Centrifugation is a term used to describe a method of separating mixtures using spinning and centrifugal force. Several characteristics can separate particles during centrifugation, including size, shape, density, and viscosity.
A centrifuge is a lab instrument for the density-based separation of fluids, gas, or liquid.

The idea for the first centrifuge originated with British military engineer Benjamin Robins, who built a device that resembled an arm rotating around an axis to gauge drag on items. The Prandtl brothers later made the first practical centrifuge to separate milk from the cream by improving this design. By rapidly rotating a container containing material, separation is accomplished; the centrifugal force forces heavier elements to the outside of the container.
Centrifuges are used in daily life as well as in science and medical research. Cells, subcellular organelles, viruses, proteins, and nucleic acids can all be purified with it.
Principle of a Centrifuge
The centrifuge utilizes the sedimentation principle due to gravitational force. The centrifugation technique uses a centrifugal field to separate particles suspended in a liquid medium. These are put in the centrifuge’s rotor either in bottles or tubes. Sedimentation is a process whereby gravity causes suspended particles to separate from fluids. The suspended substance may consist of powder or clay-like particles.
Simple filtration filters particles larger than 5 micrometers from those less than 5 micrometers, which start Brownian motion and do not sediment under gravity. These particles can be separated with the help of the central force.
Parts of a Centrifuge
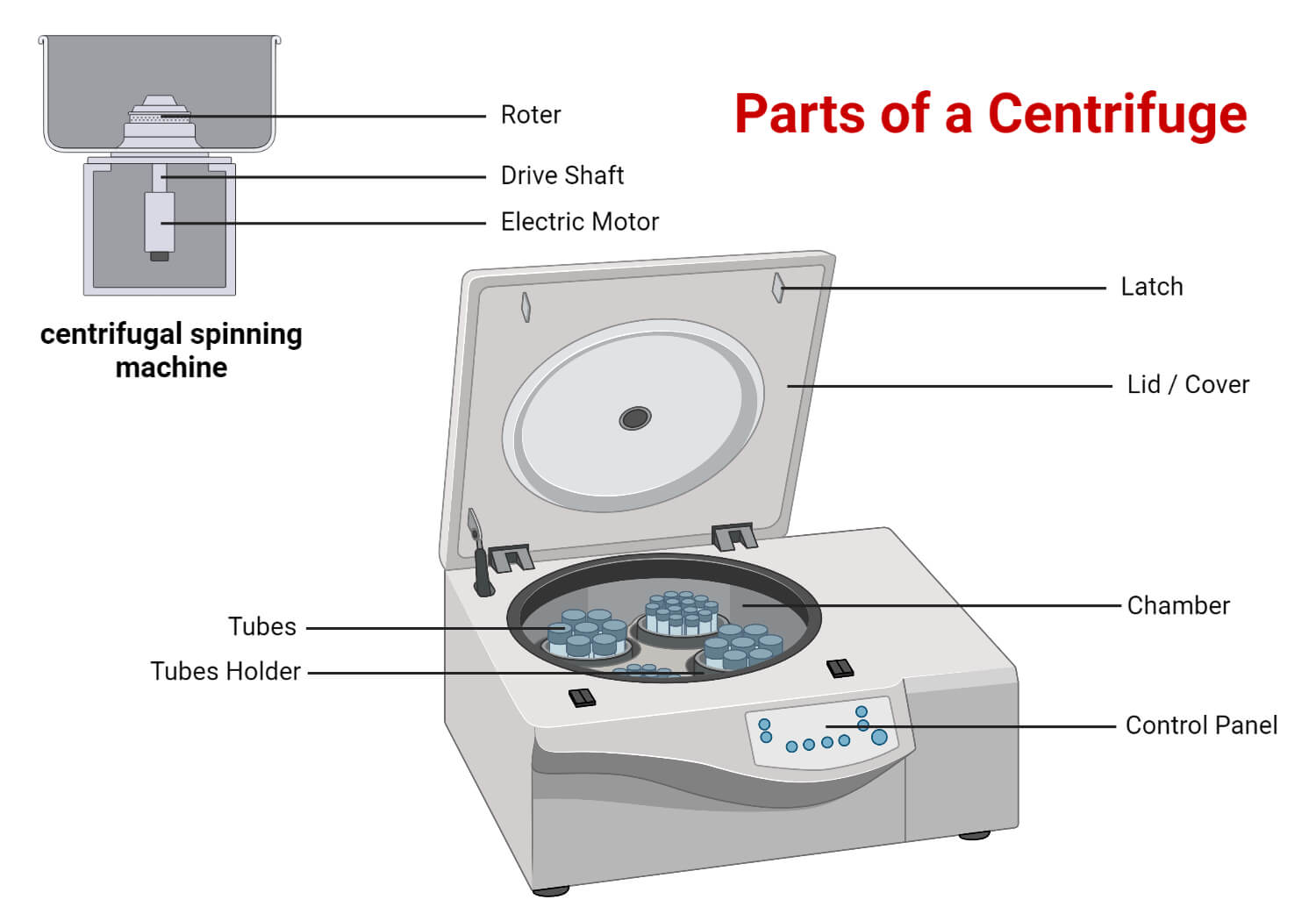
