What is Streptococcus anginosus?
Streptococcus anginosus is a Gram-positive bacterium that is present in the mouth, upper respiratory tract, gastrointestinal tract, and vagina as a normal inhabitant.
- S. anginosus belongs to the anginosus subgroup of viridans streptococci which was traditionally called the ‘milleri group’. However, the name is now pseudotaxonomic as the idea that all the members in the group belonged to the Streptococcus milleri species was discarded.
- The ability to cause abscesses is unique and sets the anginosus group apart from the rest of the streptococci groups
- It is a commensal that is present on the human body as a part of the normal flora that serves to protect the host from various pathogenic organisms.
- However, cases of S. anginosus acting as an opportunistic pathogen where it causes various infections of the oral and non-oral sites have been described.
- One of the important demonstrations of the pathogenic nature of this organism is its association with colorectal carcinoma or abscesses, which is rarely implicated with malignancy.
- S. anginosus was first isolated and identified by Andrewes and Horder in 1906 from the human gut.
- The species name ‘anginosus‘ is taken from the term ‘angina’, indicating the association between the species and the disease angina, causing chest pain due to lack of oxygen to the muscles.
Classification of Streptococcus anginosus
- The species of the genus Streptococcus have been classified on the basis of their DNA-DNA hybridization and rRNA sequences into about 60 different species and 12 subspecies.
- The genus belongs to the family Streptococcaceae which is a part of the low (<50%) G+C content branch of Eubacteria based on their rRNA sequences.
- Streptococcus species are closely related to the group of lactic acid bacteria as they produce lactic acid as their primary end product during carbon metabolism.
- These belong to the anginosus group of Streptococci, which in itself has been the subject of much taxonomic confusion, partly due to lack of international consensus on terminology and partly due to lack of reliable distinguishing phenotypic markers.
- Although the term ‘Streptococcus milleri group’ is still widely used, the term ‘Anginosus group’ proposed by Kawamura is preferred as it may eliminate much of the nomenclatural confusion.
- The group consists of two other species, namely; S. intermedius and S. constellatus, which were previously considered the subspecies of S. anginosus but were later realized as separate species based on their molecular characteristics.
- Some strains of S. anginosus belong to group F of the Lancefield antigen group while the rest are non-groupable.
The following is the taxonomical classification of S. anginosus:
| Domain: | Bacteria |
| Phylum: | Firmicutes |
| Class: | Bacilli |
| Order: | Bacillales |
| Family: | Streptoococcaceae |
| Genus: | Streptococcus |
| Species: | S. anginosus |
Habitat of Streptococcus anginosus
- Streptococcus anginosus is part of the bacterial normal flora in humans where it colonizes a wide variety of parts within the human body.
- The primary site of colonization of the organism is the oral cavity, gastrointestinal tract, and vaginal tract.
- Besides, they have also been isolated from other areas of the body like the hepatic sites, upper respiratory tract, and urinary tract.
- S. anginosus is harmless as a commensal but can cause various infections if it makes its way into the sterile sites of the body.
- The natural habitat or source of the bacteria is not yet known; however, it is assumed that it is transmitted to humans soon after birth via the environment and the primary care-giver.
- The organism has a different mechanism to survive in such parts of the human body and reduce its competition.
- The average temperature for the growth and survival of the organism is the average human body temperature, but it can survive in the temperature range of 30-40°C.
Morphology of Streptococcus anginosus
- The cells of S. anginosus are Gram-positive, round, spherical, or cocci that are small with about 0.5–1.0 µm average diameter.
- The cells are arranged in short chains with longer chains seen when grown on liquid culture media. The arrangement of the cells is characteristic of the genus Streptococci with successive division planes that are parallel resulting in cells arranged in the form of chains.
- The organism is catalase-negative, facultative anaerobe, not capsulated, and usually carries sparsely distributed, long fibrils on the surface.
- The cell wall is made up of peptidoglycan, teichoic acid, and different types of carbohydrates. The peptidoglycan type found in S. anginosus is Lys-Ala1-3.
- The teichoic acid present in the cell wall is ribitol teichoic acid and Rhamnose, Glucose and Galactose are the major cell-wall carbohydrate constituents.
- The peptidoglycan in the cell wall is a long chain of glycans that are cross-linked through short peptides, and the individual glycan is made of β-1,4-linked units alternating with N-acetylglucosamine and N-acetylmuramic acid.
