Azoles are antifungal agents against fungi causing superficial mycoses (pityriasis versicolor, Black & White piedra) and cutaneous mycoses (Tinea capitis, onchomycosis), as well as systemic mycoses (histoplasmosis, cryptococcosis).
- It is best known to interfere with the biosynthesis of ergosterol (sterol of fungal cell membrane) by attacking the cytochrome P-450-mediated enzyme “14-α-demethylase” (CYP51). This stops the demethylation of lanosterol (a precursor of ergosterol) and the reduction in ergosterol production. Hence, leaving the cell unstable and leading to cell apoptosis.
- The first compound with a prominent azole entity was benzimidazole, characterized by Woolley in 1944, but this research and the significance of azoles were undermined.
Spectrum of activity of the Azoles
Azoles have a broad spectrum of activity against the following fungi:
- Blastomyces dermatitidis
- Histoplasma capsulatum
- Cryptococcus neoformans
- Aspergillus fumigatus
- Coccidioides immitis
- Candida spp. (C. albicans, C. tropicalis, C. glabrata, C. parapsilosis, C. krusei, C. lusitaniae)
- Paracoccidiodes brasiliensis
- Microsporum spp.
- Trichophyton spp.
- Epidermophyton flocussum
Structure of the Azoles
The structure of azoles is described as a five-membered heterocyclic ring containing one nitrogen atom along with a non-carbon atom of sulphur, oxygen, or nitrogen. Azoles are aromatic, lipophilic, weakly basic, and electron-rich.
Azoles have an affinity towards an enzyme or a receptor by binding to it via a non-covalent bond, such as a hydrogen bond, an electrostatic bond, or the Van der Waals force.
- Imidazole has 2 nitrogen atoms. Its chemical formula is C3H4N2. The nitrogen atoms are not adjacent to each other.
- Triazoles have 3 nitrogen atoms. Its chemical formula is C2H3N3. The nitrogen is adjacent to each other. The aromatic stabilization is more intense in triazoles.
Source: DOI: 10.2174/0929867329666220407094430
Source: DOI: 10.2174/0929867329666220407094430
Source: DOI: 10.2174/0929867329666220407094430
Classification of the Azoles
Azoles are classified into 2 major classes:
Imidazoles
Clotrimazole, Miconazole, Econazole, Ketoconazole, Bifonazole, Oxiconazole
- Chlormidazole (chlorobenzyl imidazole) in a topical formulation was developed in 1969 by Bayer AG. It has broad activity as it is used in the treatment of oral thrush, vaginal candidiasis, against dermatophytes, and even some Gram-positive bacteria.
- Miconazole (phenethyl imidazole) was synthesized by Janssen Pharmaceutica in 1969. It is the first parenterally administered drug for the treatment of systemic mycoses. Its spectrum of activity ranges from pathogenic yeast, filamentous fungi (Aspergillus spp.), dimorphic fungi, to Gram-positive bacteria. It is very effective in the treatment of superficial candidiasis.
- Econazole has a similar spectrum of activity to miconazole. It is also very similar in structure to miconazole, except for the lack of a chlorine atom in its benzene ring. It is primarily used as a topical formulation for the treatment of superficial mycoses and is not used to treat systemic infections due to its strong affinity for serum proteins.
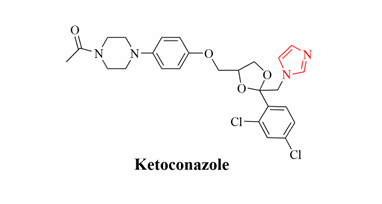
- Ketoconazole is the first commercially used azole that was orally administered to treat systemic mycoses. It was developed by Janssen Pharmaceutica in 1977.
Triazoles
First-generation triazoles: Fluconazole and itraconazole.
Second-generation triazoles: voriconazole, posaconazole, and ravuconazole.
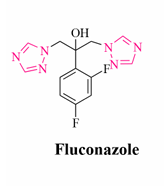
- Fluconazole was developed by Pfizer U.K. in I.V, oral, and topical formulations. This triazole has 2 azole rings and a phenyl ring with a fluoride atom at the 2nd and 4th positions.