Some of the common parts of the centrifuge are described below:
- Motor: The motor is the powerful central component of the centrifuge that creates the spin.
- Rotor assembly: A drive shaft and a rotor comprise the rotor assembly. The drive shaft provides support for the rotor components. The rotor head is attached to the motor, which bears the containers to house the tubes containing the sample to be centrifuged. It converts electrical energy to mechanical energy. Two rotors with different diameters can have the same rotational speed. Varying radii and angular momentum results in a difference in the acceleration of such rotors. Thus, relative centrifugal force (rcf) is regarded as the accepted standard unit for the rotation speed. There are mainly three types of rotors:
a) Fixed angle rotors: These rotors hold the tubes at an angle of 14 to 40° to the vertical such that particles travel a short distance while moving radially outwards and are used in differential centrifugation. The sedimentation takes place at the walls of the tubes at an angle since the sedimentation direction is the same as the direction of centrifugal force. The pellets (cluster of sediments) later settle at the corner of the base and the wall surface after colliding with the wall surface.
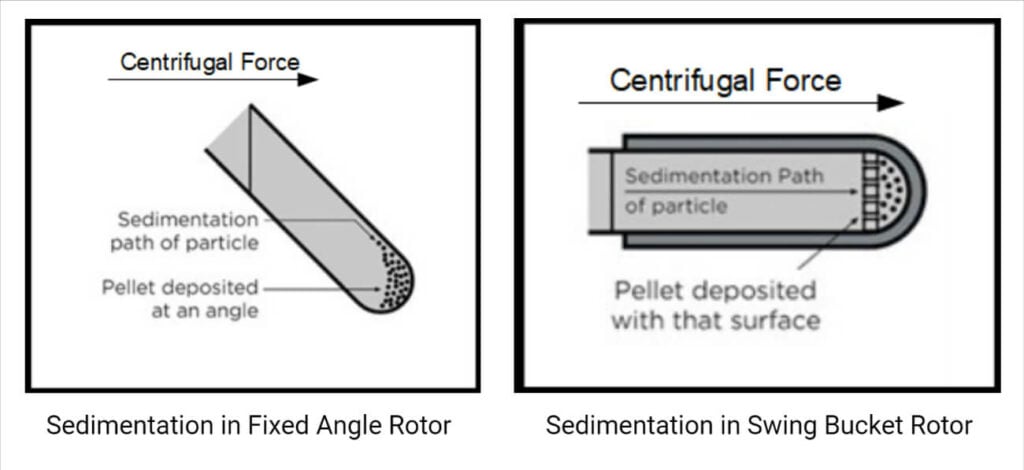
b) Swinging bucket/ Horizontal rotors: These rotors, along with the centrifuge tubes, swing out to a horizontal position during the time of acceleration such that particles travel a longer distance, thereby facilitating easier separation of supernatant from the pellet. These types of motors are employed in density gradient centrifugation.