- An outer capsular polysaccharide is also present on S. anginosus that is involved in the pathogenesis of the infections caused by the organism.
- A lipid-protein bilayer is present underneath the cell wall that functions as a cell membrane. It contains various transport chains that help in the movement of molecules between the cell and the environment.
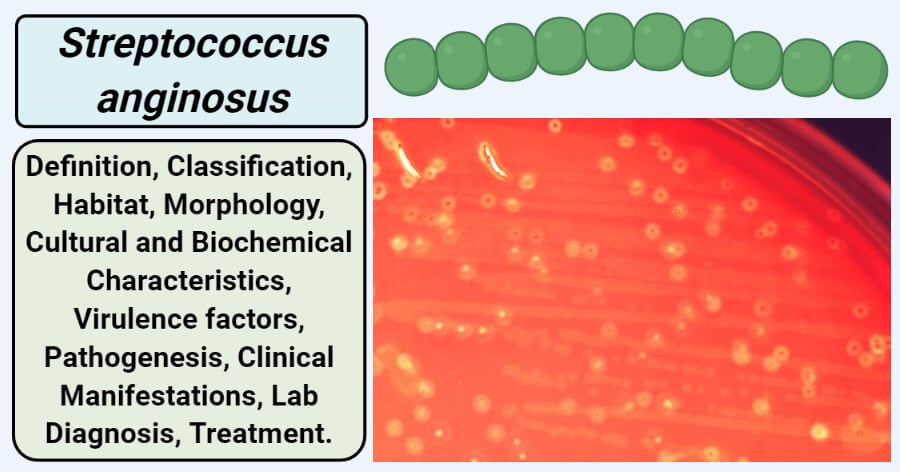
Figure: Streptococcus anginosus on trypticase soy agar medium containing 5% defibrinated sheep’s blood. Source: Public Health Image Library (PHIL), CDC.
Cultural characteristics of Streptococcus anginosus
- Streptococcus anginosus does not show profuse growth on traditional media like Nutrient Agar and might require supplements like carbohydrates and other nutrients.
- Media like Blood agar and Chocolate agar are commonly used for the observation of the pattern of hemolysis that is important during the identification of S. anginosus.
- For more selective isolation, media like Brain Heart Infusion Agar and Trypticase soy agar/broth with defibrinated sheep blood can be used.
- It is a facultative anaerobe, so abundant growth is observed in the air with 5% carbon dioxide at 37°C.
- The growth is reduced under aerobic conditions, is frequently enhanced by the addition of CO2 with some strains requiring anaerobic incubation.
- No growth is seen at 10°C, but growth is positive at 45°C. The organism can tolerate 40% bile but cannot tolerate 6.5% NaCl and pH 9.6.
1. Nutrient Agar
- White to grey colored colonies of an average size of 1 mm in diameter. The colonies were round with raised elevation and an entire margin.
- Growth is mostly poor and requires air with supplied carbon dioxide.
2. Blood Agar
- Colonies on blood (horse) agar are 0.5–2.0 mm in diameter, white or translucent, convex, and entire. Some strains might produce colonies that are white, and matte with an average diameter of 0.5–1.0 mm.
- Most strains give α-hemolysis or no hemolysis on blood agar with some strains giving β-hemolysis.
- Pronounced greening is observed on chocolate agar.
