Moraxella catarrhalis, previously known as Neisseria catarrhalis and Branhamella catarrhalis, is commonly found as a commensal organism in the human respiratory tract. However, it can act as an opportunistic pathogen, particularly in vulnerable populations.
M. catarrhalis is a frequent cause of otitis media, sinusitis, ocular infections, and lower respiratory tract infections—including bronchitis and pneumonia—especially in children and elderly individuals. In rare cases, it may also result in severe systemic diseases such as endocarditis and meningitis.
Taxonomy and Classification of Moraxella catarrhalis
Domain: Bacteria
Kingdom: Pseudomonadati
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Pseudomonadales
Family: Moraxellaceae
Genus: Moraxella
Species: M. catarrhalis
Morphology and Microscopy of Moraxella catarrhalis
- Gram-negative coccobacillary rods that occur in pairs or sometimes in short chains.
- Approximately 0.6 – 1.0µm in diameter.
- Non-motile
- Non-spore-forming
- Capsulated
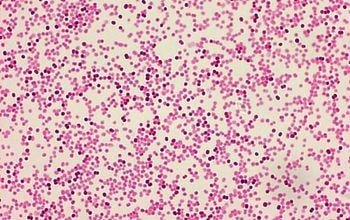
Morphologically, M. catarrhalis appears as Gram-negative diplococci, closely resembling Neisseria species on Gram stain. This similarity can make initial microscopic differentiation challenging, emphasizing the importance of further biochemical and molecular identification methods.
Source: https://microbe-canvas.com/Bacteria/gram-negative-cocci/oxidase-positive-3/moraxella-catarrhalis.html
Cultural and Growth Characteristics of Moraxella catarrhalis
- It is aerobic bacteria that grow well in suitable culture media under aerobic conditions.
- Optimum temperature: 33˚C to 35˚C
- Optimum pH: 7
- On Nutrient Agar: The colonies are small, smooth, grayish-white, convex, and firm, with a characteristic “hockey puck” appearance.
- On Blood Agar: Colonies are non-hemolytic, 1-3 mm in size, round, opaque, convex, and grayish white in color. The colonies remain intact on the surface of the agar and can be pushed across the medium without breaking apart, exhibiting the characteristic “hockey puck” phenomenon.
- On Chocolate Agar: The colonies are medium-sized, smooth, pinkish-brown, moist, and convex, with a firm consistency that demonstrates the characteristic “hockey puck” phenomenon.

Source: https://microbe-canvas.com/Bacteria/gram-negative-cocci/oxidase-positive-3/moraxella-catarrhalis.html
Biochemical and Identification Tests of Moraxella catarrhalis
| Tests | Results |
| Gram Staining | Negative |
| Catalase | Positive |
| Oxidase | Positive |
| Indole | Negative |
| Citrate | Negative |
| Glucose | Negative |
| Sucrose | Negative |
| Lactose | Negative |
| Mannitol | Negative |
| Nitrate Reduction | Positive |
| DNase Production | Positive |
Pathogenesis and Virulence Factors of Moraxella catarrhalis
Adherence
- Adherence of bacteria to the host, particularly to respiratory mucosal cells, is a critical first step in colonization. This process enables M. catarrhalis to attach firmly to the epithelial cells of the respiratory tract, facilitating persistent colonization and subsequent infection.
- M. catarrhalis produces various adhesins such as surface protein A family (UspA), the human erythrocyte agglutinin/Moraxella immunoglobulin D-binding protein (hag/MID), the outer membrane protein CD (OMP CD), M. catarrhalis adherence protein (McaP), and lipoligosaccharide (LOS), which assist in adherence of bacteria to host cells.
Invasion
- Invasion is observed mainly in vitro.
- It is regulated through the expression of UspA1, LOS, and other outer membrane proteins.
- Invasion permits M. catarrhalis to resist destruction by the host immune system and extracellular antibiotics.
- The mechanism of bacterial invasion is partially understood, and further studies are required.
Biofilm Formation
- Bacteria are capable of forming biofilms both in vitro and in vivo.