c) Vertical rotors: These hold the tubes vertically, i.e., parallel to the motor axis, and the particles move shorter distances with shorter periods for separation. It is used for isopycnic and density gradient separation; however, it is not considered useful for pelleting because the pellets are spread out along the entire outer wall of the tube by centrifugal force.
- Containers: Several types of containers, such as test tubes, blood bags, cuvettes, centrifuge tubes, etc., are held in the rotors such that the sample rotates along as the rotor rotates.
- Control Panel: It serves the purpose of controlling different parameters such as temperature, rotational speed (rcf or rpm), etc.
- Latch: When a tube breaks, or there are other issues with the centrifuge while running, the latch keeps the lid closed.
- Lid: The centrifuge will only spin if the lid is closed and locked to prevent mishaps.
Types of centrifugation techniques
There are two types of centrifugation techniques, namely, preparatory centrifugation and analytical centrifugation. Preparatory centrifugation deals with the isolation and purification of components such as tissue, cells, subcellular structure, membrane vesicles, and other particles of biochemical interest. In contrast, analytical centrifugation is carried out to characterize purified biomolecules.
Preparative Centrifugation
Based on suspension, preparative centrifugation is divided into two different types. They are:
i) Differential centrifugation
It separates particles based on shape, size, and density. A suspension of particles with varying densities or sizes will sediment at varying speeds, with the larger and denser particles sedimenting more quickly. Following a series of rising centrifugal force cycles on a suspension of cells, a series of pellets containing cells with a decreasing sedimentation rate will result.

ii) Density gradient centrifugation
It separates particles based on their buoyant density or sedimentation rate. A sample mixture is placed on the top of a preformed liquid density gradients such as CsCl for DNA banding and isolation of plasmids, nucleoproteins, and viruses; NaBr and NaI for fractionation of lipoprotein; Per coll, Ficoll, Metrizamide, Dextran for separation of whole cells and sucrose solution for the separation of DNase, RNase and Protease.
The two subtypes of density gradient centrifugation are rate-zonal and isopycnic centrifugation.
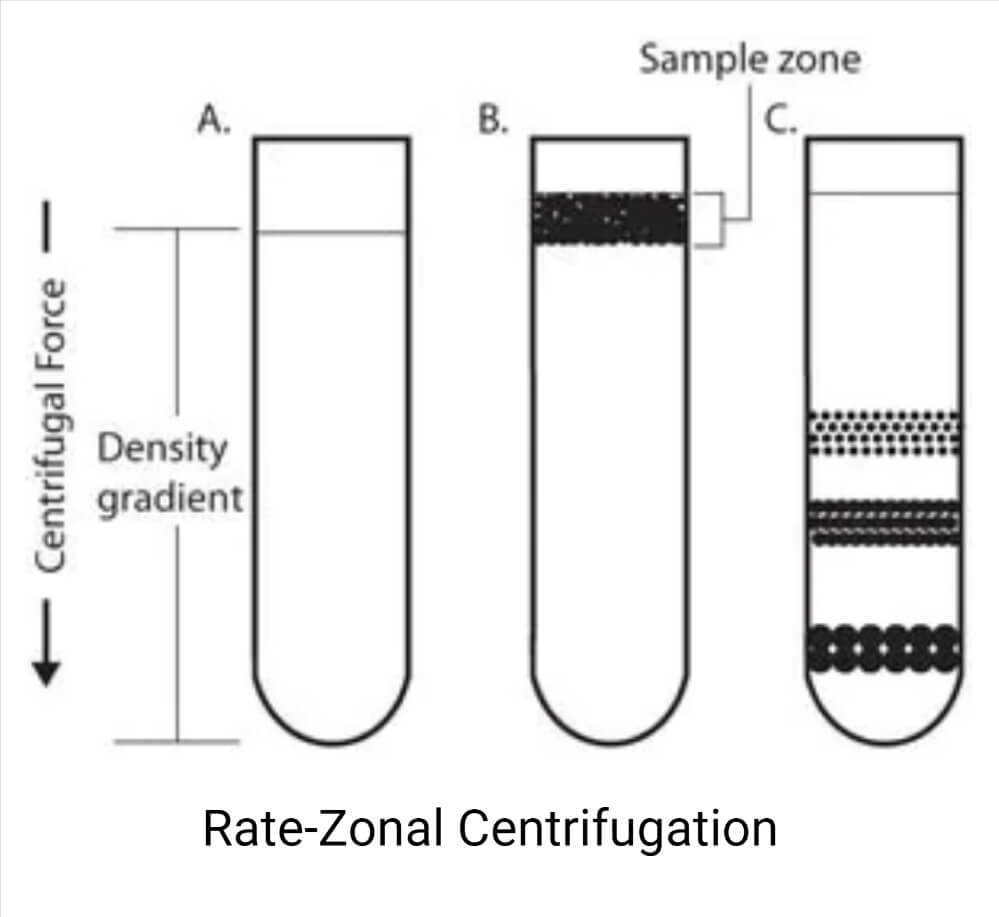
Rate-zonal centrifugation
On top of a density gradient, the sample is overlaid as a small zone. Depending on their mass, particles travel under centrifugal force at various speeds. Size and mass are the main determinants of how quickly particles settle. As the band of particles descends through the density medium, zones with particles of comparable size develop as the faster sedimenting particles pass the slower ones.
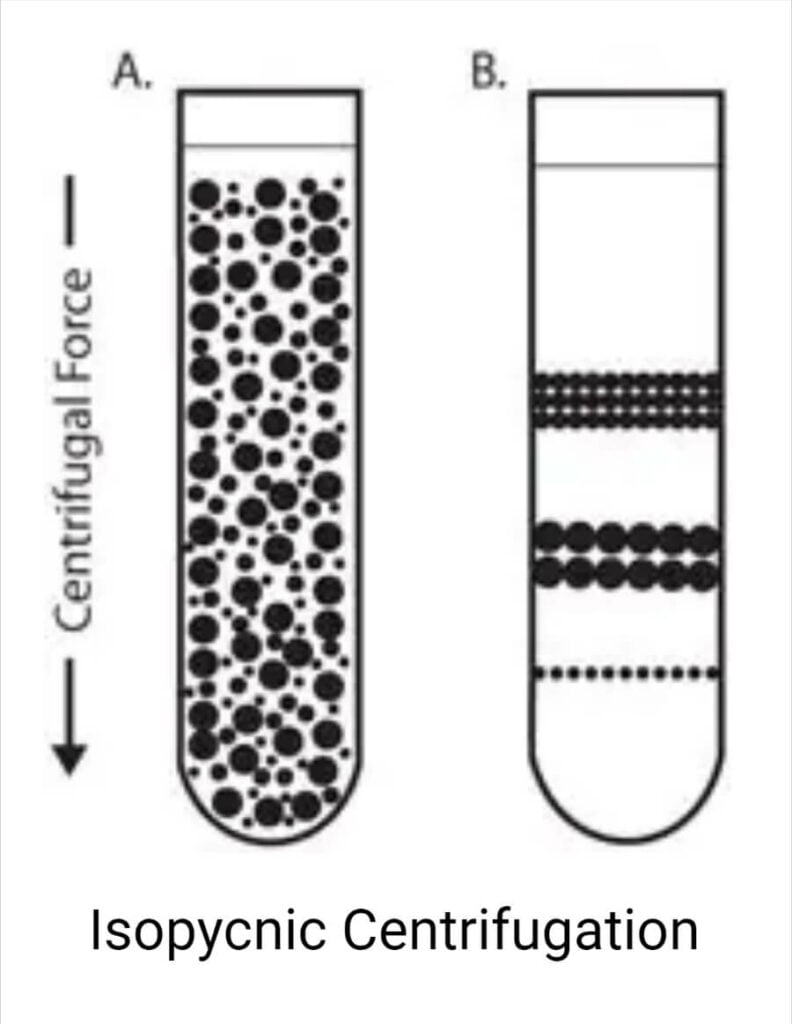
Isopycnic centrifugation
Particles are separated exclusively based on their density in an isopycnic separation, also known as buoyant or equilibrium separation. It is necessary for the gradient medium to have a higher density than the particles that need to be separated.
