- The Venereal Disease Research Laboratory test (VDRL) is a screening test for sexually transmitted infection – syphilis caused by the spirochete bacterium Treponema pallidum.
- It is a non-treponemal test, which detects antibodies IgM and IgG antibodies to lipoidal material released from damaged host cells as well as to lipoprotein-like material, and possibly cardiolipin released from the treponemes. These antibodies are traditionally referred to as ‘reagins’.
- The VDRL test is thus slide micro flocculation test used to screen for syphilis in addition to more specific tests used to diagnose the disease.
- VDRL test becomes positive 1-2 weeks after appearance of (primary lesion) chancre. The test becomes reactive (50-75%) in the late phase of primary syphilis, becomes highly reactive (100%) in the secondary syphilis and reactivity decreases (75%) thereafter.
Principle of VDRL Test
The Venereal Disease Research Laboratory (VDRL) tests are slide microflocculation test that detect antibodies produced against antigens released by damaged host cells in patients suffering from syphilis.
It uses antigen (referred to as VDRL antigen) containing 0.03% cardiolipin, 0.21% lecithin, and 0.9% cholesterol. The antigen, suspended in a buffered saline solution, forms flocculates when combined with lipoidal antibodies in serum or cerebrospinal fluid from syphilis patients.
For the test, at first a drop of antigen is placed on a slide and then a drop of serum is added to it. The slide is rotated to mix the content. In case of positive test, flocculation occurs which are read using a microscope. Clumping or agglutination indicate reactive specimen or presence of autoantibody in patient’s specimen while non-reactive specimens appear as homogeneous suspension.
Requirements of VDRL Test
Patient’s serum, water bath, freshly prepared cardiolipin antigen, VDRL slide, mechanical rotator, pipettes, and hypodermic syringe with unbeveled needle and microscope. Known reactive and non-reactive serum controls are also required.
Procedure of VDRL Test
The test can be performed both qualitatively and quantitatively. Those tests that are reactive by qualitative test are subjected to quantitative test to determine the antibody titres.
Qualitative Method
- Patients’ serum is inactivated by heating at 56oC for 30 minutes in a water bath to remove non-specific inhibitors (such as complement).
- VDRL antigen suspension (colloidal suspension of tissue cardiolipid or chemically synthesized cardiolipin), controls and samples are brought to room temperature.
- One drop (50 µl) of the test specimen, positive and negative controls is pipetted onto separate reaction circles of the disposable slide.
- A drop of diluted antigen suspension is added to the measured volume of specimen, positive and negative controls.
- Using a mixing stick the test specimen and the VDRL reagent is mixed such that it thoroughly spreads uniformly over the entire reaction circle.
- The slide gently rotated and continuously either manually or on a mechanical rotor at 180 r.p.m.
- Flocculation is checked microscopically using 10X objective and eye piece at about 8 minutes.
Quantitative Method
- Dilute serum samples to an endpoint titer. Quantitative tests for 3 serum specimens through the 1:8 dilution may be performed on one slide.
- Place 50 µl of 0.9% saline in circles numbered 2 through 4. Do not spread saline.
- Using a safety pipette device, place 50 µl of serum in circle 1 and 50 µl of serum in circle 2.
- Mix the saline and the serum in circle 2 by drawing the mixture up and down in the safety pipette eight times.
- Transfer 50 µl from circle 2 (1:2) to circle 3, and mix.
- Transfer 50 µl from circle 3 to circle 4, mix, and then discard the last 50 µl.
- Gently re-suspend the antigen suspension.
- Add exactly 1 freefalling drop (17 µl) of antigen suspension to each circle.
- Place the slide on the mechanical rotator. Rotate the slide for 4 minutes at 180 ±2 rpm.
- Immediately after rotation, read the test.
- If the highest dilution tested (1:8) is reactive, continue as follows:
- Prepare a 1:8 dilution of the test specimen in a test tube. Add 0.1 ml of serum to 0.7 ml of 0.9% saline. Mix thoroughly.
- Place 50 µl of 0.9% saline into paraffin rings 2, 3, and 4. Prepare additional serial dilutions for strongly reactive specimens.
- Add 50 µl of the 1:8 dilution of the test specimen to paraffin rings 1 and 2.
- Prepare serial twofold dilutions beginning with ring 2. Complete the test as described above.