- The virulence factors UspA and Hag/MID play key roles in regulating biofilm formation.
Immune Evasion Strategies-Complement Resistance
- M. catarrhalis expresses different mechanisms to hinder complement-mediated killing.
- It is capable of stimulating all pathways of the human complement system, as well as both surface-exposed structures and major outer membrane proteins involved in complement defense.
- M. catarrhalis can attach to the regulator protein C4b binding protein via UspA1 and UspA2.
- Similarly, UspA2 is capable of binding to C3 and vitronectin and, as a result, obstructs the activation of the alternative complement pathway.
Virulence Factors
Outer Membrane Proteins (OMPs)
- It helps in the adhesion of bacteria to host epithelial cells.
- It aids in the colonization of the respiratory mucosa.
Human Erythrocyte Agglutinin/Moraxella Immunoglobulin D-binding Protein (hag/MID)
- It is one of the important multifunctional outer membrane proteins.
- It helps in the attachment of bacteria to host respiratory tract cell lines.
- Phage variation is responsible for the expression of Hag/MID.
- It modulates immune response and helps in immune evasion.
Lipooligosaccharide (LOS)
- It acts as an endotoxin which triggers inflammation and the release of cytokines like IL-1 and TNF-α.
β-lactamase Production
- It confers resistance to penicillin and related antibiotics.
Complement Resistance
- It evades killing by the complement system.
Epidemiology of Moraxella catarrhalis
In 1896, this bacterium was originally named Mikrokokkus catarrhalis in German and later Micrococcus catarrhalis in English. Similarly, in 1963, Micrococcus catarrhalis was divided into two distinct species: Neisseria catarrhalis and Neisseria cinerea based on differences in nitrate and nitrite reduction as well as hydrolysis of tributyrin. Then, in 1970, it was named Branhamella catarrhalis as it was phylogenetically different from Neisseria.
Finally, in 1984, on the basis of the genetic relatedness of its 16S rRNA sequence to Moraxella, it was named Moraxella catarrhalis. Over the last 20 to 30 years, the bacterium has developed into a real pathogen and is now a significant cause of upper respiratory tract infection in healthy children and elderly people. Approximately 15-20% of acute otitis media in infants was caused by M. catarrhalis.
The rate of colonization of the nasopharynx is higher in infants than in adults. Similarly, each year, approximately 2-4 million of the worst cases of chronic obstructive pulmonary disease (COPD) are related to M. catarrhalis infection in the United States. This bacterium is found mainly in early childhood, and by the age of 6 months, the rate of colonization ranges from 22% to 55%.
Likewise, various nosocomial outbreaks have been reported in adults and children. Winter and spring seasons, as well as multi-bed wards, are considered important risk factors for nosocomial transmission.
One study demonstrated peak colonization in the autumn and winter seasons in healthy children. While another study demonstrated a seasonal peak in winter and spring. It has been reported that children with nasopharyngeal respiratory syncytial virus showed a high risk for the advancement of acute otitis media, which suggests that the viral infection directs the way for subsequent bacterial acute otitis media.
Transmission of Moraxella catarrhalis
M. catarrhalis is transmitted through various routes:
Respiratory Droplet Transmission
- It is the main route of transmission in which bacteria spread via cough, sneeze, or while talking.
- It is most common among children and in close-contact settings.
Direct Contact Transmission
- It is transmitted through direct contact with contaminated hands and surfaces.
- It also spreads through the secretions from the mouth or nose of one person to another person.
- It is most common in daycare centers and crowded environments.
Endogenous Transmission
- In healthy individuals, bacteria remain in nasopharyngeal flora, but disease may occur when it overgrows or spreads to other sites, such as the middle ear, causing otitis media, the sinuses, causing sinusitis, and the lower respiratory tract, causing bronchitis and pneumonia.
Clinical Manifestations of Moraxella catarrhalis
Upper Respiratory Tract Infections
-Middle Ear Infection (Otitis Media)
- It is most common in children.
- Symptoms include ear pain, fever, irritability, and difficulty hearing.
-Sinus Infection (Sinusitis)
- It is common in children and adults with weakened immune systems.
