Indole Test Definition
Indole test is a biochemical test conducted on bacterial species to detect their ability to produce indole from tryptophan in the presence of a group of enzymes called ‘tryptophanase’.
- It is a qualitative test that tests the conversion of tryptophan into indole.
- The test is performed as a part of the IMViC test that is used to differentiate the members of the Enterobacteriaceae family.
- It is important in the identification of different bacteria like Escherichia coli, Proteus, Morganella, etc.
- It plays a central role in the characterization of coliforms which is indicated by the various variations of the indole test and in combination with other biochemical tests.
- It is still used as a traditional method to distinguish indole-positive E. coli from indole-negative Enterobacter and Klebsiella.
- A variation of the test, called Ehrlich’s reagent (using ethyl alcohol instead of isoamyl alcohol) is used when the test has to be performed on non-fermenters and anaerobes.
Objectives of Indole Test
- To detect the formation of indole from tryptophan by the enzymatic action of tryptophanese.
- To differentiate members of family Enterobacteriaceae, especially E. coli from Enterobacter and Klebsiella.
Microorganisms Tested
- Fresh growth of a Gram-negative rod on a medium that does not contain dyes and contains tryptophan, e.g., BAP or CHOC.
- Anaerobic Gram-positive rods.
- Anaerobic Gram-negative rods.
Principle of Indole Test
The ability of an organism to split indole from the amino acid tryptophan is due to the presence of tryptophanase. Tryptophan is an amino acid that undergoes deamination and hydrolysis in the presence of the enzyme tryptophanase. Reductive deamination of tryptophan results in the production of indole via the intermediate molecule indole pyruvic acid. During the deamination process, the tryptophanase catalyzes the removal of the amino group (-NH2) from the tryptophan molecule. The enzyme requires pyridoxal phosphate as a coenzyme. The final products of the catalysis reaction are indole, pyruvic acid, ammonium (NH4+), and energy. Indole, if present, combines with the aldehyde in the reagent to produce a pink to red-violet quinoidal compound (benzaldehyde reagent) or a blue to green color (cinnamaldehyde reagent). The indole combines with Kovac’s reagent (hydrochloric acid and p-dimethylaminobenzaldehyde in amyl alcohol) solution to form a yellow or cherry red coloration. The amyl alcohol is water-insoluble and thus, forms a red-colored oily layer at the top of the broth. In the rapid spot test, indole is detected directly from a colony growing on a medium rich in tryptophan. The indole combines with the p-dimethylaminocinnamaldehyde (DMACA) present on the filter paper at an acid pH to produce a blue to the blue-green compound.
Media, Reagents, and Supplies Used
Reagents Used
- For the spot test, 5% p-dimethylaminobenzaldehyde or 1% pdimethylaminocinnamaldehyde is prepared in 10% (v/v) concentrated HCl.
- For the tube method, Kovac’s reagent is used for aerobic organisms, and Ehrlich’s reagent is used for anaerobes and weak indole producers.
Kovac’s reagent
| Ingredients | Concentration |
| p-dimethylaminobenzaldehydre | 50 gram |
| Hydrochloric acid | 250 ml |
| Amyl Alcohol | 750 ml |
- The medium used with this reagent is either broth containing tryptophan, motility- indole-ornithine agar, or sulfide-indole-motility agar (SIM).
Ehrlich’s reagent
| Ingredients | Concentration |
| p-dimethylaminobenzaldehydre | 1 gram |
| Hydrochloric acid | 20 ml |
| Ethyl alcohol | 95 ml |
- The medium used with Ehrlich’s reagent is heart infusion or anaerobic medium with tryptophan.
Supplies Used
- Sterile loop, swab, or stick for inoculation
- Filter paper (optional)
Procedure of Indole Test
A. Rapid Spot Test
- A piece of filter paper is taken and is moistened with the reagent.
- A well-isolated colony from an 18-24-hour culture is taken with an inoculating loop and rubbed onto the moist filter paper.
- The paper is then observed for the appearance of the color.
B. Tube Test
- The broth medium or the agar medium in the tube is stabbed with the colony taken from an 18-24-hour culture.
- For the liquid medium, a small portion of the inoculated broth is taken in a separate tube.
- The tubes are then incubated at 37°C for 24 hours.
- For Kovac’s reagent, three drops of Kovac’s’ reagent are added down the side of the tube, and the color change is observed at the meniscus.
