Antigen-Antibody (Ag-Ab) Interaction is a biochemical reaction between antibodies and specific antigens when they come closer to a distance of several nanometers. It is the binding of paratopes of antibodies to specific antigens on their epitopes that initiates a series of immunological responses to act against the respective antigens for their removal or destruction.
Antigen + Antibody ⇄ Ag-Ab complex → Immune Response
According to the law of mass action:
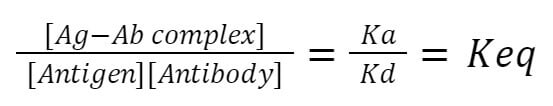
Here,
Ka = Rate of the association constant
Kd = Rate of the dissociation constant
Keq = Equivalence constant
At equilibrium, the ratio of the concentration of product i.e. [Ag-Ab complex], and reactants i.e. [Antigen]-[Antibody] are constant and called equivalence constant.
What is an Antigen (Ag)?
(Anti= against; gen=thing that produces or causes)
Any foreign substances that when entering our body sometimes self-elicit a series of immune responses and are precisely called immunogens. Whereas some of them don’t directly elicit an immune response but require the help of some other molecules (carrier proteins) to do so and are called hapten. The immunogens and hapten are collectively called antigens.
– They can be proteins, peptides, lipids, or, polysaccharides.
– Antibody binding site is called an epitope.
– Abbreviation: “Ag”
What is an Antibody (Ab)?
An antibody is simply the component produced by the immune system in response to antigens. So basically antigens are the generator of antibodies. They interact with each other to induce an immune response.
– Also called immunoglobulins (Ig)
– Y-shaped
– Glycoproteins
– Produced by plasma B-cells
– Antigen binding site is called paratope.
– Types: IgG, IgA, IgM, IgE, IgD (Pneumonics: “GAMED”)
What is an Antigen-Antibody Interaction?
The interactions between antigens and antibodies are known as antigen-antibody reactions. The reactions are highly specific, and an antigen reacts only with antibodies produced by itself or with closely related antigens. Antibodies recognize molecular shapes (epitopes) on antigens. Generally, the better the fit of the epitope (in terms of geometry and chemical character) to the antibody combining site, the more favorable the interactions that will be formed between the antibody and antigen and the higher the affinity of the antibody for antigen. The affinity of the antibody for the antigen is one of the most important factors in determining antibody efficacy in vivo.
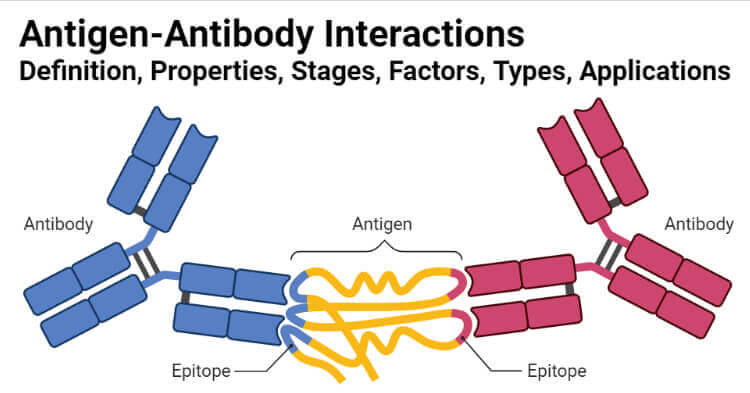
The antigen-antibody interaction is a bimolecular irreversible association between antigen and antibody. The association between antigen and antibody includes various non-covalent interactions between epitope (antigenic determinant) and variable region (VH/VL) domain of an antibody.
Antigen-Antibody Interaction Properties
– Highly specific reaction
– Occurs in an observable manner
– Non-covalent interaction ( Van der Waals forces, Ionic bonds, Hydrogen bonds, Hydrophobic interactions )
– No denaturation of antibodies and antigens
– Reversible
– Affinity: It is the strength with which one antigen binds on a single antigen-binding site on an antibody.
