T Cell (T Lymphocyte) Definition
Thymus-derived lymphocytes or T cells are cells of the immune system that are an essential part of the adaptive immune system.
- These are blood cells (leukocytes) that act by attacking invading pathogens and host cells based on the type of T cell activated.
- T cells are different from other lymphocytes as these have a T-cell receptor on the surface, which is absent in other lymphocytes.
- T cells are one of the crucial factors in the adaptive immune response as the receptors interact with MHC complexes on antigen-presenting cells exposed to antigens. The range of antigens that can activate a T cell response differs in different organisms.
- T cells can be distinguished from other lymphocytes like B cells as the maturation and differentiation of T cells occurs in the thymus.
- There are different subsets of T cells that are involved in different functions of the cells. These cells are the primary reason for immune-mediated cell death.
- T cells are produced in the bone marrow every day in a process regulated by a set of T cells called a regulatory T cell.
- T cells are a part of the adaptive immune system with a set of randomly generated membrane receptors to encounter antigens.
- T cells are also different from B cells in that they cannot function as antigen-presenting cells and require antigens to be presented to them via MHC molecules.
- T cells account for about 60-70% of the total lymphocytes present in the blood, but these are quite difficult to isolate and define compared to B cells.
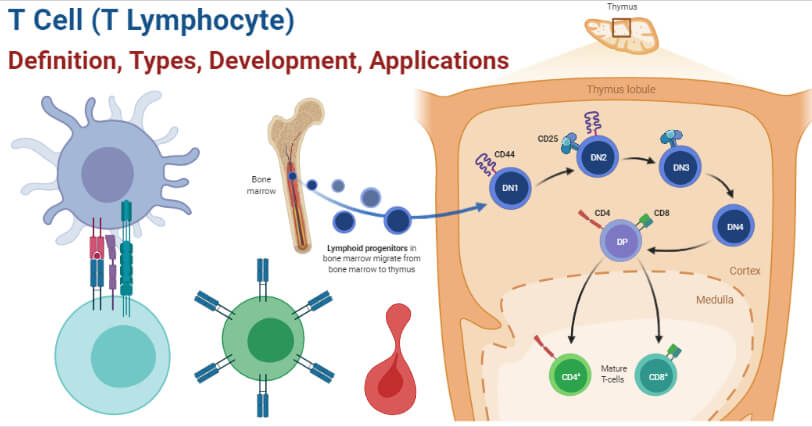
T Cell (T Lymphocyte) Types
T cells can be grouped into categories depending primarily on their function. Other factors that define these categories are gene or protein expression patterns. The following are different types of T cells;
1. Helper T cells (CD4+ T cells)
- Helper CD4+ T cells or T helper cells are lymphocytes that assist the maturation of other lymphocytes like B cells to differentiate into plasma cells and memory B cells.
- CD4+ T cells are activated by class II MHC molecules that are named after the CD4 receptors present on their membranes.
- Helper T cells account for about 50-60% of total T cells, which then further activate other immune cells to protect the body from attacks by foreign particles.
- The activation of CD4+ T cells results in the differentiation of the cells and secretion of cytokines to regulate the overall immune response.
- These cells can further be differentiated into other subtypes, which depends on the type of cytokines produced.