Biochemical characteristics of Streptococcus anginosus
The biochemical characteristics of S. anginosus can be tabulated as follows:
| S.N | Biochemical Characteristics | S. anginosus |
| 1. | Capsule | Some strains produce a polysaccharide capsule. |
| 2. | Shape | Cocci |
| 3. | Catalase | Negative (-) |
| 4. | Oxidase | Negative (-) |
| 5. | Citrate | Negative (-) |
| 6. | Methyl Red (MR) | Negative (-) |
| 7. | Voges Proskauer (VR) | Positive (+) |
| 8. | OF (Oxidative-Fermentative) | Facultative anaerobes |
| 9. | Coagulase | Negative (-) |
| 10. | DNase | Negative (-) |
| 11. | Clumping factor | Negative (-) |
| 12. | Gas | Positive (+) |
| 11. | H2O2 | Produced by some strains. |
| 12. | Hemolysis | α-hemolytic |
| 13. | Motility | Non-motile |
| 14. | Nitrate Reduction | Negative (-) |
| 15. | Gelatin Hydrolysis | Not determined |
| 16. | Pigment Production | Variable |
| 17. | Bile esculin test | Positive (+) |
| 18. | Ig A1 protease | Negative (-) |
| 19. | Urease | Negative (-) |
| 19. | Lancefield group | The majority of strains are either serologically ungroupable or belong to Lancefield group F, although strains might possess the Lancefield group A, C, or G antigen. |
Fermentation
| S.N | Substrate | S. anginosus |
| 1. | Glucose | Positive (+) |
| 2. | Fructose | Positive (+) |
| 3. | Galactose | Positive (+) |
| 4. | Lactose | Positive (+) |
| 5. | Maltose | Positive (+) |
| 6. | Mannitol | Positive (+) |
| 7. | Mannose | Positive (+) |
| 8. | Raffinose | Positive (+) |
| 9. | Ribose | Positive (+) |
| 10. | Sucrose | Positive (+) |
| 11. | Starch | Negative (-) |
| 12. | Trehalose | Positive (+) |
| 13. | Xylose | Negative (-) |
| 14. | Salicin | Positive (+) |
| 15. | Glycerol | Negative (-) |
| 16. | Dulcitol | Not determined |
| 17. | Cellobiose | Variable |
| 18. | Rhamnose | Negative (-) |
| 19. | Arabinose | Negative (-) |
| 20. | Inulin | Negative (-) |
| 21. | Sorbitol | Negative (-) |
| 22. | Pyruvate | Negative (-) |
| 23. | Glycogen | Negative (-) |
Enzymatic Reactions
| S.N | Enzymes | S. anginosus |
| 1. | Acetoin | Positive (+) |
| 2. | Acid Phosphatase | Not determined |
| 3. | Alkaline Phosphatase | Positive (+) |
| 4. | Ornithine Decarboxylase | Not determined |
| 5. | Hyaluronidase | Positive (+) for a few strains. |
| 6. | β-D-glucosidase | Positive (+) |
| 7. | Leucine aminopeptidase | Positive (+) |
| 8. | Neuraminidase | Negative (-) |
Virulence factors of Streptococcus anginosus
Even though S. anginosus is present as a commensal in the human body, an increasing number of reports have been published emphasizing the role of S. anginosus as important bacterial pathogens. Traditionally, S. anginosus was overlooked as a pathogen as it was mistaken as commensal contamination, especially in diagnostic samples of polymicrobial infections. Different structures and proteins produced by the organism help in the colonization and invasion of the host body surface, which aids in the pathogenesis of the disease. Some of the well-studied virulence factors involved in disease production by S. anginosus are:
1. Adhesins
- Attachment to the host cells is a prerequisite of bacterial colonization and plays a vital role during infection.
- The binding of bacteria to the extracellular matrix proteins is a common mechanism of streptococci that has also been observed in S. anginosus.
- A potent fibronectin receptor of about 14 kDa was isolated from one of the S. anginosus strains. It seems to be localized on the cell surface at the interface between adjacent daughter cells.
- This receptor plays an essential role in the adhesion of bacterial strains to the heart valves in infective endocarditis infections.
- Besides, adherence to porcine valve tissue is mediated by laminin that occurs at areas where the basement membrane is exposed.
- The laminin-binding protein has also been demonstrated on the cell surface of S. anginosus strains.
- Some of the amino acid sequences of S. anginosus also display high similarities to several peptide‐binding bacterial proteins of Streptococcus gordonii and Streptococcus pneumonia, which might further aid in the process of colonization.
- In addition to these proteins, homologs of several other known streptococcal adhesins are also detected in S. anginosus.
- These include the pneumococcal surface Adhesin A (PsaA) of S. pneumoniae and the pullulanase PulA of S. pyogenes, both of which have high affinity to the human plasmin.
- A similar plasminogen‐binding effect is exhibited by the enolase enzyme which in S. anginosus is encoded by the enolase gene ‘eno’.
2. Bacterial capsule
- The gene locus required for capsular polysaccharide synthesis has recently been identified in S. anginosus, which is significantly similar to that in S. pneumonia.