Result Interpretation of VDRL Test
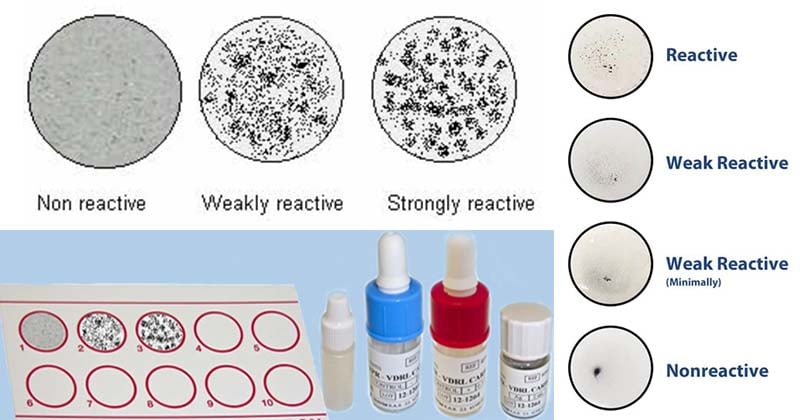
- Positive test: Presence of antigen-antibody clumps in the center or the periphery of the test circle indicate positive VDRL test.
- Negative test: Absence of antigen-antibody clumps. Indicated by a smooth, even light gray appearance with no aggregates.
- All reactive and weakly reactive serum requires serial dilution to estimate antibody titer. The titre is reported as the reciprocal of the highest dilution, which shows a positive test result.
Applications of VDRL Test
- It is used mostly as the screening test for syphilitic infection.
- Screening for syphilis is a routine part of pregnancy tests.
- VDRL test for syphilis is also performed if being treated for another STI such as gonorrhea, infected with HIV, or if engaged in high-risk sexual activity.
- If already treated for syphilis, VDRL is used for follow-up testing on recommendations of the Center for Disease Control and Prevention (CDC).
Advantages of VDRL Test
- VDRL test is most widely used simple and rapid test for syphilis.
- The VDRL tests are fast, easy to perform, and excellent for screening of samples.
- VDRL test is also helpful in the diagnosis of congenital syphilis.
- Since, the organism Treponema pallidum cannot be cultured in artificial media, the screening of syphilis via serological testing such as VDRL becomes important.
- Patient need not have the symptoms of syphilis for this test to be accurate. It can detect syphilis in patients without symptoms.
- Since it is a non-treponemal test (non-specific test) it is used to investigate syphilis along with other treponematose, Yaws and Pinta.
Limitations of VDRL Test
- Without some other evidence for the diagnosis of syphilis, a reactive non-treponemal test such as VDRL does not confirm pallidum infection. Any reactive VDRL test must be confirmed with a specific or treponemal test such as TPHA, FTA-ABS test.
- The anti lipoidal antibodies detected are not only produced as a consequence of syphilis and other treponemal diseases, but also may be produced in response to nontreponemal diseases of an acute and chronic nature in which tissue damage occurs.
- The VDRL test isn’t always accurate. For example, false-negative results may arise if an individual had syphilis for less than three months, as it could take long for the body to make antibodies. The test is also unreliable in late-stage syphilis.
- On the other hand, false-positive results can be seen in conditions like HIV, Lyme disease, malaria, pneumonia, systemic lupus erythematosus, IV drug use and tuberculosis.
- The antibodies produced as a result of a syphilis infection can stay in the body even after your syphilis has been treated.
- A prozone reaction may occur. In a prozone reaction, reactivity with undiluted serum is inhibited. The prozone phenomenon may be suspected when a specimen produces only a weakly reactive or a rough nonreactive result in a qualitative test.
- The VDRL may be reactive in persons from areas where yaws is endemic. As a rule, residual titers from these infections will be <1:8.
References
- Harris A, Rosenberg AA, Riedel LM. A microflocculation test for syphilis using cardiolopin antigen: preliminary report. J Vener Dis Inform 1946; 27: 159–172.
- https://www.cdc.gov/std/syphilis/manual-1998/chapt8.pdf
- https://laboratoryinfo.com/vdrl/
- http://www.onlinebiologynotes.com/vdrl-test-principle-procedure-result-interpretation-application/
- https://www.microrao.com/micronotes/vdrl.pdf
- https://www.healthline.com/health/vdrl-test#outlook

you’ve closed the knowledge gap I had. Thank you so much, may the Almighty Father reward you abundantly!!!
Prozone phenomenon
Q. A more serious limitations of VDRL or RPR test in late syphilis is
A. False positive
B. Prozone phenomenon
C. Biological false positive reaction
D. All
Sir, Please answer