Histoplasma capsulatum is a dimorphic fungus found in soil that causes histoplasmosis (a type of systemic infection).
Three varieties of H. capsulatum exist: H. capsulatum var. capsulatum (known as American histoplasmosis), H. capsulatum var. duboisii (known as African histoplasmosis), and H. capsulatum var. farciminosum (known as the horse pathogen).
Morphology of Histoplasma capsulatum
- H. capsulatum is a dimorphic fungus that exists in mold form (saprophyte) in the environment.
- The hyphae are septate and bear spores.
- The spores have 2 types of conidia: macroconidia and microconidia.
- A temperature change (37°C) in the human body triggers morphogenesis into yeast form.
Habitat of Histoplasma capsulatum
- H. capsulatum dwells particularly in the soil contaminated with bat and avian excrement, which is enriched with nitrogen.
- The spores are aerosolized and dispersed in the air that enters the lung via the respiratory system.
- After invading the lung, they remain intracellularly on the alveolar macrophage.
Epidemiology of Histoplasma capsulatum
- H. capsulatum var. capsulatum is distributed all over the world. It is the most endemic fungal infection in the US, where higher incidence is seen in regions like Ohio and the Mississippi River Valley.
- H. capsulatum var. duboisii is localized in Africa.
- The soil contaminated with bat and avian excrement (nitrogen-enriched soil) has a higher occurrence. The air aerosolized by its microconidia significantly infects poultry farmers, spelunkers (cave explorers), and bat guano miners.
Pathogenesis of Histoplasma capsulatum
Inhalation
- The microconidia, after inhalation, enter the respiratory tract. When manifestation of pulmonary infection is seen, the microconidia have successfully evaded the innate defenses, like antibodies, nasal and pharyngeal mucus, and mucociliary clearance, and make their way into alveoli.
- This is likely due to the minute nature of microconidia.
Morphogenesis
- The yeast is the virulence form of H. capsulatum. The dimorphism process (from mould to yeast) is prompted by a rise in temperature (37°C in the human host).
- When the microconidia enter the human body, the mycelium form ceases, which is followed by activation of shunt pathways mediated by sulfhydryl and cysteine that bring about morphogenesis into the yeast form by creating a reduced environment in the cell critical for maintaining the configuration of protein.
- DRK1 (Dimorphism Regulating Kinase) senses the temperature fluctuation and induces the activation of RYP (Required for Yeast Phase growth) genes, which are crucial for the morphogenesis. It acts upstream of RYP.
- RYP1 (part of WOPR family) is the master regulator that activates RYP2, RYP3 & RYP4. RYP2 & RYP3 (part of the velvet family) form a heterodimeric complex which stabilizes RYP1 activity and represses the mould genes.
- RYP4 transcription factor maintains the yeast form by regulating yeast-phase-specific genes and virulence-associated genes. It acts downstream of RYP1, RYP2 & RYP3.
Surfactant proteins
- Lung surfactant is a fluid that is composed of proteins (four types: SP-A, SP-B, SP-C and SP-D) and phospholipids. It lines the alveoli to reduce surface tension.
- SP-A and SP-D belong to collectin (a subfamily of C-type lectin). SP-A & SP-D are the first line of defense, which bind to the surface of pathogens through the Carbohydrate Recognition Domain (CRD) and opsonize the pathogen via C3b cleaved from C3 for phagocytosis.
- The calcium-dependent binding of SP-A & SP-D firmly binds to the surface of the pathogen, causing cellular disintegration.
- The cellular remodeling/masking of the cell wall, secretion of protease enzymes, and intracellular sustenance inside the alveolar macrophage lower the fungicidal property of surfactant proteins to a greater extent.
Dwelling inside the macrophage
Complement Receptor-Mediated Phagocytosis: The receptors on the surface of alveolar macrophage are: LFA-1 (CD11a/CD18), CR3 (CD11b/CD18) & CR4 (CD11c/CD18). CD11a/b/c are β subunits unique to each receptor, while α is the common subunit. The binding of yeast cells is prevented when the α subunit is blocked.
The complement receptor-mediated phagocytosis of yeast cells of H. capsulatum occurs via 2 ways:
- The direct binding of HSP60 (Heat shock Protein) present on the yeast cell surface with the CR3 receptor of macrophage. This minimizes the macrophage attack/inflammatory cascade.
- Opsonization by C3b tags, which are recognized by the CR3 receptors, hence the internalization.
Cell wall modification: Dectin-1 is another pathogen-recognizing receptor (PRR) of macrophages capable of recognizing fungal Pathogen-Associated Molecular Pattern (PAMPS). Dectin-1 recognizes the β-glucan (a major component of the yeast cell wall) and triggers a cascade of inflammatory response.