- Inhibition of phagocytosis and phagocytic killing through capsule production is observed in S. anginosus, but studies regarding its role as a potent virulence factor are yet to be done.
3. H2S production
- Another factor produced by S. anginosus that may interact with hemoglobin is hydrogen sulfide (H2S), which is highly toxic.
- Exposure of erythrocytes to H2S leads to the production of sulfhaemoglobin, methemoglobin, and erythrocyte lysis.
- The l‐cysteine desulfhydrase of S. anginosus generates H2S from l‐cysteine, resulting in an increased abscess formation.
4. Hydrolytic enzymes
- Hydrolytic enzymes destroying nucleic acids or components of the extracellular matrix-like hyaluronic acid and chondroitin sulfate are also some of the important virulence factors in S. anginosus. Nucleases are important in immune evasion of neutrophil extracellular traps, while hyaluronidase and chondroitin sulphatase represent spreading factors through the digestion of extracellular matrix.
Pathogenesis of Streptococcus anginosus
Traditionally, S. anginosus has been considered non-pathogenic, but new studies have indicated that S. anginosus is involved in various infections throughout various parts of the body. The exact mechanism of infection is not yet clearly understood but the involvement of some structures and proteins that are homologous to that found in other pathogenic Streptococcal species.
1. Colonization
- Streptococcus anginosus is a commensal that reaches different parts of the body via different sources after birth.
- The organism primarily colonizes the gastrointestinal tract where it adheres to the epithelial cells of the mucosal membrane.
- Adhesion is the first step in the pathogenesis of the infection, which is brought about by different structures and proteins produced by the bacteria.
- The binding of bacteria to the extracellular matrix proteins is a common mechanism of streptococci that has also been observed in S. anginosus.
- S. anginosus codes for both fibrinogen-binding and laminin-binding proteins that bind to the extracellular matrix of the host cells.
- The attachment is further enhanced by surface adhesins that are homologous to the pneumococcal surface Adhesin A (PsaA) of S. pneumoniae and the pullulanase PulA of S. pyogenes.
- Besides, the enzyme enolase produced by the bacteria helps in the colonization of the epithelial surface.
2. Invasion
- Colonization of the epithelial surface is then followed by invasion, which is supported by various enzymes that help in disrupting the outer barrier.
- S. anginosus is surrounded by capsular polysaccharide that inhibits phagocytosis and phagocytic killing and allows the bacteria to divide.
- The l‐cysteine desulfhydrase of S. anginosus generates H2S from l‐cysteine, resulting in an increased abscesses formation by an interaction with hemoglobin.
- Exposure of erythrocytes to H2S leads to the production of sulfhaemoglobin, methemoglobin, and erythrocyte lysis.
- S. anginosus also produces hydrolytic enzymes that destroy nucleic acids or components of the extracellular matrix-like hyaluronic acid and chondroitin sulfate and aids in the destruction of the mucosal layer.
- The hydrolytic enzyme nucleases play an important role in immune evasion of neutrophil extracellular traps, while hyaluronidase and chondroitin sulphatase act as spreading factors by the digestion of extracellular matrix.
- The bacteria then move through the bloodstream and reach different parts of the body like the heart valves.
- In some cases, S. anginosus induces inflammation reaction and causes the release of IL-8 which, in turn, causes the release of carcinogens that might initiate malignancy.
Clinical manifestations of Streptococcus anginosus
- Streptococcus anginosus is associated with different forms of diseases with infective endocarditis being the most prominent one.
- Besides, the colonization of the gastrointestinal tract results in the formation of abscesses through the tract. The abscesses formation is characteristic of the members of the anginosus group.
- Most of the patients usually have an underlying condition that weakens the immune response and results in severe infections.
- Most patients had fevers lasting for 1 to 33 days, including those with septicemia. In severe cases, ardent fever, chills, and systemic toxemia might be observed.
- Infections of the oropharynx were associated with odynophagia, cervicodynia, and trachelophyma, leading to difficulties in swallowing and opening the mouth.
- Patients with underlying conditions such as pneumonia and pulmonary abscess are likely to have respiratory symptoms such as coughing and shortness of breath.
- In the cases of intra-abdominal abscess and peritonitis induced ardent fever, abdominal pain, vomiting, and signs of peritoneal irritation might be observed.
- Some patients might even experience painful swelling at the anus, while some patients might have abscess diabrosis and watery discharge.