Particles migrate under the influence of centrifugal force from a uniformly mixed sample and density gradient until their densities are equal to those of the surrounding medium. After centrifugation, particles of a certain density settle until their density equals that of the gradient media (i.e., the equilibrium position).
Analytical Centrifugation
It aims to collect information to characterize the spun sample (sedimentation velocity, viscosity, concentration, etc.), determine the relative molecular weight of the solutes, purity of biomolecules, detect conformational changes of protein structure, etc.
Types of Centrifuges
Benchtop or Tabletop centrifuges
- They can be handy for tiny labs with limited space because of their diminutive size.
- These are compact and are frequently employed in research and clinical laboratories.
- A tabletop centrifuge is furnished with a lid that covers the apparatus used to run the centrifuge and a rotor with racks for the test tubes.

Gas centrifuges
- These are used to separate molecules based on their masses and to separate gases based on their isotopes.
- Specifically, they are employed in the extraction and separation of uranium-235 and uranium-238.
Haematocrit centrifuge
- Haematocrit centrifuges operate between 7000 and 15000 rpm.
- The main purpose of hematocrit centrifuges is to calculate the volume-based erythrocyte percentage in blood. It is used to produce plasma for photometric analysis of the bilirubin concentration of neonatal blood.
Microcentrifuge
- They have a very small footprint and take up minimal room on the workstation because of their highly compact form.
- These work well with small tubes (up to 2.0 ml) and are frequently employed in biological applications.
- They are used to microfilter small amounts of aqueous samples and hold pelleted nucleic acids, proteins from solutions, and other substances.
Refrigerated Centrifuges
- These centrifuges run at their top speeds while keeping a constant temperature.
- It is used to analyze DNA, RNA, PCR, and antibodies because its temperature range is between -20 and -40 degrees Celsius.
- They are frequently used to collect sedimenting materials quickly, including yeast cells, chloroplasts, and more.
High-Speed Centrifuges
- A high-speed centrifuge is a type of centrifuge that can work at somewhat faster rates ranging between 15,000 and 30,000 revolutions per minute.
- High-speed centrifuges contain a device for regulating both the temperature and speed of the operation for the critical analysis of delicate biological molecules.
- These centrifuges employ three rotors: fixed angle, swinging bucket, and vertical.
Low-speed centrifuges
- These are frequently used in laboratories for routine particle sorting operated at a maximum speed of 4000-5000 rpm.
- There are few instances of temperature regulation, and they are often operated at room temperature.
- These centrifuges employ swinging bucket and fixed-angle rotor types.
Continuous flow centrifuges
- It enables the centrifugation of large quantities of samples without influencing sedimentation rates.
- They also have greater capacities, which saves time by eliminating the need to load and unload the sample repeatedly as is necessary with standard centrifuges.
Ultracentrifuges
- The ultracentrifuge is a highly developed and sophisticated centrifuge that can separate tiny molecules that conventional centrifuges can’t separate at a fast rate.
- Ultracentrifuge rotor speeds can range from 60,000 to 150,000 rpm.
- They run samples in groups or as continuous flow systems and are larger.