- H. capsulatum evades this process via production of α-glucans, which mask the β-glucans and thus increase the virulence. α-glucans are produced by α-(1,3)-glucan synthase encoded by AGS1 gene.
- Other genes that are involved in the synthesis of α-glucans indirectly involve AMY1, which produces α-(1,4) linked oligosaccharides, which are then used by AGS1 to synthesize α-glucans.
- Similarly, the UGP1 gene that generates UTP-glucose-1-phosphate uridylltransferase to synthesize UDP-glucose monomers is subsequently used by AGS1.
Inerting Reactive oxidation: H. capsulatum dwells inside phagocytic vacuoles. The phagosome membrane-associated complex: NADPH oxidase activates oxidative burst (a surge of reactive O2). NADPH oxidase reduces O2 into superoxide, which attacks the pathogen extracellularly. It has a fungicidal nature.
- Superoxide can further be broken down into H2O2, which can attack both intracellularly and extracellularly.
- However, when the phagocytosis is CR3-mediated, the NADPH oxidase does not get strongly triggered.
- H. capsulatum contains superoxide dismutase (Sod3) that decreases the free radicals. To subsidize the effect of H2O2, catalases, CatB, and CatP are produced. They reduce hydrogen peroxide into water and oxygen.
Inerting the effect of reactive nitrogen: Superoxide combines to form nitric oxide, which produces reactive nitrogen. It causes DNA damage and stops cell replication. Reactive nitrogen has a fungistatic effect.
- The NOR1 gene encodes nitric oxide reductase, which reduces reactive nitrogen into less toxic substances.
Neutralization of phagosome acid and lysosomal fusion: The Vacuolar ATPases are pumps on the membrane surface of the phagosome that bring in protons, which decrease the pH. Further fusion with the lysosome intensifies the acidity.
- The mechanism of pH neutrality brought by H. capsulatum is unknown. However, studies done by Issac et al. have speculated the correlation of HMG CoA lyase (encoded by HCL1) for growth within the macrophage (Issac et al., 2013).
- Inside the phagosome, leucine is more abundant and lacks sufficient glucose. The metabolism of leucine by H. capsulatum may give a basic byproduct, thus reducing the acidity.
Apoptosis of the host cell and dissemination of Histoplasma capsulatum
To disseminate, H. capsulatum must inflict cell death via apoptosis.
- Pitangui et al. emphasized the intracellular movement and aggregation of yeast cells around the nucleus, which damages the DNA, and eventual cell death occurs (Pitangui et al, 2015).
- Deepe et al. give details on the use of extrinsic pathways for apoptosis, triggered by TNF-α (that binds to its receptor TNFR1 on the surface of macrophage), which activates caspase 1 & 3. Contrary to this, there is an increase in IL-10 (an anti-inflammatory cytokine that inhibits apoptosis) in neighbouring cells. This makes the pathogen easy to dwell in phagocytes of neighbouring cells, sustaining and disseminating itself (Deepe and Buesing, 2012).
- The presence of CBP1 was crucial for macrophage lysis (Sebghati et al., 2000).
- The mutants of H. capsulatum that lacked CBP1 were not able to induce expression of stress-responsive host genes that would have activated caspases 3 and 7. (Isaac, et al., 2015)
Clinical features of Histoplasma capsulatum
- The disease manifests into 3 types: Acute histoplasmosis, Chronic histoplasmosis, and Disseminated histoplasmosis.
- Acute histoplasmosis: In an immunocompetent individual, acute histoplasmosis is often transient and asymptomatic. Symptomatic patient develops fevers, chills, cough, “flu-like” symptoms. It can also manifest as pneumonia or mediastinal lymphadenopathy.
Depending on inoculum size and host immune response, the lung tissue is left with calcified lesions (histoplasmoma).
- Chronic histoplasmosis: It is prevalent in older demographics predisposed to injured lung tissue. The disease is slowly progressive from pulmonary infiltrates to fibrosis and cavity lesions. Apical bullae and bronchopleural fistulas are a complicated form of chronic histoplasmosis. The cavity lesions can be confused with tuberculosis.
- Disseminated histoplasmosis: Disease can be disseminated throughout the reticuloendothelial system and other primary organs such as the liver, lymph nodes, and bone. Patients with low CD4 count (HIV/AIDs) or those undergoing immunosuppressive therapy become susceptible to dissemination.
- H. duboisii infection is mainly concentrated in the skin, which can ulcerate. Bone abscess is also observed, and in fatal cases, dissemination into the liver and spleen is seen.
Cultural characteristics of Histoplasma capsulatum
- Sabouraud agar: The mould takes 2-3 weeks to grow at 25-30°C. Colonies appear white to buff-brown in color and fluffy. Microconidia (diameter: 2-5 μm) are elliptical and smooth-walled, while macroconidia (diameter: 8-15μm) are tuberculate and round with age. Mycelium is septate.