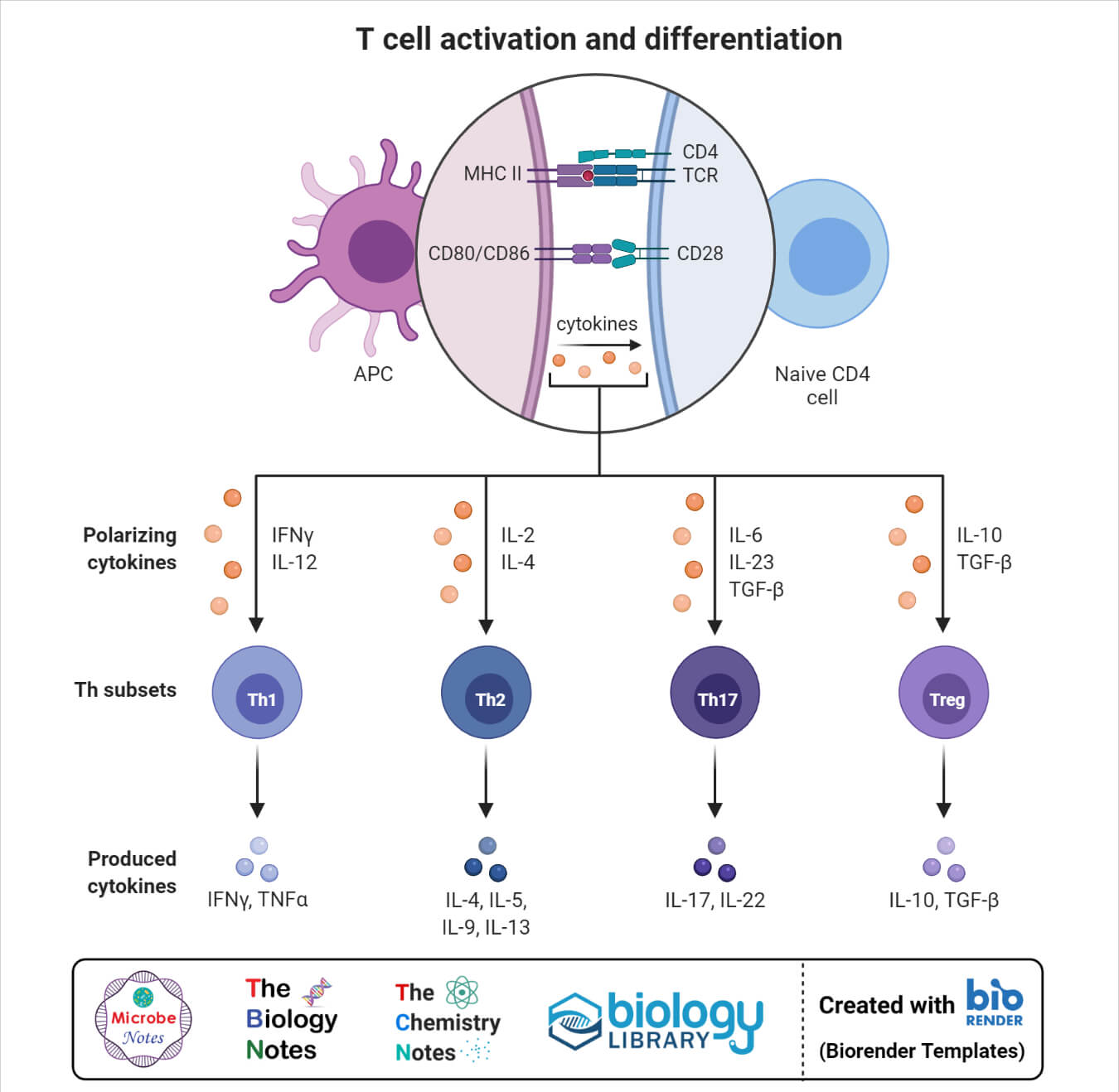
- The two subsets of helper T cells are Th1 and Th2 cells based on the type of toll-like receptors involved in the activation of the T cells.
- The Th1 cells are involved in attacks on pathogens by increasing the phagocytic activity of the macrophages, whereas the Th2 cells are involved in B cell activation.
- Th2 cells are also involved in opsonization, where they coat the surface of pathogens to induce phagocytosis.
2. Cytotoxic T cells (CD8+ T cells)
- Cytotoxic T cells or CD8+ T cells are the T cells activated by the class I MHC molecules on the antigen-presenting cells.
- These are named CD8+ T cells as these contain the CD8 receptors on the surface. The receptor is present in about 40% of the total T cells.
- The CD8+ T cells, after activation, differentiate into the cytotoxic or killer T cells by the process of clonal expansion.
- Cytotoxic T cells usually destroy virus-infected cells or tumor cells as well as cells involved in transplants.
- These cells recognize their target cells by binding to short peptides present together with class I MHC molecules.
- Cytotoxic T cells also secrete cytokines like IL-2 and IFN-γ, which regulate the effector functions of other immune cells.
3. Memory T cells
- Memory T cells are the cells that are differentiated from naïve T cells as a result of encountering a complete antigen by an MHC molecule.
