Phagocytosis is the process by which cells are used for the ingestion and degradation of particles larger than 0.5µm in diameter, such as microorganisms, foreign bodies, and apoptotic cells. This process occurs in both unicellular (such as Amoeba, Dictyostelium discoideum, and other protozoa) and multicellular organisms (Depierre et al., 2025).
Examples of phagocytic cells include neutrophils, macrophages, monocytes, dendritic cells, osteoclasts, and retinal epithelial cells. The term “Phagocytosis” means cell eating, namely the ability to engulf cells and digest pathogens, helping the body to get rid of the unwanted cells and pathogens. Phagocytes help to remove microorganisms, present antigen to lymphocytes, activate the adaptive immune system, and maintain tissue homeostasis (Uribe-Querol & Rosales, 2020; Cleveland Medical Clinic, 2024).
Phagocytosis is involved in the removal of:
- Infected cells
- Damaged, dead, or aging cells
- Tumor cells
- Cell debris
- Inactive or less active synapses
- Viruses, bacteria, fungi, or other pathogens
History: Discovery of Phagocytosis and the Role of Élie Metchnikoff
Phagocytosis was discovered by Élie Metchnikoff in 1882. While studying the larvae of transparent fish, he observed freely moving cells and proposed that these cells could engulf and destroy microorganisms and other foreign bodies. Through these observations, he realized the significance of phagocytic cells in organismal homeostasis through mesodermal amoebocytes, as well as in host defense (Tan & Dee, 2009).
Although Metchnikoff is credited with establishing the concept of phagocytosis, he was not the first person to observe phagocytic cells. This had been noted earlier by Rudolf Virchow, the father of cellular pathology, and by Claus, who later coined the term “Phagocytosis” from the Greek words phagein (meaning “eating”) and kytos (meaning “cell”) (Gordon, 2016).