- Symptoms include greenish-yellow discharge from your nose, pressure or pain in the face, and nasal discharge.
-Pharyngitis
- It is less common and causes a sore throat and mild fever.
Lower Respiratory Tract Infections
-Bronchitis
- It especially occurs in adults with lung conditions in hospitals.
- Symptoms include cough (often productive), wheezing, and discomfort in the chest.
-Pneumonia
- It is more common in elderly and COPD patients.
- Symptoms include fever, dyspnea, and productive cough.
Chronic Respiratory Disease
- Symptoms include coughing, wheezing, coughing up mucus, chest tightness, shortness of breath, and difficulty breathing.
- It can get worse over time and can cause serious complications, including death.
Pink Eye
- Bacteria cause infection of the outer layer of the eye, known as pink eye, especially in newborns and children.
Meningitis
- It can cause meningitis in newborns in very rare cases.
Bacteremia
- It is very rare and usually occurs in immunocompromised individuals.
- Symptoms include fever, chills, and systemic signs of infection.
Laboratory Diagnosis of Moraxella catarrhalis
Sample Collection
The sample depends on the site of infection:
| Site of Infection | Samples |
| Middle Ear Infection | Ear Discharge |
| Sinus Infection | Nasal Discharge |
| Lower Respiratory Tract Infection | Sputum |
| Bacteremia | Blood |
Microscopy
- On Gram staining, it appears as Gram-negative diplococci arranged in pairs similar to those of Neisseria species.
Culture
- Organism is inoculated in suitable media under aerobic conditions and incubated at 35-37˚C for 18-24 hours.
- On Nutrient Agar: Colonies are small, smooth, grayish-white, convex with a firm hockey puck appearance.
- On Blood Agar: Colonies are non-hemolytic, 1-3 mm in size, round, opaque, convex, and grayish white in color. Colony remains intact over the surface of the media, showing the hockey puck phenomenon.
- On Chocolate Agar: Colonies are medium-sized, smooth, pinkish-brown, moist, convex with a firm hockey puck consistency.
Biochemical Tests
- After culture, colonies from incubated plates are tested for biochemical tests and identified as M. catarrhalis based on the following results:
| Tests | Results |
| Gram Staining | Negative |
| Catalase | Positive |
| Oxidase | Positive |
| Indole | Negative |
| Citrate | Negative |
| Glucose | Negative |
| Sucrose | Negative |
| Lactose | Negative |
| Mannitol | Negative |
| Nitrate Reduction | Positive |
| DNase Production | Positive |
Molecular Methods (PCR)
- It is used for rapid and accurate identification in an advanced laboratory.
Treatments of Moraxella catarrhalis
First-line Antibiotics
- Amoxicillin-clavulanic acid 625mg 3 times a day for 14 days.
- Trimethoprim-sulfamethoxazole
- Macrolides like Azithromycin
- Extended-spectrum Cephalosporins like Cefixime
Alternative Antibiotics
- Clarithromycin 500mg 2 twice daily for 14 days.
- Doxycycline 100mg 2 twice daily for 14 days.
- Ciprofloxacin 500mg or 750mg twice daily for 14 days.
Supportive Treatment
- Proper and adequate hydration and proper rest.
- Use of analgesics and antipyretics such as Ibuprofen to control pain and fever.
Prevention and Control of Moraxella catarrhalis
- Regularly washing hands with soap and water and using sanitizer while outside.
- Avoid touching your nose, mouth, and eyes with unclean hands.
- Covering the mouth and nose while coughing and sneezing.
- Avoid close contact with infected patients.
- Always wear masks in crowded or high-risk areas.
- Proper sterilization of surfaces and objects touched by an infected person.
- Special care should be given to children, elderly individuals, and patients with COPD.
- Prompt and appropriate antibiotics should be given for the treatment of infections.
- Proper management of underlying conditions such as COPD and asthma.
- Early treatment of upper respiratory tract infections to reduce complications.
