ????
What a great website! Information was clear, concise, and the diagrams were useful and easy to understand.
Thank you
Why is the structure for starch and glycogen the same?
I appreciate you in your explanation