What are Phagocytes?
Phagocytosis is performed by professional phagocytes, such as neutrophils, macrophages, monocytes, dendritic cells, and osteoclasts, as well as by non-professional phagocytes, including retinal epithelial cells. Professional phagocytes function efficiently because they are immune system cells dedicated to pathogen recognition and clearance, whereas non-professional phagocytic cells are non-immune cells dedicated to engulfing cellular debris and maintaining tissue homeostasis (Uribe-Querol & Rosales, 2020).
Neutrophils can detect bacteria via Fc-gamma or complement receptors on their surface, triggering cytoskeletal changes. Inside the phagosome, cytoplasmic enzymes like granzymes, collagenase, and myeloperoxidase, combined with an oxidative burst that generates reactive oxygen species, destroy the trapped particle. This process is known as neutrophilic phagocytosis, a key innate immune defense mechanism (Deshpande & Wadhwa, 2023).
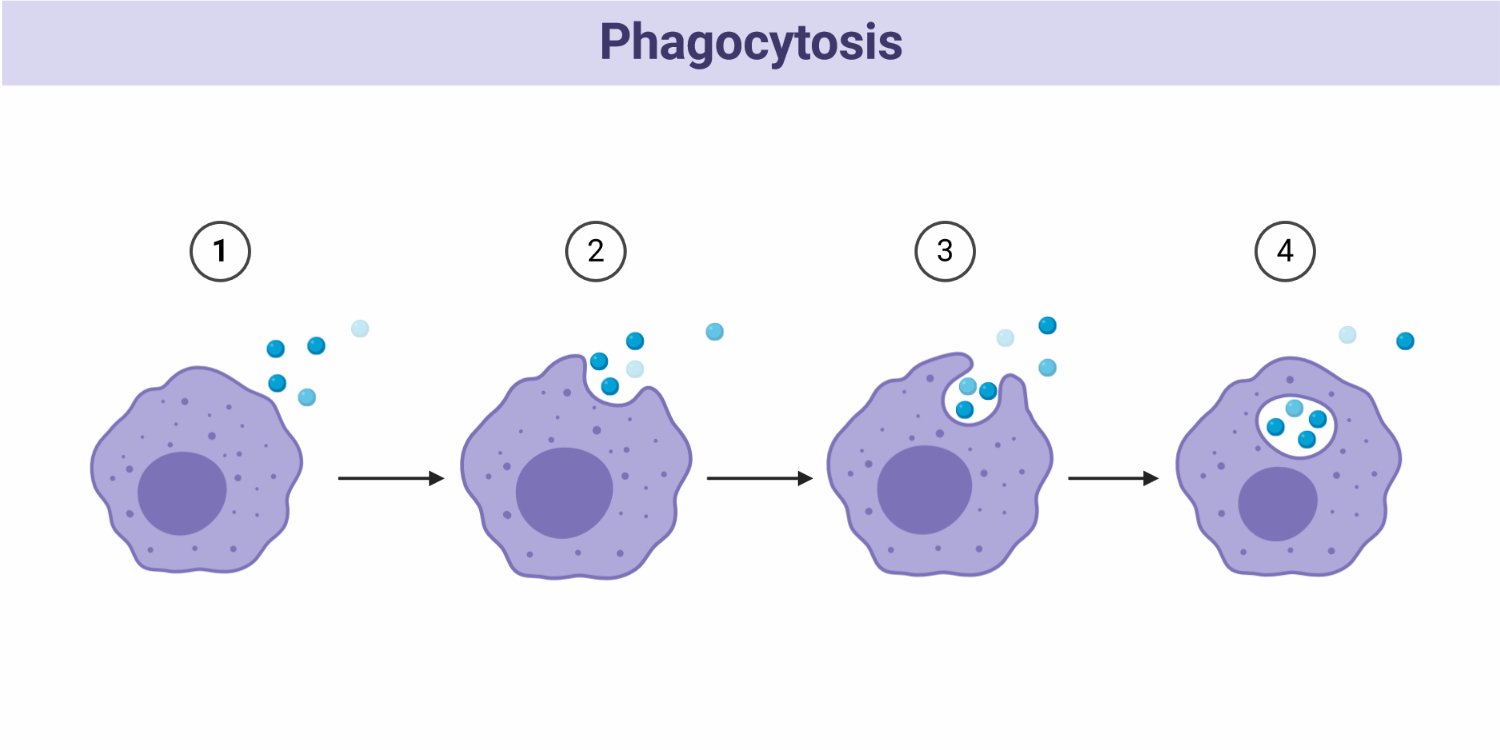
Steps of Phagocytosis
The steps of phagocytosis involve:
- Recognition and Binding: Cell-surface receptors in the cell membrane recognize and bind to antibodies (e.g., IgG) on the target (such as bacteria).
- Engulfment: Engulfment initiates when the membrane protrusions, also known as pseudopodia, form in a zipper-like mechanism to surround the target as more receptors bind to ligands.
- Phagosome formation: The membrane engulfs the target, creating a phagosome with an arc length that increases from 0 to πR for a spherical particle. The phagosome then fuses with lysosomes. It undergoes a series of fission and fusion events with endocytic organelles to acquire necessary proteins, a process known as maturation of phagosomes.
- Digestion: Acidification of the phagosome occurs, which leads to the digestion of the target.