Types of Ultracentrifuges
- Preparative Ultracentrifuges
- Preparative ultracentrifuges are centrifuges that are used to isolate and separate particles inside an experiment using centrifugation.
- When a run is prepared for an ultracentrifuge, the contents of the tubes are examined after the centrifugation procedure, as opposed to the centrifuge for analysis, which examines during the centrifugation.
- Analytical Ultracentrifuges
- Analytical centrifuges are ultracentrifuges used to examine different particles inside the specimen.
- It is utilized to perform a qualitative examination of macromolecules in solution.
- These are fitted with sensing devices that track the spin and movement of the constituents in real time to calculate the sedimentation coefficient.
Centrifuge Operating Procedure
- Check the centrifuge to ensure it operates properly, is unharmed, and can move without restriction.
- After choosing the proper centrifuge tubes or containers, check them to ensure no blemishes or cracks. Any tubes or containers with damage or flaws should be discarded.
- Fill the preferred liquid in the tubes. Never overfill or underfill tubes or containers.
- Check that the centrifuge tubes are balanced; weigh each tube separately on a scale to confirm that their weights are equal. Avoid balancing solely on volume! This is especially true for solutions containing various sample types or varied sample concentrations.
- Screw the centrifuge tubes’ lids on firmly.
- Before inserting the centrifuge tubes, ensure their exteriors are dry and clean.
- Keep the tubes in the centrifuge balanced.
- Shut the lid. Ensure that the lid fits tightly.
- Configure the run time and speed.
- When the centrifuge appears to be running and is running at maximum speed, do not leave the device. Check for any unusual vibrations or noises.
- Turn the centrifuge off and remove the sample as soon as you hear an odd noise or feel strong shaking. A typical cause of this is an improperly balanced centrifuge. Do not use the centrifuge until it has been repaired if the issue does not go away despite the centrifuge being properly balanced.
- After the centrifuge has finished its cycle, wait until it has stopped rotating before opening the cover. Never touch or open the centrifuge’s lid before it has stopped rotating. Mechanical failure and harm might result from prematurely stopping the equipment.
- To allow the aerosols that were emitted during centrifugation to settle, it is best to wait at least 10 minutes after the rotation has stopped before opening the lid.
- Samples can be taken out of the centrifuge once it has stopped spinning.
Applications of Centrifuge
- Centrifuges are employed in chemistry, biology, biochemical, and clinical laboratories, such as testing the sedimentation rates of various blood cells.
- These are utilized in dairy industries to separate cream (fat) from milk, and this process is known as churning.
- Giant centrifuging machines are used in water treatment, where it spins the mud and sludge out of the water to produce cleaner water. Likewise, solid matter is separated from freshly drilled-out petroleum in oil rigs.
- Moreover, big spinning wheels are used to simulate a high-gravity environment to practice for the pilots. Hence, these are important in aeronautics and space.
- Centrifugation is used to produce biological products and bulk drugs and perform biopharmaceutical analysis of drugs.
- It is applied in removing water from lettuce after washing it in a salad spinner and separating chalk powder from water.
- It is useful for separating the isotopes for nuclear weapon programming.
Advantages of Centrifuge
- Enclosed operation and consequently clean appearance
- Quick start-up and shutdown
- Easy automation and continuous operation if necessary
- Low capital cost-to-capacity ratio
- Quick adjustment of operating parameters
- High flexibility and outstanding performance
- Simple operation and easy installation
Limitations of Centrifuge
- Separating light particles (with almost negligible mass) is exceedingly difficult.
- A centrifuge, despite its kind, is a very sophisticated electrical device that requires specialist servicing in times of malfunction, making device maintenance extremely difficult and expensive.
- High energy usage because it has so many power-consuming features such as temperature control and rotor spinning.
- Due to their high rotation rates per minute, centrifuges produce noise disruptions. Since it uses rotations, vibrations are unavoidable, which will cause noise pollution and prevent its use in some areas.