- If left untreated, bacteria might reach the heart valves, which results in endocarditis as the bacteria forms biofilms around the valves.
Lab diagnosis of Streptococcus anginosus
The diagnosis of infections caused by S. anginosus begins with the collection of samples, which depends on the site of infection and its extent. The sample can either be observed directly under the microscope or in other cases, cultured to obtain viable colonies of the organism for further diagnosis. The diagnosis of diseases caused by S. anginosus is mostly involved with the identification of the organism.
1. Morphological, cultural, and biochemical characteristics
- The observation of the organism under the microscope, followed by its culture and biochemical tests is the most common method during the diagnosis of S. anginosus.
- Isolation of the organism from primary clinical specimens is obtained on selective culture media like blood agar supplemented with 5 percent sheep blood, following an incubation period of 18–24 h in the air at 35–37°C.
- Some strains might require anaerobic incubation while others grow well in air supplemented with about 5% carbon dioxide.
- Colony morphology and hemolysis can then provide a basis for biochemical testing for species-level identification.
- Different biochemical tests beginning with catalase and oxidase can provide a better distinction of the species.
2. Rapid diagnosis systems
- Nowadays, different techniques utilize different properties of the species like the fatty acid profile and surface antigens for rapid identification of the organism.
- For Gram-positive organisms, different test kits like MicroScan Conventional Pos ID, Rapid Pos ID, and BBL Crystal Gram-Pos ID are available.
- These tests, however, might not very reliable and thus should only be used as a primary identification method followed by identification through culture or molecular methods.
3. Molecular methods
- Molecular methods of diagnosis are based on the identification of the organism based on its nuclear sequences.
- This is the most accurate method of identification as the gene sequences of an organism are mostly unique.
- In molecular methods, techniques that study the DNA sequences and rRNA sequences are used, some of which are PCR and ribotyping.
- These methods use restriction fragment polymorphism technique and DNA-DNA hybridization.
Treatment of Streptococcus anginosus
- Treatment of S. anginosus might be tricky as some strains are notorious for being resistant against antimicrobials like bacitracin and nitrofurazone.
- However, other strains have shown to be susceptible to penicillin, ampicillin, erythromycin, and tetracycline.
- In some severe cases, invasive treatments like surgery, puncture, drainage might be required.
- Patients with underlying conditions can be provided with nutritional support and treatment of the underlying condition.
- The rate of mortality by S. anginosus infections is relatively low in some cases are known to support the infections caused by other pathogenic streptococci.
References
- Topley WWC (2007). Topley and Wison’s Microbiology and Microbial Interactions; Bacteriology, 2 Vol. Tenth Edition. John Wiley and Sons Ltd.
- Bergey, D. H., Whitman, W. B., De, V. P., Garrity, G. M., & Jones, D. (2009). Bergey’s manual of systematic bacteriology: Vol. 3. New York: Springer.
- Jiang, S., Li, M., Fu, T. et al. Clinical Characteristics of Infections Caused by Streptococcus Anginosus Group. Sci Rep 10, 9032 (2020). https://doi.org/10.1038/s41598-020-65977-z
- Asam, D. and Spellerberg, B. (2014), Molecular pathogenicity of Streptococcus anginosus. Mol oral Microbiol, 29: 145-155. doi:10.1111/omi.12056
- Tao, Z., Zhang, L., Zhang, Q., Lv, T., Chen, R., Wang, L., Huang, Z., Hu, L., & Liao, Q. (2019). The Pathogenesis Of Streptococcus anginosus In Aerobic Vaginitis. Infection and drug resistance, 12, 3745–3754. https://doi.org/10.2147/IDR.S227883
- Noguchi, S., Yatera, K., Kawanami, T., Yamasaki, K., Naito, K., Akata, K., … Mukae, H. (2015). The clinical features of respiratory infections caused by the Streptococcus anginosus group. BMC Pulmonary Medicine, 15(1). doi:10.1186/s12890-015-0128-6
- Tabitha M. Gana, Olugbenga Awolaran, Sobia Akhtar, “Streptococcus milleri and Recurrent Intra-Abdominal Abscesses: A Case Report and Literature Review”, Case Reports in Surgery, vol. 2016, Article ID 6297953, 3 pages, 2016. https://doi.org/10.1155/2016/6297953
- Mamie Hui. Streptococcus anginosus Bacteremia: Sutton’s Law Journal of Clinical Microbiology Dec 2005, 43 (12) 6217; DOI: 10.1128/JCM.43.12.6217.2005
- Masood, U., Sharma, A., Lowe, D., Khan, R., & Manocha, D. (2016). Colorectal Cancer Associated with Streptococcus anginosus Bacteremia and Liver Abscesses. Case reports in gastroenterology, 10(3), 769–774. https://doi.org/10.1159/000452757
- Jensen et al. Taxonomy of the Anginosus group of the genus Streptococcus and description of Streptococcus anginosus subsp. whileyi subsp. nov. and Streptococcus constellatus subsp. viborgensis subsp. nov. International Journal of Systematic and Evolutionary Microbiology (2013), 63, 2506–2519.