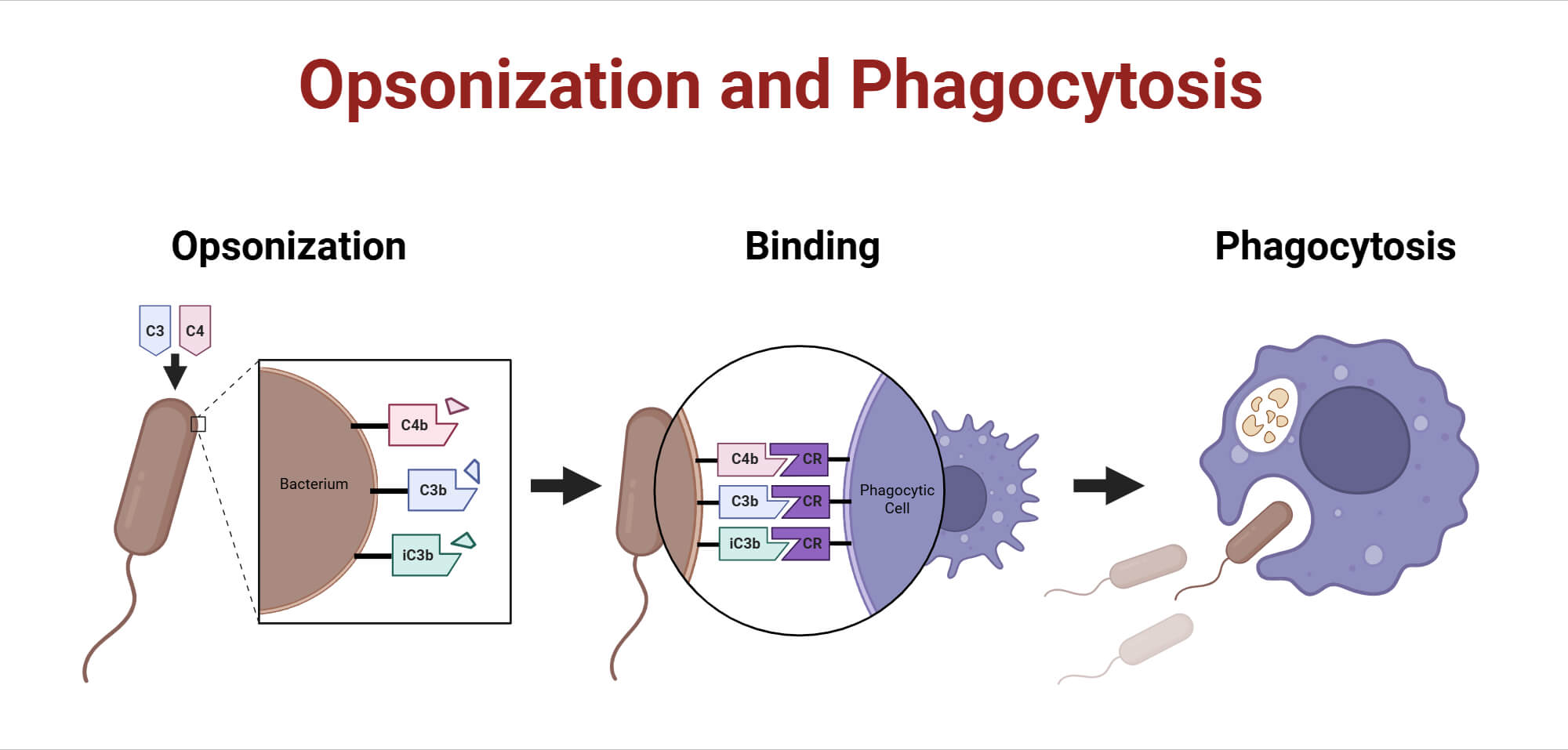
How Cells Recognize Targets: Receptors, Opsonins, and “Eat-Me” Signals
Cells recognize their target with the help of the opsonization process. This process facilitates the identification and elimination of pathogens and dead cells. Opsonins are proteins that coat pathogens, helping phagocytes recognize, internalize, and destroy or digest cellular debris ( Cleveland Clinic, 2024).
Phagocytic signals:
Phagocytes rely on different signals to determine whether to engulf a target. These include:
- Find-me signals: These help phagocytes locate cells or pathogens for destruction.
- Eat-me or “don’t eat me” signals: These signals indicate to phagocytes whether to engulf a target or not.
- Opsonins: These proteins attach to the surface of cells or pathogens and signal phagocytes to engulf and destroy them. They also act as a bridge, making binding easier.
- Don’t eat-me signals (e.g., CD47): These signals prevent phagocytes from attaching to and destroying healthy cells.
