Radioimmunoassay is one of the sensitive immunoassay techniques which helps in the determination of antigens or antibodies in a sample with the use of radioisotopes.
It is an in vitro type of antigen-antibody interaction.
When radioisotopes instead of enzymes are used as labels to be conjugated with antigens or antibodies, the technique of detection of the antigen-antibody complex is called radioimmunoassay (RIA). Radioimmunoassay (RIA) is an in vitro assay that measures the presence of an antigen with very high sensitivity. RIA was first described in 1960 for the measurement of endogenous plasma insulin by Solomon Berson and Rosalyn Yalow of the Veterans Administration Hospital in New York.
The classical RIA methods are based on the principle of competitive binding. In this method, an unlabeled antigen competes with a radiolabeled antigen for binding to an antibody with the appropriate specificity. Thus, when mixtures of radiolabeled and unlabeled antigen are incubated with the corresponding antibody, the amount of free (not bound to antibody) radiolabeled antigen is directly proportional to the quantity of unlabeled antigen in the mixture.
Radioimmunoassay (RIA) Requirements
Radiolabeled antigens: The antigens are generally labeled with gamma-ray emitting isotopes such as I-125 and beta-ray emitting isotopes such as Tritium. They are also called hot antigens.
Specific Antibodies: They are required in smaller amounts than antigens.
Unlabeled antigens (sample antigens): They are also called cold antigens.
Microtitre plates: 96 wells microtitre plate
Washing Buffer solutions: Wash buffer such as 1% Trifluoroacetic acid is used.
Radioimmunoassay (RIA) Principle
Antigens and antibodies bind specifically to form the Ag-Ab complex. The antigen can be labeled or conjugated with radioisotopes. The unlabeled antigens from the sample compete with radiolabeled antigens to bind on paratopes of specific antibodies. The unlabeled antigens replace labeled antigens that are already linked with the antibodies. The unlabeled antigens when bind with antibodies, increases the amount of free radiolabeled antigens in the solution. Hence the concentration of free labeled antigens is directly proportional to the bound unlabeled antigens.
It involves a combination of three principles.
- An immune reaction i.e. antigen, antibody binding.
- A competitive binding or competitive displacement reaction. (It gives specificity)
- Measurement of radio emission. (It gives sensitivity)
Immune Reaction
When a foreign biological substance enters the body’s bloodstream through a non-oral route, the body recognizes the specific chemistry on the surface of the foreign substance as antigen and produces specific antibodies against the antigen so as nullify the effects and keep the body safe. The antibodies are produced by the body’s immune system so, it is an immune reaction. Here the antibodies or antigens bind and move due to chemical influence. This is different from the principle of electrophoresis where proteins are separated due to charge.
Competitive binding or competitive displacement reaction
This is a phenomenon wherein when there are two antigens that can bind to the same antibody, the antigen with more concentration binds extensively with the limited antibody displacing others. So here in the experiment, a radiolabelled antigen is allowed to bind to a high-affinity antibody. Then when the patient serum is added to unlabeled antigens it starts binding to the antibody displacing the labeled antigen.
Measurement of radio emission
Once the incubation is over, then washings are done to remove any unbound antigens. Then radio emission of the antigen-antibody complex is taken, and the gamma rays from the radiolabeled antigen are measured.
The target antigen is labeled radioactively and bound to its specific antibodies (a limited and known amount of the specific antibody has to be added). A sample, for e.g. blood serum, is added in order to initiate a competitive reaction of the labeled antigens from the preparation, and the unlabeled antigens from the serum sample, with the specific antibodies. The competition for the antibodies will release a certain amount of labeled antigen. This amount is proportional to the ratio of labeled to an unlabeled antigen. A binding curve can then be generated which allows the amount of antigen in the patient’s serum to be derived. That means as the concentration of unlabeled antigen is increased, more of it binds to the antibody, displacing the labeled variant. The bound antigens are then separated from the unbound ones, and the radioactivity of the free antigens remaining in the supernatant is measured.
Antigen-antibody complexes are precipitated either by crosslinking with a second antibody or by means of the addition of reagents that promote the precipitation of antigen-antibody complexes. Counting radioactivity in the precipitates allows the determination of the amount of radiolabeled antigen precipitated with the antibody. A standard curve is constructed by plotting the percentage of antibody-bound radiolabeled antigen against known concentrations of a standardized unlabeled antigen, and the concentrations of antigen in patient samples are extrapolated from that curve.
The extremely high sensitivity of RIA is its major advantage.