- Memory T cells are a class of T cells that are long-lived and can quickly expand into a large number of effector T cells when re-exposed to the antigen that activated them in the first place.
- These cells can express either of the CD4+ and CD8+ receptors but more commonly, these express CD45RO.
- Memory T cells can be further classified into subtypes like central memory T cells, effector memory T cells, tissue-resident memory T cells, and virtual memory T cells.
- The central memory T cells usually express CD44 and can be found in the lymph nodes and peripheral circulation.
- The effector memory T cells express CD45RO and remain in the peripheral circulation and in tissues. These cells terminally differentiate into effector T cells in the presence of a specific antigen.
- Virtual memory T cells are different from other memory T cells in that they do not originate as a result of clonal expansion. These cells are comparatively less in number and mostly remain in the peripheral circulation.
4. Regulatory CD4+ T cells
- Regulatory T cells are the T cells involved in the maintenance of immunological responses and tolerances.
- These cells primarily shut down T-cell mediated immunity at the end of an immune response and also suppress autoreactive T cells that escape the activation during the negative selection process.
- Regulatory T cells are of two classes CD4+ regulatory T cells; FOXP3+ T cells and FOXP3- T cells.
- The group of regulatory T cells can be formed either during the normal process of T cell development in the thymus or be induced peripherally.
- The regulatory cells formed in the thymus are called thymic Treg cells, and the induced cells are called peripherally derived Treg cells.
- Both of these classes of regulatory T cells require the expression of the transcription factor FOXP3, which is also used to identify these cells. Mutations on the FOXP3 genes can affect the regulatory T cell development, leading to autoimmune diseases.
T Cell (T Lymphocyte) Development
- T cells, like all other blood cells, originate from the hematopoietic stem cells present in the bone marrow. In some cases, the development might begin in the fetal liver during embryonic development.
- The cells then develop into multipotent progenitors that retain the ability to differentiate into myeloid and lymphoid cells.
- Further differentiation leads to the formation of a common lymphoid progenitor that can differentiate into T, B, or NK cells.
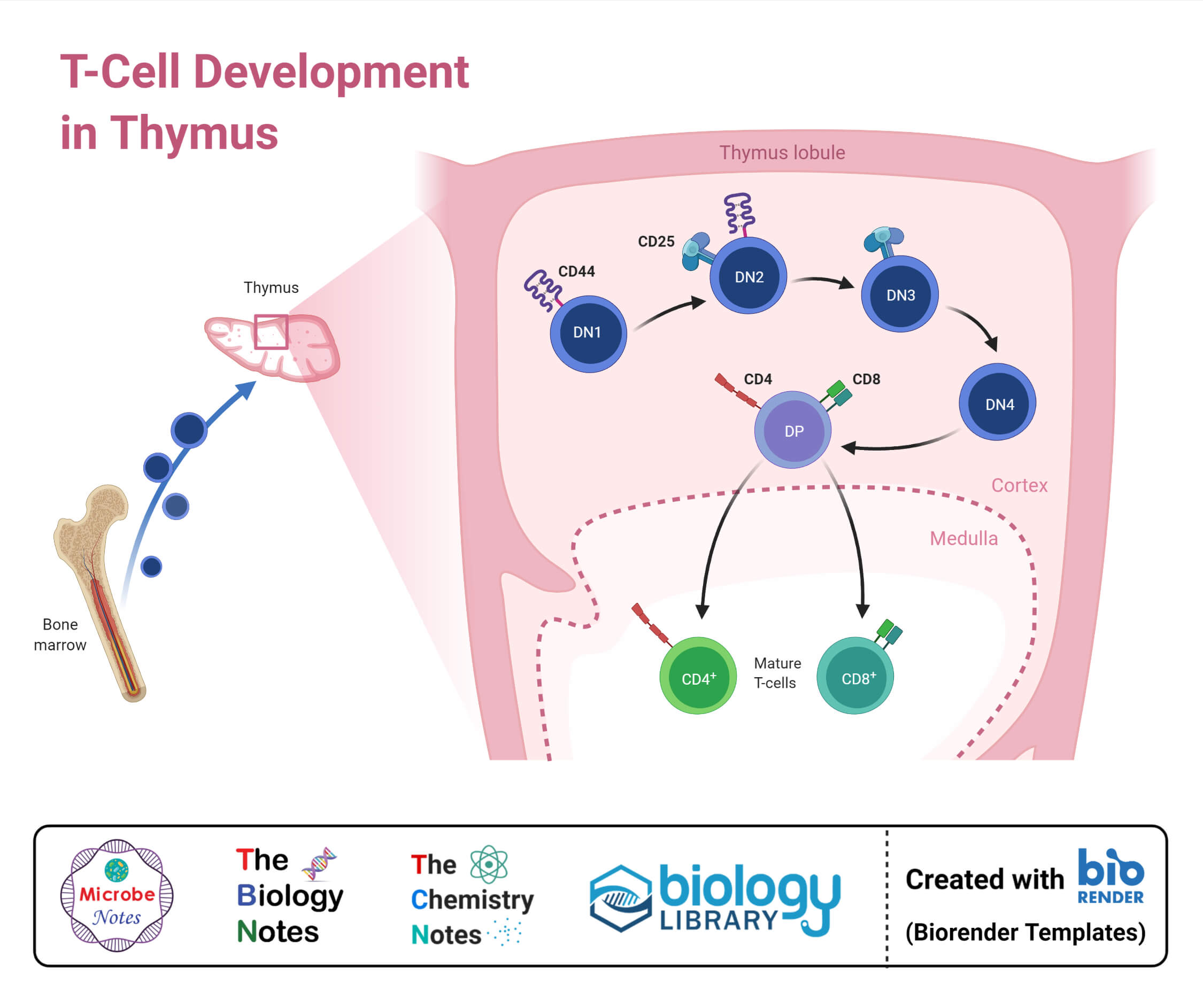
- In the case of T cells, the common lymphoid progenitor cells move to the thymus via blood, where they become engrafted.
- The cells that reach the thymus are called double-negative cells as these do not express any of the CD4 or CD8 co-receptor.
- The cells present in the thymus are not self-renewing the thymopoiesis (formation of T cells) depends on the continuous recruitment of cells from the bone marrow.