– Avidity: It is a broader term than affinity. It is a measure of the overall strength of the Ag-Ab complex. It depends on:
- The affinity of the antibody
- Valency(no. of binding sites) of antibody and antigen
- And the structural arrangement of epitopes and paratopes.
– Cross-Reactivity: It refers to the ability of an antibody to bind to similar epitopes of different antigens.
Stages of Antigen-Antibody Interaction
1. Primary Stage: It is the initial interaction between antigens and antibodies.
– Rapid
– Reversible
– Without any visible effects
2. Secondary Stage: It is the irreversible interaction between antigens and antibodies.
– Slow
– With visible effects
Factors Affecting Antigen-Antibody Interaction
Many factors affect Ag-Ab reactions. Some of the common factors are:
- Temperature: It depends on the chemical nature of epitopes, paratopes, and, bonds involved. Eg. hydrogen bonds are stable at low temperatures and hydrophobic bonds are stable at high temperatures.
- pH: Optimal pH range is 6.5 to 8.5. Extreme pH values change the conformation of antibodies and inhibit the reaction.
- Ionic strength: The effect of ionic is important in blood group serology. Here the reaction is significantly influenced by sodium and chloride ions. In normal saline solution, Na+ and Cl− cluster around the complex and partially neutralize charges, potentially interfering with antibody binding to antigen. This could be problematic when low-affinity antibodies are used.
- Enzyme Treatment: Many proteolytic enzymes are used to enhance the Ag-Ab reactions. Some of the commonly used ones are papain, ficin, bromelin.
- Concentrations of Ag and Ab: Increase in the concentration of antigen and antibody enhances the reaction.
- No. of antigen-binding sites: More the no. of antigen-binding sites on the antibody, the more the chances of interaction. For eg., IgM is a pentamer and hence has 10 binding sites whereas IgG is a monomer and hence has only 2 binding sites so IgM will bind more efficiently with antigens.
- Structural arrangement: If the structure of epitope and paratope is such that they could fit well as lock and key then it enhances the interaction between antigen and antibody.
Chemical Bonds Responsible for the Antigen-Antibody Reaction
The interaction between the Ab-binding site and the epitope involves exclusively non-covalent bonds, in a similar manner to that in which proteins bind to their cellular receptors, or enzymes bind to their substrates. The binding is reversible and can be prevented or dissociated by high ionic strength or extreme pH. The following intermolecular forces are involved in Ag–Ab binding:
- Electrostatic bonds: This results from the attraction between oppositely charged ionic groups of two protein side chains; for example, an ionized amino group (NH4+) on lysine in the Ab, and an ionized carboxyl group (COO_) on an aspartate residue in the Ag.
- Hydrogen bonding: When the Ag and Ab are in very close proximity, relatively weak hydrogen bonds can be formed between hydrophilic groups (e.g., OH and C=O, NH, and C=O, and NH and OH groups).
- Hydrophobic interactions: Hydrophobic groups, such as the side chains of valine, leucine, and phenylalanine, tend to associate due to Van der Waals bonding and coalesce in an aqueous environment, excluding water molecules from their surroundings. As a consequence, the distance between them decreases, enhancing the energies of attraction involved. This type of interaction is estimated to contribute up to 50% of the total strength of the Ag–Ab bond.
- Van der Waals bonds: These forces depend upon interactions between the “electron clouds” that surround the Ag and Ab molecules. The interaction has been compared to that which might exist between alternating dipoles in two molecules, alternating in such a way that, at any given moment, oppositely oriented dipoles will be present in closely apposed areas of the Ag and Ab molecules.
Each of these non-covalent interactions operates over a very short distance (generally about 1 Å) so, Ag-Ab interactions depend on a very close fit between antigen and antibody.
Strength of Ag-Ab interactions
- Affinity
- The combined strength of total non-covalent interactions between a single Ag-binding site of Ab and a single epitope is the affinity of Ab for that epitope.
- Low-affinity Ab: Bind Ag weakly and dissociates readily.
- High-affinity Ab: Bind Ag tightly and remain bound longer.
- Avidity
- The strength of multiple interactions between multivalent Ab and Ag is avidity. Avidity is a better measure of the binding capacity of antibodies than affinity. High avidity can compensate for low affinity.
