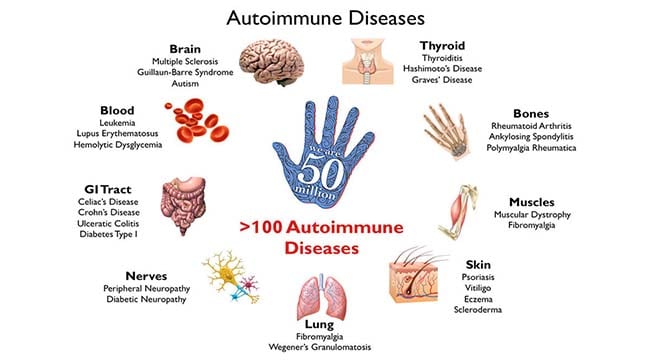
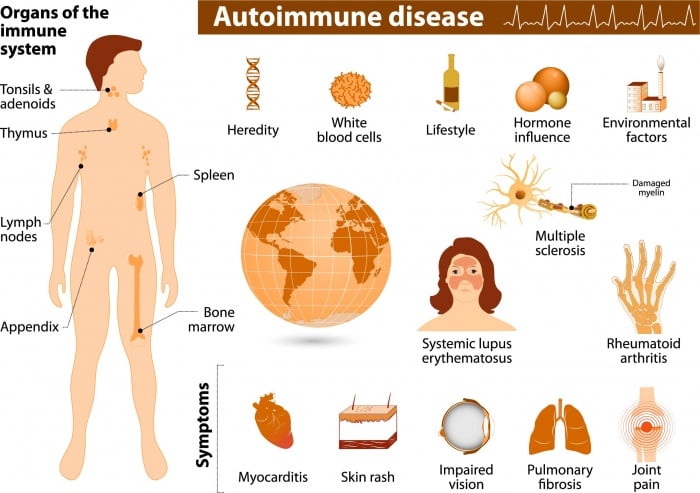
What is an Autoimmune disease?
Autoimmune disease occurs when a specific adaptive immune response is mounted against self-antigens. The normal consequence of an adaptive immune response against a foreign antigen is the clearance of the antigen from the body. Virus-infected cells, for example, are destroyed by cytotoxic T cells, whereas soluble antigens are cleared by the formation of immune complexes of antibody and antigen, which are taken up by cells of the mononuclear phagocytic system such as macrophages. When an adaptive immune response develops against self-antigens, however, it is usually impossible for immune effector mechanisms to eliminate the antigen completely, and so a sustained response occurs. The consequence is that the effector pathways of immunity cause chronic inflammatory injury to tissues, which may prove lethal. The mechanisms of tissue damage in autoimmune diseases are essentially the same as those that operate in protective immunity and in hypersensitivity diseases.
Hypersensitivity Reaction
There are four types of hypersensitivity reaction mediated by immunological mechanisms that cause tissue damage. Types I-III are antibody-mediated and are distinguished by the different types of antigens recognized and the different classes of antibody involved.
- Type I responses are mediated by IgE, which induces mast-cell activation, whereas types II and III are mediated by IgG, which can engage Fc-receptor and complement-mediated effector mechanisms to varying degrees, depending on the subclass of IgG and the nature of the antigen involved.
- Type II responses are directed against cell-surface or matrix antigens.
- Type III responses are directed against soluble antigens, and the tissue damage involved is caused by responses triggered by immune complexes.
- Type IV hypersensitivity reactions are T cell-mediated and can be subdivided into three groups. In the first group, tissue damage is caused by the activation of macro-phages by TH1 cells, which results in an inflammatory response. In the second, damage is caused by the activation by TH2 cells of inflammatory responses in which eosinophils predominate; in the third, damage is caused directly by cytotoxic T cells (CTL).