The mold culture is considered a biohazard; hence, screw-capped bottles should be used.
- Blood agar enriched with cysteine: The medium is used to grow the yeast form, which appears as wrinkled, heaped colonies (2-4 μm) ranging from creamy to tan color.
Laboratory diagnosis of Histoplasma capsulatum
- Various diagnostic methods are available, such as pathology, culture, antigen detection, and serology.
- The culture is considered a gold standard.
- Antigen detection involves the detection of the Histoplasma polysaccharide by antibodies. Urine is the most commonly used fluid for the test.
- Serology of chronic histoplasmosis involves techniques like complement fixation, enzyme immunoassay, and immunodiffusion.
- In pathology, sputum sample (H. capsulatum) and pus from the lesion (H. duboisii) should be Giesma-stained. In disseminated histoplasmosis, Grocott-Gomori methenamine-silver of lung or liver biopsies is done. H. capsulatum appears as small, round, oval cells with macrophages; H. duboisii appears as large oval to round cells.
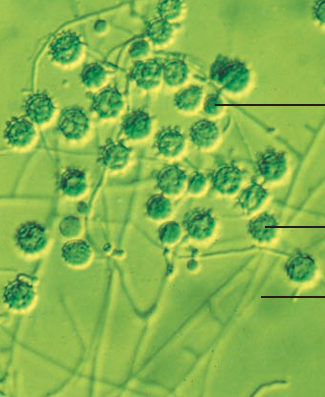
Source: Prescott, Harley & Klein’s Microbiology (7th edition)

Source: 10.7759/cureus. 751
Treatment of Histoplasma capsulatum
- Itraconazole is administered to treat acute histoplasmosis when symptoms are observed.
- For severe acute pulmonary disease, liposomal amphotericin B is administered as the first line of drug, followed by itraconazole.
- In disseminated histoplasmosis, intravenous amphotericin B is recommended.
Conclusion
H. capsulatum is a type of dimorphic fungi, capable of causing infection in both immunocompromised and immunocompetent individuals. The strategy through which it maneuvers inside the host by evading many host immune responses is a dire concern. Its diaspora is ubiquitous; therefore, the pathogenesis and the adaptation of new pathogenic mechanisms should be intensely studied.
Reference
- Cheesbrough, M. (2006). District Laboratory Practice in Tropical Countries (2nd edition). Cambridge University Press.
- Deepe, G.S. & Buesing, W.R. (2012). Deciphering the pathways of death of Histoplasma capsulatum-infected macrophages: implications for the immunopathogenesis of early infection. J Immunol, 188 (1), 334-344. doi: 10.4049/jimmunol.1102175
- Greenwood, D., Barer, M., Slack, R. & Irving, W. (2012). Medical microbiology (18th edition). Churchill Livingstone Elsevier.
- Issac, D.T., Coady, A., Van Prooyen, N. & Sil, A. (2013). The 3-hydroxy0methylglutaryl coenzyme A lyase HCL1 is required for macrophage colonization by human fungal pathogen Histoplasma capsulatum. Infect Immun, 81(2), 411-420. doi: 10.1128/iai.00833-12
- Issac, D.T., Berkers, C.A., English, B.C., Murray, D.H., Lee, T.N., Coady, A. & Sil, A. (2015). Macrophage cell death and transcriptional response are actively triggered by fungal virulence factr Cbp1 during H. capsulatum infection. Mol Microbiol, 98(5), 910-929. doi: 10.1111/mmi.13168
- Mittal, J., Ponce, M.G., Gendlina, I. & Nosanchuk, J.D. (2020). Histoplasma capsulatum: Mechanisms for Pathogenesis. Curr Top Microbiol Immunol, 422, (157-191). doi: 10.1007/82_2018_114.
- Piatangui, NeS., Sardi, JeC., Volton, A.R., Dos Santos, C.T., da Silva, JeF., da Silva, R.A., Souza, F.O., Soares, C.P., Rodriguez-Arellanes, G., Taylor, M.L., Mendes-Giannini, M.J. & Fusco-Almeida, A.M. (2015). An intracellular arrangement of Histoplasma capsulatum yeast-aggregates nuclear damage to the cultured murine alveolar macrophages. Front Microbiol, 6, 1526. doi: 10.3389/fmicb.2015.01526
- Tille, P.M. (2014). Bailey & Scott’s Diagnostic Microbiology (13th edition, pp. 705-757). Elsevier Mosby.
- Willey, J.M., Sherwood, L.M. Woolverton, C.J. (2008). Prescott, Harley and Klein’s microbiology (7th edition). McGraw-Hill.