1. Maturation
- The cells that enter the thymus encounter the thymic epithelium before progressing to the early thymic progenitor cells (ETP). The cells at this stage are CD4-, CD8-, CD44+ CD25- and ckit+ cells.
- The microenvironment in the thymus restricts the potential of these cells to convert into myeloid and DC cells.
- The thymus consists of four major compartments, where each of them performs a distinct function and regulates different stages of T cell development.
- The compartments are the subcapsular zone, the cortex, the medulla, and the corticomedullary junction.
- The development of the thymocyte progresses through different stages in different regions of the thymus and can be traced by the alterations in the cell-surface marker expression of the molecules.
- The double negative (DN) cells can be further classified into four different stages and are identified by their lack of receptors.
- The DN1 cells are ETPs that exhibit high levels of CD117 and account for about 0.01% of the total thymic T cell pool. The DN1 cells move from the corticomedullary junction into the deeper cortex towards the subcapsular region.
- Here, the cells differentiate into DN2 thymocytes, including CD24+, CD25+, CD44+, and CD117+ cells. The DN2 thymocytes then experience a rearrangement of genes and the secretion of cytokines like IL-7.
- The cells further differentiate into the DN3 stage, where the T cells expressed an invariant α-chain called pre-Tα. The arrangement of genes together with the invariant chain produces signals to proceed with the process of T cell maturation.
- At the DN3 stage, the cells mature into DN4, which is further upregulated into CD4 and CD8 cells achieving a double positive status in the maturation process.
- The specificity and binding strength of the αβ T cell receptor determine the survival and differentiation of the cells.
- The process is followed by two distinct processes; positive selection and negative selection.
a. Positive Selection
- Positive selection is the process of movement of double-positive T cells (CD4+ and CD8+) to the cortex, where they encounter self-antigens.
- The thymic cortal epithelial cells express self-antigens on MHC molecules where the T cells interact with the molecules. The cells that do not interact with the molecules strongly enough due whereas others with high affinity to MHC cells survive.
- A large portion of the developing thymocyte due during the process, which lasts for a number of days.
- In the positive selection, the CD4+ cells interact well with class MHC molecules, whereas the CD8+ cells interact well with class II MHC molecules.
b. Negative Selection
- The cells that survive the positive selection move into the medulla and undergo negative selection, which eliminates thymocytes with a high affinity for self-antigens.