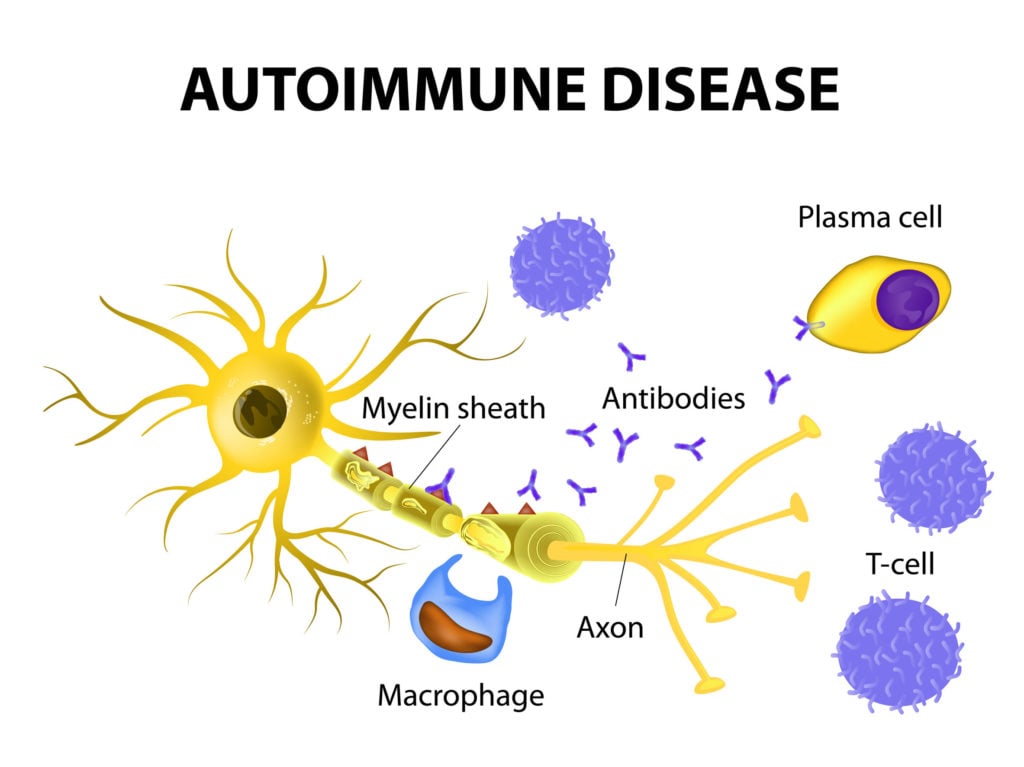
Adaptive immune responses are initiated by the activation of antigen-specific T cells, and it is believed that autoimmunity is initiated in the same way. T-cell responses to self-antigens can inflict tissue damage either directly or indirectly. Cytotoxic T-cell responses and inappropriate activation of macrophages by TH1 cells can cause extensive tissue damage, whereas inappropriate T-cell help to self-reactive B cells can initiate harmful autoantibody responses. Autoimmune responses are a natural consequence of the open repertoires of both B-cell and T-cell receptors, which allow them to recognize any pathogen. Although these repertoires are purged of most receptors that bind with high affinity to self-antigens encountered during development, they still include receptors of lower affinity reactive to some self-antigens. It is not known what triggers autoimmunity, but both environmental and genetic factors, especially MHC genotype, are clearly important. Transient autoimmune responses are common, but it is only when they are sustained and cause lasting tissue damage that they attract medical attention.
Organ-specific and systemic autoimmune diseases
The classification of disease is an uncertain science, especially in the absence of a precise understanding of causative mechanisms. This is well illustrated by the difficulty in classifying autoimmune diseases. It is useful to distinguish two major patterns of autoimmune disease, the diseases in which the expression of autoimmunity is restricted to specific organs of the body, known as ‘organ-specific’ autoimmune diseases, and those in which many tissues of the body are affected, the ‘systemic’ autoimmune diseases. Examples of organ-specific autoimmune diseases are Hashimoto’s thyroiditis and Graves’ disease, each predominantly affecting the thyroid gland, and type I insulin-dependent diabetes mellitus (IDDM), which affects the pancreatic islets. Examples of systemic autoimmune disease are systemic lupus erythematosus (SLE) and primary Sjögren’s syndrome, in which tissues as diverse as the skin, kidneys, and brain may all be affected.
Self-antigen / Auto-antigen
Tissue injury in autoimmune disease results because the self-antigen is an intrinsic component of the body and, consequently, the effector mechanisms of the immune system are directed at the body’s own tissues. Also, because the adaptive immune response is incapable of removing the offending autoantigen from the body, the immune response persists, and there is a constant supply of new autoantigen, which amplifies the response. An important exception to this rule is type I IDDM, in which the autoimmune response destroys the target organ completely. This leads to a failure to produce insulin one of the major autoantigens in this disease. Lack of insulin is in turn responsible for the phenotype of diabetes mellitus.

informative
very nice