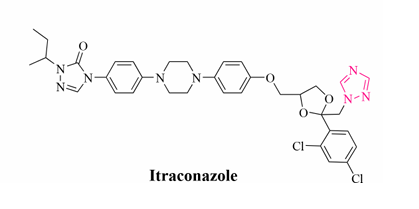
- Itraconazole was developed by Janssen Pharmaceutica. It has a broad antifungal spectrum, particularly against Aspergillus spp. and Sporothrix schenckii.
Pharmacokinetics of Azoles
Azoles are lipophilic weak bases, and this attribute correlates with their bioavailability after oral administration.
Azoles require low gastric pH for dissolution, and their lipophilic nature prompts higher chances of permeability into the cell membrane.
Ketoconazole: It is orally administered. The bioavailability varies depending upon the food intake and pH of the stomach (high pH levels decrease the absorption). Ketoconazole poorly penetrates the blood-brain barrier, hence less effective in the treatment of fungal meningitis (mainly caused by Cryptococcus neoformans). It is metabolized in the liver by CYP3A4 into inactive metabolites. It is excreted through bile.
Fluconazole: The drug is administered either intravenously or orally. It is water-soluble and binds weakly with serum proteins. It has high bioavailability because it is absorbed very well and has less toxicity compared to imidazoles. The bioavailability is not affected by food intake and gastric pH. It penetrates well into the cerebrospinal fluid. The route of elimination is the renal system. The drug remains unchanged when excreted in urine.
Itraconazole: It is mainly administered orally; intravenous administration is complexed with hydroxypropyl-β-cyclodextrin (to avert the high lipophilicity of itraconazole and make it more soluble), which can induce nephrotoxicity. The absorption depends upon food intake. The drug gets metabolized in the liver (CYP3A4) into hydroxyl-itraconazole. 45% drug is excreted through faeces, while the remaining 35% is excreted through urine.
Mode of Action of Azoles
- The primary mode of action involves one nitrogen atom (N-3 of imidazole and N-4 of triazole) of azole binding to the iron atom of the heme group in the active site of CYP51, this impede the demethylation of lanosterol (by inhibiting CYP-dependent 14-α-demethylase) and in turn prevents the biosynthesis of ergosterol. This action hinders the stability of the plasma lemma.
- Secondarily, azoles trigger changes in the oxidative and peroxidative enzyme system, leading to high concentration buildup of H2O2 intracellularly.
- The primary mode of action of triazoles is similar to that of imidazole. Triazoles like fluconazole and voriconazole additionally completely inhibit the biosynthesis of obtusifoliol.
- In C. neoformans, itraconazole and fluconazole suppress the activity of 3-ketoreductase that otherwise catalyzes the reduction of 3-ketosteroid obtusifolione to obtusifoliol.
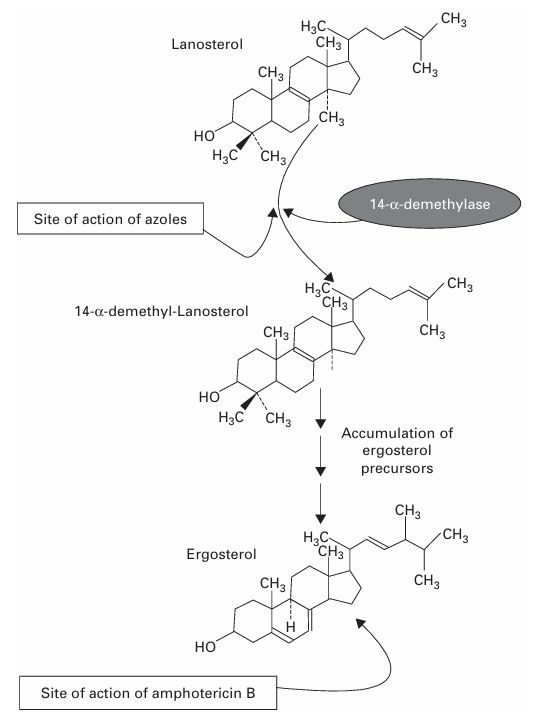
Source: Hugo and Russell’s Pharmaceutical Microbiology (7th edition)
Toxicity of the Azoles
The most common side effects imposed by azoles administered via the oral route include:
- Gastrointestinal abnormalities
- Hepatic necrosis/hepatitis
- Pulmonary edema
- Dizziness
- Rash
- Hypokalemia
Ketoconazole inhibits the corticosteroid synthesis, prompting adrenocortical inadequacy.