Sources
- 3% – https://microbenotes.com/streptococcus-bovis/
- 1% – https://www.researchgate.net/publication/11817834_Identification_of_the_psaA_Gene_Coding_for_Pneumococcal_Surface_Adhesin_A_in_Viridans_Group_Streptococci_other_than_Streptococcus_pneumoniae
- 1% – https://microbenotes.com/staphylococcus-hominis/
- 1% – https://microbenotes.com/staphylococcus-capitis/
- <1% – https://www.webmd.com/cancer/common-cancers-that-metastasize-to-the-bones
- <1% – https://www.ukessays.com/essays/biology/temperature-on-the-growth-and-survival-of-bacteria-biology-essay.php
- <1% – https://www.sweet-cures.com/research/health-and-wellbeing/streptococcus.htm
- <1% – https://www.sciencedirect.com/topics/medicine-and-dentistry/streptococcus-anginosus
- <1% – https://www.sciencedirect.com/science/article/pii/S1931312814001802
- <1% – https://www.sciencedirect.com/science/article/pii/S1389172311004117
- <1% – https://www.sciencedirect.com/science/article/pii/S0378113500001802
- <1% – https://www.sciencedirect.com/science/article/pii/S0196439916300101
- <1% – https://www.sciencedirect.com/science/article/pii/S0161589016302644
- <1% – https://www.sciencedirect.com/science/article/pii/0021968178900917
- <1% – https://www.researchgate.net/publication/8546731_The_role_of_the_capsule_of_the_Streptococcus_milleri_group_in_its_pathogenicity
- <1% – https://www.researchgate.net/publication/237737875_lcd_from_Streptococcus_anginosus_encodes_a_CS_lyase_with_ab-elimination_activity_that_degrades_L-cysteine
- <1% – https://www.ncbi.nlm.nih.gov/pubmed/28840807
- <1% – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4683221/
- <1% – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3264080/
- <1% – https://www.medicowesome.com/2012/12/difference-between-blood-agar-and.html
- <1% – https://www.lion.com/lion-news/july-2019/what-is-hydrogen-sulfide
- <1% – https://www.healthline.com/health/elderly-pneumonia
- <1% – https://www.britannica.com/science/human-body
- <1% – https://quizlet.com/44097356/micro-final-qbank-cy-flash-cards/
- <1% – https://quizlet.com/176386574/cell-surface-interface-flash-cards/
- <1% – https://pubmed.ncbi.nlm.nih.gov/29488863/
- <1% – https://onlinelibrary.wiley.com/doi/full/10.1111/omi.12278
- <1% – https://onlinelibrary.wiley.com/doi/abs/10.1002/art.41078
- <1% – https://medical-dictionary.thefreedictionary.com/hydrolytic+enzyme
- <1% – https://lookformedical.com/en/search/streptococcus-anginosus
- <1% – https://en.wikipedia.org/wiki/Streptococcus_anginosus_group
- <1% – https://en.wikipedia.org/wiki/Streptococcus_anginosus
- <1% – https://en.wikipedia.org/wiki/Streptococcus
- <1% – https://diabetes.diabetesjournals.org/content/21/Supplement_2/461
- <1% – https://bcdairy.ca/milk/articles/the-probiotic-effects-of-lactic-acid-bacteria

Strep. anginosus is not oxidase positive. It is oxidase negative.
Hi Sam,
Thanks for the correction. It has been updated to Oxidase Negative (-ve).