Antimicrobial Resistance of Moraxella catarrhalis
Before the mid-1970s, M. catarrhalis was thought to be non-β-lactamase producing; β-lactamase-positive strains are now ubiquitous. β-lactamases such as BRO-1, BRO-2, and BRO-3 are closely associated with resistant M. catarrhalis, which is evidently different from other β-lactamases and TEM. β-lactamase genes are located chromosomally on a conjugative transposon, yet the existence of plasmids and shift of β-lactamase have been recorded. These β-lactamase-producing strains are resistant to amoxicillin (MIC 16-64mg/L), penicillin, and ampicillin and show decreased susceptibility towards various Cephalosporins such as cefuroxime MIC 16-128mg/L. The resistance rate was raised notably in Europe from 70% in 1992 to 82% in 1993, though not remarkably, but there was a slight increase in the resistance rate in the USA from 85% to 92%. Similarly, no remarkable resistance was observed against macrolides, and even less resistance was reported from Spain. Likewise, remarkable resistance was not found against fluoroquinolones.
Conclusion
M. catarrhalis is a Gram-negative, diplococcus that frequently colonizes the human upper respiratory tract as a part of the normal flora, especially in children. Although usually harmless in healthy individuals, it can act as an opportunistic pathogen, causing infections like otitis media, sinusitis, bronchitis, and pneumonia, particularly in children, the elderly, and patients with underlying diseases such as COPD. Its pathogenicity is mainly due to factors like adhesion, biofilm formation, complement resistance, and production of β-lactamase, which contribute to the persistence of bacteria and antibiotic resistance. Overall, it is a clinically significant respiratory pathogen whose impact can be reduced through good hygiene, early diagnosis, control of infection, and timely treatment of respiratory infections.
References
- Public Health Agency of Canada. (2011). Pathogen safety data sheet: Infectious substances—Moraxella spp. Government of Canada. https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/pathogen-safety-data-sheets-risk-assessment/moraxella.html
- Verduin, C. M., Hol, C., Fleer, A., van Dijk, H., & van Belkum, A. (2002). Moraxella catarrhalis: From emerging to established pathogen. Clinical Microbiology Reviews, 15(1), 125–144. https://doi.org/10.1128/CMR.15.1.125-144.2002
- Bernhard, S., Spaniol, V., & Aebi, C. (2012). Molecular pathogenesis of infections caused by Moraxella catarrhalis in children. Swiss Medical Weekly, 142, w13694. https://doi.org/10.4414/smw.2012.13694
- Public Health England. (2015). UK Standards for Microbiology Investigations: ID 11i3 – Identification of Moraxella species and morphologically similar organisms. Royal College of Pathologists. https://www.rcpath.org/static/512832c6-58fb-4e62-9c2f597965c1c892/uk-smi-id-11i3-identification-of-moraxella-species-and-morphologically-similar-organisms-february-2015-pdf.pdf
- Farajzadeh Sheikh, A., Feghhi, M., Torabipour, M., Saki, M., & Veisi, H. (2020). Biochemical characteristics of Moraxella catarrhalis isolated from conjunctival swab [Table]. ResearchGate. https://www.researchgate.net/figure/Biochemical-characteristics-of-Moraxella-catarrhalis-isolated-from-conjunctival-swab_tbl1_346325263
- MechPath. (2021, November 16). Moraxella catarrhalis. https://mechpath.com/2021/11/16/moraxella-catarrhalis/
- Dix, M. (2019, January 30). Moraxella catarrhalis: Infection, causes, symptoms, and treatment. Healthline. https://www.healthline.com/health/Moraxella-catarrhalis
- Morris, D. E. (2022). The epidemiology of Moraxella catarrhalis (Doctoral thesis, University of Southampton). University of Southampton Institutional Repository. https://eprints.soton.ac.uk/475339/
- DrOracle. (2025, March 18). What are the treatment options for Moraxella catarrhalis infections? https://www.droracle.ai/articles/35662/what-are-the-treatment-options-for-moraxella-catarrhalis-infections
- Bell, A. (2020, May 6). Moraxella catarrhalis: Linked conditions, treatment, and prevention. Medical News Today. https://www.medicalnewstoday.com/articles/moraxella-catarrhalis