- Cross-reactivity
- Antibodies elicited by one Ag can cross-react with unrelated Ag if they share identical epitopes or have similar chemical properties.
Types of Antigen-Antibody Interaction
Ag-Ab reactions are basically of two types:
1. In Vivo (Occurring in natural condition): It includes the general antibody-mediated immune response occurring in our body against any antigen.
2. In Vitro (Done in artificial conditions): It includes a series of serological tests performed in laboratories to detect antigens or antibodies in case of many diseases.
In Vivo Reactions
- Agglutination
- Precipitation
- Complement fixation
- Neutralization
- Ab Dependent Cell-Mediated Toxicity
- Immobilization
- Opsonization
In vitro Reactions
- Agglutination
- Precipitation
- Complement fixation
- Neutralization
- ELISA
- Radioimmunoassay (RIA)
- Western Blotting
- Immunochromatograhy (ICT)
- Immunofluorescence
Precipitation Reaction
The reaction between soluble (small) antigens and soluble antibodies forms an insoluble precipitate. The proportion of Ag and Ab in the reaction must be equivalent for the reaction to occur.
– In the presence of electrolytes
– At specific pH and Temperature
– Antibodies involved are called precipitins
– Types: In solution (Flocculation and Ring Test)
In Agar (Immunodiffusion)
In Agar with an electric field (Immunoelectrophoresis)
Agglutination Reaction
The reaction between insoluble (large) antigens and soluble antibodies leads to agglutination.
– Formation of visible clumps occurs.
– Occurs on the surface of the particle involved.
– Antibodies involved are called agglutinins.
– Types: Direct/Active (Slide, Tube, Heterophile agglutination test and, Coombs’ test,)
: Passive (Latex, Haemagglutination and, Coagglutination test)
Haemagglutination
It is a passive agglutination reaction that involves RBCs as carrier particles.
– RBCs of sheep, humans, chicks, etc. are used
– Used in Blood Typing
– In the detection of parasitic infections and viral diseases such as influenza, mumps, and measles.
Note: In the case of sensitivity agglutination reaction is more sensitive than precipitation reaction.
Complement Fixation
It is the process of binding or fixing the serum complement proteins with the Ag-Ab complex which further initiates a series of immune responses against the antigen. It is used in the detection of any specific antigen or antibody in the patient’s serum.
– Reagents: Antigen, Antibody, Complement, and, Indicator System
– Common complement fixation test is Wasserman’s Test: For detection of Syphilis
Enzyme-Linked Immunosorbent Assay (ELISA)
It is one of the sensitive techniques for the detection of the presence of antigen or antibody and quantification as well in case of clinical diagnosis of many diseases such as AIDS, Ebola, Pernicious anemia, different parasitic infections, etc. Enzymes are used for labeling.
– Detects free antigens or antibodies.
– Reagents: Coating buffer, Blocking buffer, Wash Buffer, Substrates, Sample and, Assay Diluents.
– Types: Direct, Indirect, and, Sandwich ELISA.
Hemolysis
When Ag-Ab interactions result in the rupture or lysis of RBC, it is called hemolysis which results in the release of Haemoglobin. It can be catalyzed by enzymes called hemolysins. It is the demonstrable endpoint of some reactions.
Immunofluorescence
It is a type of immunoassay technique in which fluorescent dyes are used for the visualization of Ag-Ab reactions.
– Detects surface antigens or antibodies.
– Fluorescent dyes such are fluorescein isothiocyanate and lissamine rhodamine used.
– Types: Direct and Indirect.
Neutralization
In neutralization, the biological effects of viruses and toxins are neutralized by homologous antibodies called neutralizing antibodies.
Types:- Virus Neutralization Tests (Eg. Viral hemagglutination inhibition test)
– Toxin Neutralization Tests (Eg. Schick test, antistreptolysin O test, etc.)
Radioimmunoassay (RIA)
It is a type of immunoassay in which radioisotopes are used for labeling the antigen or antibody to detect the formation of the Ag-Ab complex.
– Can determine very small quantities of Ag and Ab in the serum.