Resistance factors of the Azoles
Azoles, particularly fluconazole, have been prone to resistance by C. albicans. The major contributing factor for this drawback includes the following scenarios:
Increased drug efflux: CaCdr1p & CaCdr2p belong to the pleiotropic drug resistance (PDR) subfamily of ATP-binding Cassette transporter proteins are mainly responsible for azole resistance. The conformational change in these transporters pumps out the azoles. The simultaneous overexpression of CDR1 & CDR2 also leads to resistance.
MFS transporter protein contains 2 families responsible for azole resistance i.e. DHA1 & DHA2. In C. albicans, Mdr1p and Flu1p of DHA1 expel azoles from the cell.
Increased drug target: Overexpression of ERG11 increases the target enzyme and requires increased concentration of azoles. The sequestering mechanism of azole prompts upregulation of UPC2 via gain-of-function mutations. UPC2 encodes Upc2p, a zinc-cluster transcription factor that helps encourage amplification of Erg11p. Administration of high concentrations amplifies the persisting side-effects of azoles, hence decreasing their efficacy and use for treatment.
Point mutation of ERG11: This phenomenon leads to structural changes in Erg11p and results in less affinity of azole to the heme portion of 14-α-demethylase. In C. albicans, the Y132F and K143R substitutions have been clinically significant in causing fluconazole resistance.
Conclusion
Increased mycotic infection by use of broad- spectrum antibiotics that clear bacterial load but initiate/stimulate fungal colonization, increasing the number of immunocompromised patients, unintentional invasive trauma in surgical procedures that damages anatomical barrier (a part of innate immunity), and use of immunosuppressive agents (myeloablative cytotoxic therapy) all lead to vulnerability towards a mycotic infection. Taking in the consideration of pharmacokinetics of azoles and its sysnergistic interaction with other antifungal can provide better treatment or prophylaxis to combat prevalent mycoses.
References
- Bellmann, R. & Smuszkiewicz, P. (2017). Pharmacokinetics of antifungal drugs: practical implications for optimized treatment of patients. Infection, 45(6), 737-779. doi: 10.1007/s15010-017-1042-z
- Denyer, S.P., Hodges, N.A. & Gorman, S.P. (2004). Hugo and Russell’s Pharmaceutical Microbiology (7th edition, pp. 51). Blackwell Science
- El-Garhy, O.H. (2015). An overview of the azoles of interest. Int. J. Curr Pharma Res. 7(1), 1-6.
- Emami, L., Faghih, Z., Ataollahi, E., Sadeghian, S., Rezaei, Z. & Khabnadideh. (2022). Azole Derivatives: Recent Advances as Potent Antibacterial and Antifungal Agents. Current Medical Chemistry, 30(2), 220.249. doi: 10.2174/0929867329666220407094430
- Fromtling, R.A. (1988). Overview of medically important antifungal azole derivatives. Clinical Microbiology Reviews, 1(2).
- Li, Y., Hind, C., Furner-Pardoe, J., Sutton, J.M. & Rahman, K.M. (2025). Understanding the mechanism of resistance to azole antifungals in Candida spp. JAC Antimicrob Resist, 7(3). doi: 10.1093/jacamr/dlaf106
- Saag, M. & Dismukes, W.E. (1988). Azole Antifungal Agents: Emphasis on New Triazoles. Antimicrob. Agents Chemother. 32(1), 1-8.