- For Ehrlich’s reagent, 0.5 ml of xylene is added to the tube and inverted to mix well. Further, six drops of Ehrlich’s indole reagent are added down the side of the tube, and the color is observed below the xylene layer.
Result Interpretation of Indole Test
A. Spot Test
- The development of blue color within 20 seconds indicates the presence of indole.
- A negative result is indicated by no color or the appearance of a slightly pink color on the filter paper.
Figure: Spot Indole Test. Image Source: VetBact.
B. Tube Test
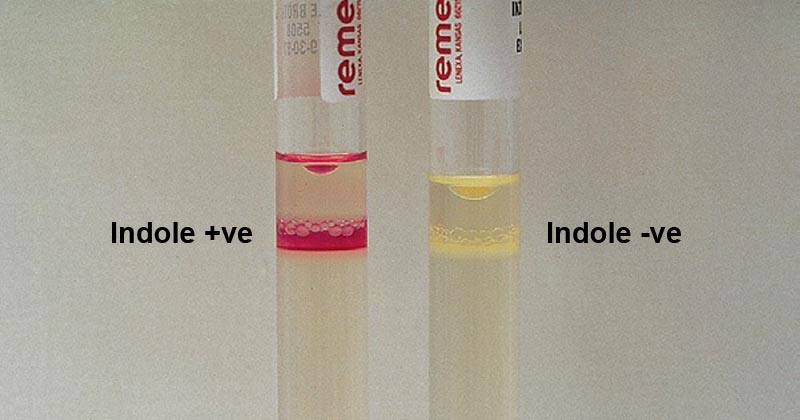
- The formation of pink-red coloration (cherry red ring) in the reagent at the point of contact between the reagent and the medium indicates a positive result.
- The absence of color or the appearance of slightly yellow color indicates a negative result.
Figure: Indole Test. Image Source: Bailey and Scott’s Diagnostic Microbiology. Elsevier.
Uses of Indole Test
- The indole test is used to test an organism’s ability to utilize tryptophan and produce indole.
- The test is used to differentiate members of the Enterobacteriaceae family as a part of the IMViC test.
- The test also differentiates Proteus mirabilis from other Proteus species.
- This test differentiates indole-positive E. coli from indole-negative Enterobacter and Klebsiella.
- The test further differentiates K. pneumoniae(indole negative) from K. oxytoca (indole positive) and Citrobacter freundii (indole negative) from Citrobacter koseri (indole positive).
Limitations of Indole Test
- Detectable indole will diffuse to colonies within 5 mm of a 2- to 3-mm colony, giving false-positive results.
- Media that contain dyes (e.g., EMB, MAC) shouldn’t be used.
- The growth medium must contain an adequate amount of tryptophan. Do not use Mueller-Hinton agar for the test, because tryptophan is destroyed during the acid hydrolysis of casein.
- Only the cinnamaldehyde reagent can be used for spot testing of anaerobic microorganisms. It is a more sensitive reagent, but it is less stable.
- Do not use a plate with a nitrate disk to perform the indole test, as nitrate can interfere with the spot indole test by inducing false-negative results.
- If the rapid indole test is negative, the isolate could be positive in the more sensitive tube test. Extraction with xylene is the most sensitive test. Xylene substitutes are less sensitive.
- For fastidious Gram-negative rods, such as C. hominis, a heavy inoculum and extraction are necessary.
References and Sources
- Biochemical Tests for the Identification of Aerobic Bacteria. (2016). Clinical Microbiology Procedures Handbook, 3.17.1.1–3.17.48.3.DOI:10.1128/9781555818814.ch3.17.1
- Maria P. MacWilliams. 2009. Indole test protocol.
- Kovac’s Indole Reagent R008. HiMedia Laboratories.
- 5% – https://asm.org/getattachment/200d3f34-c75e-4072-a7e6-df912c792f62/indole-test-protocol-3202.pdf
- 3% – https://microbiologyinfo.com/indole-test-principle-reagents-procedure-result-interpretation-and-limitations/
- 2% – https://himedialabs.com/TD/R008.pdf
- 2% – http://universe84a.com/indole-test-introduction/
- 1% – https://en.wikipedia.org/wiki/Indole_test
- 1% – https://catalog.hardydiagnostics.com/cp_prod/Content/hugo/IndoleTestRgnts.htm


ótimo.