Lactose is a disaccharide sugar molecule formed by the combination of glucose and galactose. It is mainly present in milk and milk products. Some bacteria can ferment lactose into glucose and galactose and derive their energy. The breakdown of lactose depends upon two inducible enzymes, the permease, and the β-galactosidase. The production of these enzymes is induced if the culture media contains abundant lactose but lacks glucose. If the bacteria don’t produce the β-galactosidase enzyme, they can’t ferment lactose at all. But, if the bacteria lack only permease enzyme, they may ferment it.
Some bacteria produce both of these enzymes. The permease facilitates the entry of lactate inside the bacterial cell, and the β-galactosidase breaks the lactose into glucose and galactose. The resulting glucose and galactose can be used by bacteria to release metabolic energy. Such bacteria that have the ability to encode both enzymes are rapid fermenters of lactose.
However, some bacteria lack permease enzymes and/or both enzymes. If the bacteria lack the permease, they appear as late lactose fermenters or non-lactose fermenters. Therefore, if the bacteria lack a permease enzyme, it is very difficult to separate the lactose fermenter, late lactose fermenter, and non-lactose fermenter using a traditional lactose fermentation test using lactose as a substrate.
In order to remove this difficulty, a new testing method using ‘o -Nitrophenyl -β- D- galactopyranoside’ (ONPG) was devised called the ONPG test. The ONPG is a colorless synthetic compound similar to lactose in structure which splits and develops color in the presence of the β-galactosidase enzyme.
Objectives
- To verify the ability of the bacteria to encode the β-galactosidase enzyme.
- To differentiate lactose fermenters and non-lactose fermenters.
Principle of ONPG Test
Two enzymes, the β-galactosidase enzyme and permease enzyme, play a major role in the lactose fermentation process. Lactose fermenting bacteria can either synthesize the β-galactosidase enzyme only or both of the enzymes. If bacteria synthesize both enzymes, then the permease facilitates the rapid intake of lactose molecules so that the bacteria can quickly ferment the lactose. However, if bacteria synthesize the β-galactosidase enzyme only, then it ferments lactose very slowly.
If ONPG is added in the culture medium as a substrate instead of the lactose, it is acted upon by the β-galactosidase enzyme in a manner similar to the lactose. The ONPG is structurally similar to lactose and can penetrate the bacterial cell without the permease enzyme. Once inside the bacterial cell, the β-galactosidase enzyme breaks the β-galactoside bond in the ONPG, releasing galactose and o-nitrophenol. The o-nitrophenol is yellow in color and hence causes the development of visible yellow color, indicating the bacteria is a lactose fermenter.
As ONPG doesn’t require a permease enzyme, it gives similar results in a very short time even though the test bacteria lack permease and contain only β-galactosidase.
Requirements for ONPG Test
a. Culture Media
The ONPG test can be performed by using ONPG broth or ONPG disk.
Composition of ONPG Broth per 1000 mL
Casein Peptone (or casitose)- 7.50 grams
o -Nitrophenyl -β- D- galactopyranoside- 1.50 grams
Disodium hydrogen phosphate- 0.35 grams
Sodium Chloride- 3.75 grams
Final pH 7.5±0.2 at 25°C
(Reference: ONPG Broth (himedialabs.com))
Preparation of ONPG Broth
- Measure the appropriate amount of ONPG broth medium as directed by the manufacturing company and dissolve it with the sterile water of the required volume in a conical flask (or glass bottle)
- Heat to dissolve the medium, but do not autoclave the medium (The broth can be stored at or below – 10°C away from light.)
- Dispense about 5 mL (or the required quantity) of the broth in sterile test tubes.
For the ONPG disk method, bacteria can be isolated (grown) in any suitable culture media.
b. Reagents
ONPG disk for performing disk ONPG test
Normal Saline
McFarland Standard No. 2
Composition
1% anhydrous barium chloride (BaCl2) solution
1% sulfuric acid (H2SO4) solution
Preparation of number 2 McFarland Standard Suspension
In a clean and clear test tube, add 9.8 mL of 1% H2SO4 and 0.2 mL of 1% BaCl2 solution
c. Equipment
| Sterile Test-tubes Inoculating Loop | Weighing Machine Autoclave & Incubator | Bunsen burner | Micropipette PPE Other laboratory materials |
d. Test Organism (sample bacteria)
Positive Control: E. coli ATCC 25922 or Neisseria lactamica ATCC 23971
Negative Control: Proteus mirabilis ATCC 12453 or Neisseria gonorrhoeae ATCC 43069
Procedure of ONPG Test
a. Tube Method (ONPG Broth Method)
- Prepare a suspension of pure and fresh (18 to 24 hours old) sample bacteria with turbidity equal to McFarland Standard number 2.
- Heavily inoculate a tube of ONPG broth with the bacterial suspension using a sterile inoculating loop.
- Incubate the tube at 35±2°C aerobically.