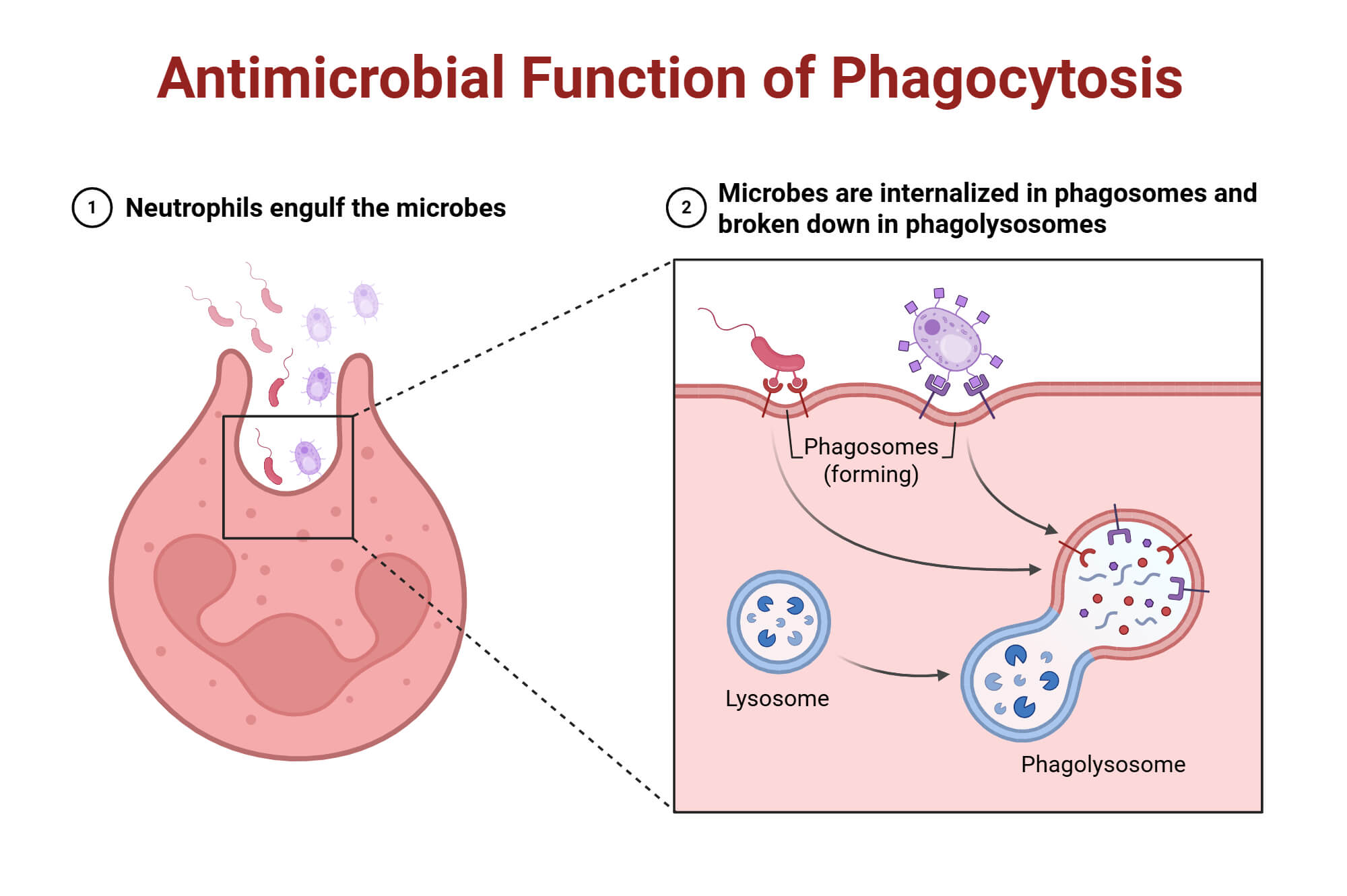
Phagosome Maturation: Fusion with Lysosomes and Pathogen Destruction
The phagosome maturation begins with transforming the newly formed phagosome into a degradative phagolysosome through sequential fusion with endocytic compartments (Roy & Kagan, 2013). It can be divided into three stages:
- Early phagosome stage: This stage acquires Rab5 and early endosomal markers. Initial acidification begins with V-ATPase recruitment.
- Intermediate phagosome stage: This stage transitions from Rab5 to Rab7, and fuses with late endosomes, thereby increasing acidity and hydrolase content.
- Late phagosome stage (phagolysosome): This stage involves full lysosomal fusion, delivering acid hydrolases, proteases, and MHC II. Its contents degrade in a highly acidic, oxidative environment.