- Most often, a backup machine is required to step in when the primary equipment breaks down.
- Extensive use of polymers
Precautions
- Before the operation, always check that the centrifuge is on a suitable surface.
- Keep the lid closed while the rotor is operating.
- When the centrifuge shakes or vibrates, unplug it.
- Make sure tubes work with programs and settings before using them. The tubes used for the centrifuge should be in a set that is matched.
- Balanced neighboring and opposing loads should be used to load tubes symmetrically. Use mass instead of volume to balance the tubes.
Centrifuge Examples
Laboratory centrifuge MC-20000 (Medline Scientific)
- RCF value is automatically shown
- Equipped with microprocessor control, brushless motor, and widescreen digital display showing speed, time, temperature, etc.
- Built-in safety systems include electronic door locks, unbalance, over-speed, and over-temperature alerts.
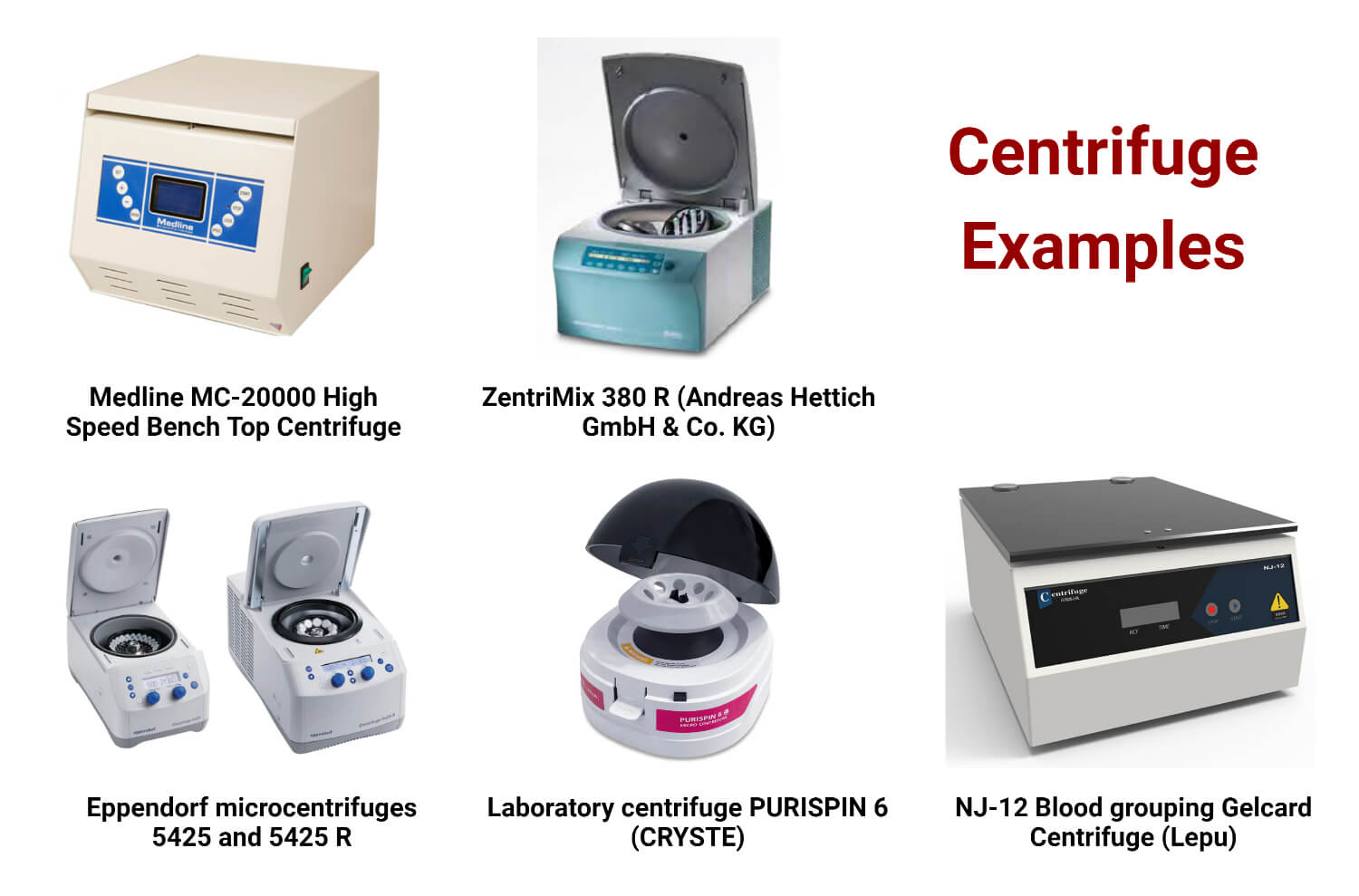
Analytical centrifuge ZENTRIMIX 380 R (Andreas HETTICH GmbH & Co.KG)
- Numerous common operations in research and analytical laboratories are facilitated by the dual centrifuge ZentriMix 380 R.