– Used for the quantification of hormones, drugs, and, viral antigens.
Sensitization
The first step in the Ag-Ab interactions involves the formation of the Ag-Ab complex and it is called sensitization. It is not observable and is observed only after agglutination of the formed complex using different reagents. IgG antibodies can sensitize red cells to the corresponding antigens and hence are called sensitizing antibodies.
Western Blotting
It is called so because of its similarity to Southern Blotting.
– Protein separation is done by electrophoresis.
– Used in the detection of proteins.
– Confirmatory test in the diagnosis of HIV.
Applications of Antigen-Antibody Interaction
- The most common use is the determination of blood groups i.e. blood typing.
- Rapid diagnosis test kits used for pregnancy detection as well as detection of several diseases such as malaria, dengue, etc. are based on this principle. They require very little time for the tests.
- Serological ascertainment of exposure to infectious agents.
- Quantification of drugs, hormones, viral antigens, etc.
- Detection of presence or absence of proteins in serum.
- To study the characteristics of different immunodeficiency diseases.
- To perform confirmatory tests for infections such as Western Blotting for HIV infection.
Limitations of Antigen-Antibody Interaction
- Require higher expertise and equipment which are not mostly available in some developing or underdeveloped countries. Hence these techniques are not used in those places mostly.
- RDTs do not reliably detect low-density parasitemia (≤200 parasites/µL).
- In different cases of antibody detection, it’s hard to differentiate between early and late infections as the antibodies remain in our blood for a long time even after the cure of the infection.
References and Sources
- Goldsby R.A., Kindt T.J., Osborne B.A., (1999) Kuby Immunology, 4th edition, W.H.Freeman & Co Ltd., pg. 137- 160
- Parija S.C., (2009), Textbook of Microbiology and Immunology, 2nd edition, Elsevier, a division of Reed Elsevier India Private Limited, pg. 94-115
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2581910/
- https://onlinelibrary.wiley.com/doi/full/10.1111/j.1751-2824.2008.00185.x
- https://www.ramauniversity.ac.in/online-study-material/faculty_sciences/mscbiotechnology/iiisemester/immunology/lecture-9.pdf- 2%
- http://www.microbiologybook.org/mayer/ab-ag-rx.htm- 1%
- https://www.brainkart.com/article/Neutralization-Tests—Antigen-Antibody-Reactions_17923/- 1%
- https://microbenotes.com/immunofluorescence/– 1%
- https://www.differencebetween.com/difference-between-precipitation-and-vs-agglutination-reactions/- 1%
- https://biodifferences.com/difference-between-igm-and-igg.html- <1%
- https://www.thermofisher.com/us/en/home/life-science/antibodies/immunoassays/elisa-kits/elisa-blocking-buffers-reagents.html- <1%
- http://ndvsu.org/images/StudyMaterials/Micro/Serological-Technique-II.pdf- <1%
- https://www.researchgate.net/publication/228031747_Antigen-Antibody_Binding- <1%

Very helpful, thank you so much
Thank you very much. This has been very helpful
thank you so much 😊🙏🏻notes are beautiful and simple words example thank you for this amazing notes ❤️
Thank you for the clear notes. And you interacted well with the readers. Thank you❤
excellent explanation.
Thank you so much.
excellent explanation thank you!
i have one question;
you said the antigen-antibody interaction is a bimolecular irreversible association between antigen and antibody.
Is ag-ab interaction reversible or irreversible?
Irreversible
College of health science and technology filiya gombe state shangom L. C. G filiya word
It’s helpful information so to speak!!!
Notes are good but the advts. between the notes irritates sometimes…hope you understand and modify the things.
Thank you so much for the suggestion. But ads are only the way to sustain from cost incurred like servers, domain, writers, etc. so that we can keep the contents or notes always free for the users or visitors. We will try to minimize the adverts as low as possible. Thanks, Sagar
The overall article is very good but I found a small mistake in the Types of Ab-Ag Reactions. There you have written Enzyme inked Immunosorbent Assay (ELISA), the inked word should be replaced with ‘LINKED’.
Thank you so much for the correction.
Thank you so much