- The cells that interact too strongly with the self-antigens receive an apoptotic signal resulting in cell death.
- During the same process, however, some cells are selected to form Treg cells. The cells that successfully complete the selection process exit the thymus as mature naïve T cells.
2. Activation
- The mature naïve T cells leave the thymus and reach the bloodstream, where they circulate until they recognize their specific antigens on the surface of antigen-presenting cells.
- The activation of CD4+ cells occurs as a result of interaction between the T-cell receptor and a co-stimulatory molecule (CD28 or ICOS) present on the T cell.
- The activation of CD4+ cells is essential for the initial antigenic activation of naïve CD8 T cells and memory T cells.
- The initial signal is provided by the binding of the T cell receptor to the cognate peptide present on the class II MHC. A similar peptide is present on the class II MHC which activates the CD8+ cells.
- The second signal is provided as a result of co-stimulation, where the surface receptors are induced by a relatively small number of stimuli which are products of pathogens or breakdown products of cells.
- Naïve T cells only express the CD28 as a co-stimulatory receptor that interacts with CD80 and CD86 proteins present on APCs.
- The two-step activation process prevents inappropriate responses to self-antigens as self-peptides do not provide suitable co-stimulation.
- After the two signals, T cells also receive stimulation in the form of cytokines. The cytokine signal determines the fate of T cells, especially in the case of helper T cells.
3. Differentiation
- The differentiation of T cells into different types of T cells usually occurs in the form of lineage commitment which is based on the affinity of the T-cell receptor towards self-antigen.
- The decision is to be made during the double-positive thymocyte stage, where the cells determine whether to join the CD8+ cytotoxic T cells or the CD4+ helper T-cell.
- The lineage commitment requires changes in genomic organization and gene expression that results in the silencing of the gene and the expression of a gene associated with a particular lineage function.
- The exact mode of differentiation is not yet clearly understood, but the most recent model that explains the lineage indicates that it is based on the affinity for one of the two MHC classes.