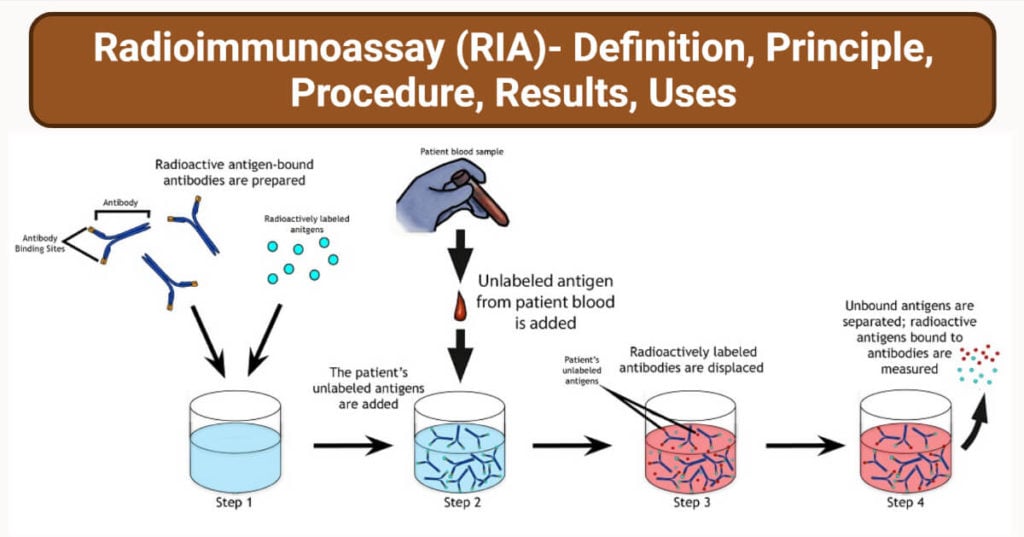
Radioimmunoassay (RIA) Procedure
- Specific antibodies of known concentration are fixed in the microtitre well.
- A known amount of hot antigens is then added to the well
- Washed carefully to remove any unbound antigens
- At this point, the radioactivity of the well will be maximum.
- Unlabeled antigens are then added to the well
- The unlabeled antigens will bind to the antibodies and there will be free labeled antigens in the well.
- Again washed carefully to remove the free labeled antigens.
- Radioactivity of wells is then measured by gamma-counter.
Radioimmunoassay (RIA) Result Interpretation
At first, the labeled antigens will bind to the antibodies hence radioactivity will be maximum.
If the sample contains specific antigens of interest, it will bind to the antibodies releasing labeled antigens and hence the radioactivity of the solution will decrease.
So by observation of decreasing radioactivity, it can be confirmed that the antigen of interest is present in the sample. And if the radioactivity remains the same, it can be called a negative test.
With the increasing concentration of unlabeled antigens, the radioactivity decreases. By plotting a graph of radioactivity(in percentage) vs concentration of unlabeled antigens, a standard curve is obtained.
The sample to be assayed is run parallel following a similar procedure and the radioactivity measured is calibrated with the standard curve to determine the concentration of the antigen.
Radioimmunoassay (RIA) Applications
- It was first used for the detection of peptide hormones.
- Detection of different viral antigens
- Detection of many hormones and drugs
- Detection of Hepatitis B surface antigens
- Detection of mycotoxins
- Detection of the early stage of cancer
Radioimmunoassay (RIA) Advantages
- High specificity
- High sensitivity
- Can detect a very small amount (nanograms) of antigen or antibodies.
Radioimmunoassay (RIA) Limitations
- Working with radioactive substances makes it a bit risky.
- Disposal of radioactive substances can be problematic.
- Equipment and reagents are expensive.
- Radiolabeled substances used have a short shelf-life.
References
- Goldsmith SJ. Radioimmunoassay: review of basic principles. Semin Nucl Med. 1975 Apr;5(2):125-52. DOI: 10.1016/s0001-2998(75)80028-6. PMID: 164695.
- Goldsby R.A., Kindt T.J., Osborne B.A., (1999) Kuby Immunology, 4th edition, W.H.Freeman & Co Ltd., pg. 147-148.
- Parija S.C., (2009), Textbook of Microbiology and Immunology, 2nd edition, Elsevier, a division of Reed Elsevier India Private Limited, pg. 113-114.
- Howell-Moroney, Madeleine, “Radioimmunoassay”. Embryo Project Encyclopedia (2022-01-24). ISSN: 1940-5030 http://embryo.asu.edu/handle/10776/13324.

Thank you sir 👍😊 It’s just awesome.
Great explanation, thank you for sharing.
this is so helpful for my studies
good presentation. the explanation is very nice.
Well explained just some diagrams would be much helpful. Thankyou
Thank you so much for sharing this information in easy language
Good
Please tell me about references