Phagolysosomes are acidic (pH 5-5.5) due to abundant V-ATPase molecules on their membrane. They also contain degenerative enzymes, such as cathepsins, proteases, lysozymes, and lipases, along with other microbicidal components such as lactoferrin, which sequesters the iron required by bacteria and acts as a scavenger molecule (Uribe-Querol & Rosales, 2017).
Oxygen-Dependent vs. Oxygen-Independent Killing Mechanisms
Phagocytic cells trigger a rapid respiratory burst upon ingestion of microorganisms, oxidizing NADPH to produce singlet oxygen (O2–) and hydrogen peroxide (H2O2) and energy (ATP) through the cytochrome complex in the oxidative phosphorylation pathway (Clinical Gate, 2025).
Oxygen-Dependent Mechanisms
Electrons flow from cytochrome b558, the primary superoxide (O2–), releasing phagosomal free radicals. “Superoxide dismutase” converts O2– to O2 and H2O2 (2O2– + 2H+ → O2 + H2O2). Additional reactions between O2– and H2O2 produce hydroxyl radicals (OH–), while O2– and nitric acid (NO–) yield peroxynitrite (ONOO–) or its acid (ONOOH), damaging proteins, lipids, and DNA. Myeloperoxidase (MPO) from neutrophil azurophilic granules or monocyte lysosomes uses H2O2 and Cl– to generate hypochlorous acid (Cl– + H2O2 + MPO → H2O + OCl–), which kills microorganisms via acidity and cell wall dissolution.
ROS Effects on Microorganisms
Singlet oxygen disrupts bacterial walls, while H2O2 and hydroxyl radicals can harm membranes and DNA. Enzymes like superoxide dismutase, catalase, and glutathione peroxidase regulate these reactive oxygen species (Clinical Gate, 2025).
Oxygen-Independent Mechanisms
Primary granules release proteinases and lysozyme, which cleave 𝛃-1,4-glucosidic bonds between N-acetylmuramic acid and N-acetylglucosamine to breach cell walls. Secondary granules provide lactoferrin, which chelates ions to block heme cytochromes, and defensins, which form pores leading to osmotic lysis. Tertiary granules deliver gelatinase, acetyltransferase, and lysozyme for further antimicrobial action (Clinical gate, 2025).
Phagocytosis in Single-Celled Organisms: Feeding in Amoebae and Protists
Protists can exhibit many forms of nutrition and be aerobic or anaerobic. They may store energy through photosynthesis, primarily as photoautotrophs that possess chloroplasts. Other kinds of protists may be heterotrophic, obtaining nutrition by consuming organic materials to obtain nutrition. Amoeba and other heterotrophic protists ingest particles via a process called phagocytosis, in which the cell membrane engulfs food particles, forming an intracellular membranous sac, or vesicle, called a food vacuole. The food particle is broken down into small molecules, which diffuse into the cytoplasm via cellular metabolism. Undigested materials are expelled from the cell through exocytosis (Libretexts, 2022).
When Phagocytosis Fails: Diseases, Immune Evasion and Chronic Infections
Pathogens use two main strategies to subvert host immune responses and ensure prolonged survival (Andrews & Sullivan, 2003). They are,
- Evasion of immune recognition:
Pathogens modify or hide on the surface components, they secrete immunomodulators, undergo antigenic variation, or reside within protected host cells to avoid detection. Examples include,
- Lipid A or peptidoglycan modification (Salmonella, Leptospira)
- Secretion systems (Type III, IV, ESX) to modulate host defence (Mycobacteria, Salmonella, Brucella)
- Antigenic variation through gene rearrangements (Influenza Simplex virus, Epstein-Barr Virus, Leishmania)
- Modulation and Suppression of Host Immune Responses:
Pathogens inhibit host immune mechanisms, such as phagocytosis, lysosome fusion, innate immune sensors, and cytokine responses. They also resist host effector mechanisms or induce inappropriate immune reactions like immunosuppression or regulatory T cells. Some of their examples include,
- Inhibition of lysosomal fusion and acidification (Brucella, Salmonella)
- Escape into host cytosol (Listeria, Rickettsia, Francisella)
- Blockage of antigen processing and humoral immunity (Cytomegalovirus, Herpesvirus)
- Induction of regulatory T cells and immunosuppressive environments (Hepatitis C virus, HIV, Mycobacterium tuberculosis, Leishmania, Plasmodium)
These strategies enable intracellular persistent pathogens, such as bacteria, viruses, and protozoa, to evade host defense, establish chronic infection, and survive within the host for extended periods (Thakur et al., 2019).