T Cell (T Lymphocyte) Applications
There are different functions and applications of T cells that can be used to protect the body as a part of the immune system. The following are some applications of such applications;
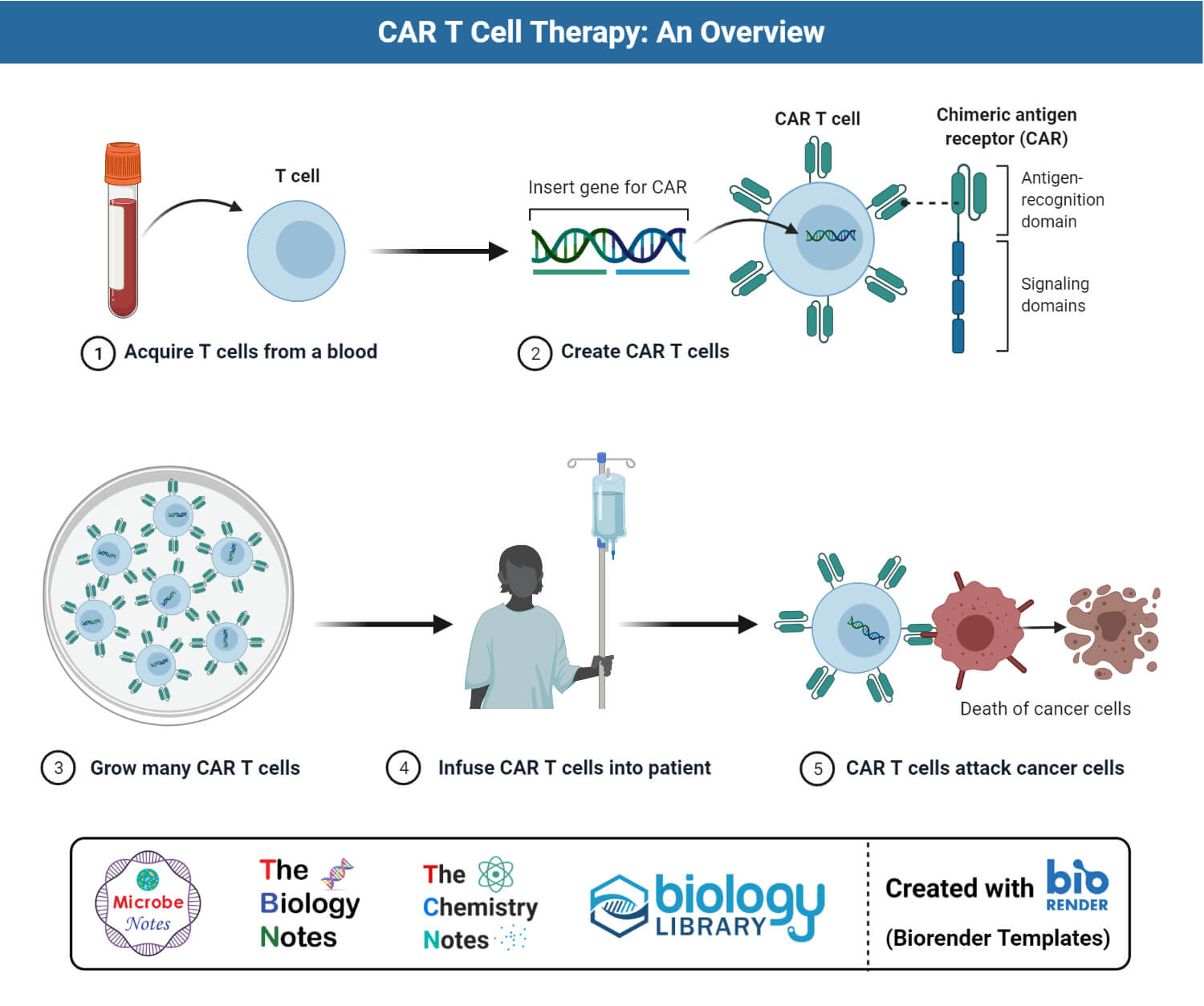
Chimeric antigen receptors (CAR) T cell therapy
Figure: Step-wise overview of Chimeric Antigen Receptor (CAR) T Cell generation and cancer therapy.
- Chimeric antigen receptor (CAR) T cell therapy is one of the promising immunological treatments for cancer which involves the use of engineered chimeric antigen receptors to redirect T cells to targeted malignant cells.
- CAR T cell therapy has been successfully used in clinical trials for the treatment of hematologic malignancies.
- The antigen used as a target is CD19 which is usually expressed on precursor, and mature B cells can be used to treat B cell malignancies as well.
- The technique can also be used for solid tumors, but the efficiency is not as high as in the case of blood cancer therapy.
References
- Peter J. Delves, Seamus J. Martin, Dennis R. Burton, and Ivan M. Roitt(2017). Roitt’s Essential Immunology, Thirteenth Edition. John Wiley & Sons, Ltd.
- Judith A. Owen, Jenni Punt, Sharon A. Stranford (2013). Kuby Immunology. Seventh Edition. W. H. Freeman and Company.
- Koch U, Radtke F. Mechanisms of T cell development and transformation. Annu Rev Cell Dev Biol. 2011;27:539-62. doi: 10.1146/annurev-cellbio-092910-154008. Epub 2011 Jul 5. PMID: 21740230.
- Alberts B, Johnson A, Lewis J, et al. Molecular Biology of the Cell. 4th edition. New York: Garland Science; 2002. T Cells and MHC Proteins. Available from: https://www.ncbi.nlm.nih.gov/books/NBK26926
- Kumar, Brahma V et al. “Human T Cell Development, Localization, and Function throughout Life.” Immunity vol. 48,2 (2018): 202-213. doi:10.1016/j.immuni.2018.01.007
- Zijun Zhao, Yu Chen, Ngiambudulu M. Francisco, Yuanqing Zhang, Minhao Wu. The application of CAR-T cell therapy in hematological malignancies: advantages and challenges. Acta Pharmaceutica Sinica B. Volume 8, Issue 4. 2018. Pages 539-551. https://doi.org/10.1016/j.apsb.2018.03.001.

i like this syllabus
how to cite this article
Sapkota, A. (2022, July 11). T cell (T lymphocyte)- definition, types, development, applications. Microbe Notes. https://microbenotes.com/t-cell-t-lymphocyte/