- Creating nanoparticles in sealed (sterile) vials or mixing heavy or difficult-to-mix components are two common applications.
- The noise level is minimal during the performance compared to typical lab shakers.
Laboratory microcentrifuge 5425 (Eppendorf SE)
- The Centrifuge 5425 R’s upgraded cooling system guarantees optimum separation outcomes and consistent cooling at 4°C, even when the rotor operates at its highest speed.
- Centrifuging samples in 18 spin columns and 1.5 or 2.0 mL tubes at a maximum speed of 18,565 x g is possible with the Eppendorf Kit rotor F-45-18-11-Kit.
Laboratory centrifuge PURISPIN 6 (CRYSTE)
- Excellent Durability.
- Easy to Operate.
- Quiet Operation.
- Compact Design.
Blood bank centrifuge NJ-12 (Lepu Medical Technology)
- Maximum speed: 3000 rpm.
- Precision at high speed.
- Little noise.
- Automated oversight of the entire procedure.
References
- https://www.genfollower.com/centrifuges-types-uses-in-laboratories/
- https://study.com/academy/lesson/what-is-centrifugation-definition-process-uses.html
- https://druckerdiagnostics.com/knowledge/how-a-centrifuge-works/
- https://www.biocompare.com/Lab-Equipment/Laboratory-Centrifuges/
- https://www.toppr.com/guides/chemistry/is-matter-around-us-pure/centrifugation/
- https://www.tec2med.com/centrifuge-rotor-rcf-rpm/
- https://www.iqsdirectory.com/articles/centrifuge.html
- https://www.sigmaaldrich.com/NP/en/technical-documents/technical-article/protein-biology/protein-pulldown/centrifugation-separations
- https://www.mls.be/en/p/lab-apparatus/centrifuges/centrifuges-hettich/hematocrit-centrifuges/
- https://microbiologynote.com/ultracentrifuge-definition-principle-types-uses/
- https://www.ehs.uci.edu/sop/hazardous-operations/centrifuge-sop.pdf
- https://www.medicalexpo.com/prod/andreas-hettich-gmbh-cokg/product-67600-888341.html
- https://www.medicalexpo.com/prod/medline-scientific/product-76782-1005134.html
- https://www.medicalexpo.com/prod/eppendorf-se/product-68382-849100.html
- https://www.medicalexpo.com/prod/cryste/product-105953-1030380.html
- https://www.medicalexpo.com/prod/lepu-medical-technology/product-95737-988153.html

you should’ve add ideal rpm speed for different types of specimen
Thank you Prakriti, I had to prepare a presentation on centrifugation in my M.sc 1st semester and your notes have been really helpful to understand this topic.
These are really nice and organized notes.
smith
thanks for that
I found it very easy to understand.
Thank you.
Very educative
Really, it’s a very good article.