Phagocytosis Beyond Immunity: Wound Healing, Clearing Debris, and Cell Turnover
Phagocytosis helps not only with immune responses but also in wound healing, clearing of cell debris, and removal of necrotic debris at injury sites. The process prevents inflammation and supports tissue regeneration. Necrotic cells release damage-associated molecular patterns (DAMPs), such as formyl-peptides, chemokines, and lipids, that recruit phagocytes like neutrophils and macrophages (Westman et al., 2020).
Conclusion
Phagocytosis is a key process in which cells, such as neutrophils and macrophages, engulf and break down pathogens, dead cells, and debris to maintain bodily health. Since its discovery, the role is in fighting infections, healing wounds, and clearing waste. The process involves various steps, primarily attraction, engulfing, and destroying targets. When phagocytosis fails, it leads to diseases and chronic infection. However, it balances destruction with repair, making it vital for immunity and tissue health in organisms ranging from unicellular protists like amoeba to multicellular organisms such as humans.
References
- Andrews, T., & Sullivan, K. E. (2003). Infections in patients with inherited defects in phagocytic function. Clinical Microbiology Reviews, 16(4), 597–621. https://doi.org/10.1128/CMR.16.4.597-621.2003
- Cleveland Clinic medical. (2024, April 26). Opsonization. Cleveland Clinic. https://my.clevelandclinic.org/health/body/opsonization
- Clinical Gate. (2025, April 22). Phagocytosis and intracellular killing. Clinical Gate. https://clinicalgate.com/phagocytosis-and-intracellular-killing/
- Depierre, M., Pompili, C., & Niedergang, F. (2025). Phagocytosis at a glance. Journal of Cell Science, 138(12), jcs263833. https://doi.org/10.1242/jcs.263833
- Deshpande, O. A., & Wadhwa, R. (2023). Phagocytosis. StatPearls.
- Gordon, S. (2016). Phagocytosis: The legacy of metchnikoff. Cell, 166(5), 1065–1068. https://doi.org/10.1016/j.cell.2016.08.017
- Libretexts. (2022, March 8). 5.4: Protists. Libretexts. https://bio.libretexts.org/Courses/CT_State_Northwestern/General_Biology_I_and_II/05%3A_Unit_V-_Biological_Diversity/5.04%3A_Protists
- Roy, C. R., & Kagan, J. C. (2013, January 1). Evasion of Phagosome Lysosome Fusion and Establishment of a Replicative Organelle by the Intracellular Pathogen Legionella pneumophila. NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK6111/
- Tan, S. Y., & Dee, M. K. (2009). Elie Metchnikoff (1845-1916): Discoverer of phagocytosis. Singapore Medical Journal, 50(5), 456–457.
- Thakur, A., Mikkelsen, H., & Jungersen, G. (2019). Intracellular pathogens: Host immunity and microbial persistence strategies. Journal of Immunology Research, 2019, 1356540. https://doi.org/10.1155/2019/1356540
- Uribe-Querol, E., & Rosales, C. (2017). Control of phagocytosis by microbial pathogens. Frontiers in Immunology, 8. https://doi.org/10.3389/fimmu.2017.01368
- Uribe-Querol, E., & Rosales, C. (2020). Phagocytosis: Our current understanding of a universal biological process. Frontiers in Immunology, 11. https://doi.org/10.3389/fimmu.2020.01066
- Westman, J., Grinstein, S., & Marques, P. E. (2020). Phagocytosis of necrotic debris at sites of injury and inflammation. Frontiers in Immunology, 10, 3030. https://doi.org/10.3389/fimmu.2019.03030

the notes were well written and summarised well for reading purposes