- After one hour, observe for color change (development of yellow color). If no color change is observed, continue incubating for up to 24 hours and look for color change.
b. Disk Method
- In a sterile test tube, add 0.5 mL of sterile saline and prepare heavy bacterial suspension (turbidity equal to about number 2 McFarland standards).
- Place an ONPG disk in the suspension.
- Incubate aerobically at 35±2°C aerobically for about 4 to 6 hours.
- Observe for color change (development of yellow color) in the ONPG disk.
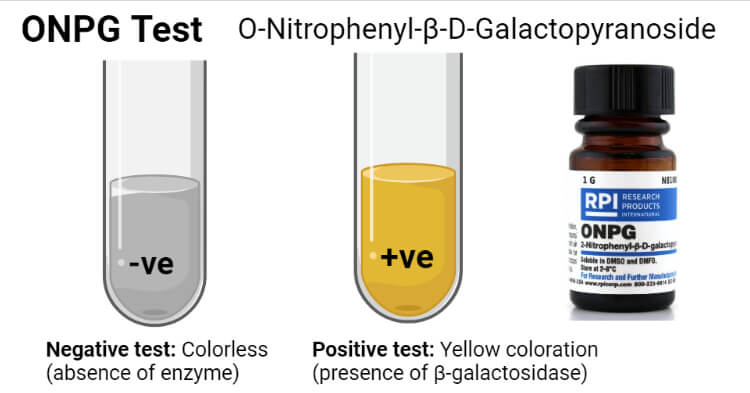
Result and Interpretation of ONPG Test
Positive ONPG test (production of β-galactosidase) = Development of yellow color
Negative ONPG test (lack of β-galactosidase production) = No color development (change)
ONPG Positive Bacteria
E. coli, Citrobacter freundii, Salmonella Choleraesuis, Shigella dysenteriae, Klebsiella pneumoniae (and most Klebsiella spp.), Vibrio cholerae, Serratia marcescens, N. lactamica, etc.
ONPG Negative Bacteria
Proteus vulgaris, P. mirabilis, Salmonella Typhimurium, Vibrio parahaemolyticus, Providencia spp., Pseudomonas aeruginosa, N. gonorrhoeae, etc.
Quality Control
E. coli ATCC 25922 or Neisseria lactamica ATCC 23971 are ONPG positive and cause the development of yellow color in broth (and color change of ONPG disk to yellow)
Proteus mirabilis ATCC 12453 or Neisseria gonorrhoeae ATCC 43069 are ONPG negative and there is no color change in broth (as well as no color change in ONPG disk)
Precautions
- ONPG is highly unstable and it must be purchased or made by skilled personnel in a controlled environment.
- ONPG broth and disk must be stored in a dark area because ONPG is highly photosensitive.
- ONPG broth should not be autoclaved; hence every piece of equipment and saline must be pre-sterilized.
- Use heavy inoculum for testing.
Applications of ONPG Test
- Differentiation of Enterobacteriaceae and Neisseria spp. based on the ability to synthesize β-galactosidase
- Coupled with the lactose fermentation test, it can also determine the ability of bacteria to synthesize the permease enzyme.
- It is used in the biochemical characterization and identification of Gram-negative bacteria.
Limitations of ONPG Test
- Bacteria that naturally produce yellow pigment are not suitable for the ONPG test.
- For the rapid ONPG test, the bacterial colonies cultured on the lactose-containing medium are required.
- The bacterial colonies from the glucose-containing medium show lower activity against ONPG.
- This test is not enough for bacterial identification.
References
- Leber, Amy L., editor in chief. (2016). Clinical microbiology procedures handbook (Fourth edition). Washington, DC : ASM Press 1752 N St., N.W., [2016]
- Tille, P. M., & Forbes, B. A. (2014). Bailey & Scott’s diagnostic microbiology (Thirteenth edition.). St. Louis, Missouri: Elsevier.
- Definition of Lactose – Lactose Intolerance, Causes, Diagnosis & Treatment (byjus.com)
- ONPG: β-galactosidase Test • Microbe Online
- 42: Beta-Galactosidase Test (ONPG) – Biology LibreTexts
- O-Nitrophenyl-b-D-Galactopyranoside (ONPG) Test Principle, Procedure (microbiologynote.com)
- ONPG Test (for β-galactosidase) – How to do (Principle), Results Interpretation and Uses – Laboratoryinfo.com
- O-Nitrophenyl-β-D-Galactopyranoside (ONPG) test: Principle, Procedure and Results – Online Biology Notes
- ONPG test (microbiologie-clinique.com)
- ONPG Test – Principle, Procedure, Uses and Interpretation (microbiologyinfo.com)

S dysenteriae is not ONPG positive
But Shigella dysenteriae is positive for ONPG test. Some of the other sources are given below which tell that S dysenteriae is ONPG positive.
https://pmc.ncbi.nlm.nih.gov/articles/PMC480699/
https://www.bio-rad.com/webroot/web/pdf/fsd/literature/Technical_Sheets/TS_ONPG%20Disks_V3_05-08-11.pdf