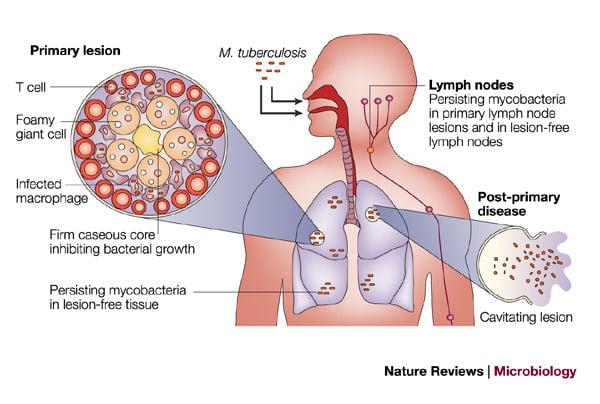
Habitat of Mycobacterium tuberculosis
- Human is only a known reservoir.
- Usually found in the lungs but can also live in any part of the body.
- They may be obligate pathogen, facultative or opportunistic pathogens, or free living.
- Usually infect the mono-nuclear phagocytes.
- Can also be infected by the consumption of unpasteurized milk.
- Can survive for weeks in the dust, on carpets or clothes, for months in sputum.
- They are also found in soil and water.
Morphology of Mycobacterium tuberculosis
- Straight or slightly curved thin rod-shaped bacilli.
- Non-sporing, non-motile, non-capsulated bacteria.
- Acid-fast bacilli, neither gram +ve nor gram –ve.
- During acid-fast stain, they appear bright red to intensive purple with green/blue background.
- They measure 0.5 µm x 3 µm.
- Appears in single, in pairs, or small chumps.
- The high content of mycolic acid (50 to 60 %).
- The cell wall is rich in lipids and waxes.
- They are wrapped together due to the presence of fatty acids.
- Capable of intracellular growth.
- They are resistant to disinfectants, detergents, common antibiotics, dyes, stains, osmotic lysis, lethal oxidation, etc.
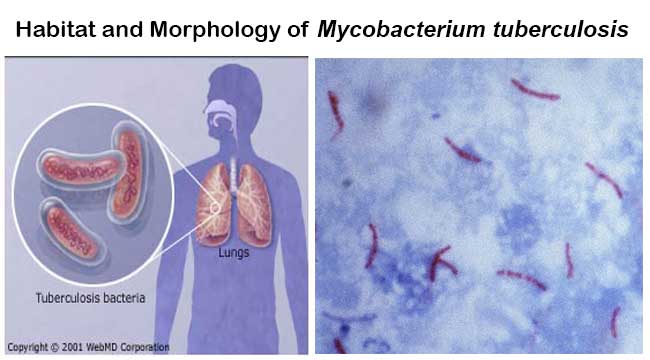
Cultural Characteristics of Mycobacterium tuberculosis
- Culture is the gold standard for laboratory confirmation of Tuberculosis.
- Growing bacteria are required to perform drug-susceptibility testing and genotyping.
In the Lowenstein-Jensen (LJ) medium
- It is an egg-based medium and growth is quite slow.
- It takes 6-8 weeks to get visual colonies on this type of media.
- Colonies are non-pigmented, dry, rough, raised, irregular with a wrinkled surface
- They are creamy-white initially, becoming yellowish or buff-colored on further incubation.
- Growth is eugonic (grows more luxuriantly in culture).
- The optimum temperature is 35-37°C and the optimum pH is 6.4 to 7.
- It is an obligate aerobe.
- Increased carbon dioxide (5-10%) enhances the growth of Mycobacterium tuberculosis.
- 5% Glycerol also stimulates the growth.
- LJ media contain inhibitors to keep contaminants from out-growing Mycobacterium tuberculosis.
- The green color of the medium is due to the presence of malachite green which is one of the selective agents to prevent the growth of most other contaminants.
- A faster result can now be obtained using the Middlebrook medium or BACTEC.
Other Culture Media used for Mycobacterium tuberculosis
Solid media
- Egg-based – Petragnini medium and Dorset medium
- Middlebrook 7H10 Agar
- Middlebrook 7H11 Agar
- Blood-based – Tarshis medium
- Serum based – Loeffler medium
- Potato based – Pawlowsky medium
Liquid media
- Dubos’ medium
- Middlebrook 7H9 Broth
- Proskauer and Beck’s medium
- Sula’s medium
- Sauton’s medium
Biochemical Characteristics of Mycobacterium tuberculosis
| S.N. | Biochemical Test | Results |
| 1 | 5% NaCl Tolerance | Negative (-ve) |
| 2 | 68°C Catalase Test | Negative (-ve) |
| 3 | Acid Phosphatase | Negative (-ve) |
| 4 | Amidase Test | Positive (+ve) |
| 5 | Arylsulphatase Test | Negative (-ve) |
| 6 | Growth on P-Nitrobenzoic Acid | Negative (-ve) |
| 7 | Growth on TCH (10mg/ml) | Positive (+ve) |
| 8 | Iron Uptake | Negative (-ve) |
| 9 | Neutral Red Test | Positive (+ve) |
| 10 | Nicain Test | Positive (+ve) |
| 11 | Nitrate Reduction Test | Positive (+ve) |
| 12 | Peroxidase Test | Positive (+ve) |
| 13 | Pyrazinamidase Test | Positive (+ve) |
| 14 | Semi-Quantitative Catalase Test | Negative (-ve) |
| 15 | Tellurite Reduction Test | Variable |
| 16 | Tween 80 Hydrolysis | Negative (-ve) |
| 17 | Urease Test | Variable |
Virulence factors of Mycobacterium tuberculosis
A. Mycolic acid and Lipoarabinomannan (LAM)
- Waxy layer that protects the bacteria from many host factors and also to many antibiotics including beta-lactamases.
- Muramyl dipeptide (from peptidoglycan) complexed with mycolic acids can cause granuloma formation, phospholipids induce caseous necrosis.
- LAM is structurally and functionally related to the O antigenic lipopolysaccharides of other bacteria.
- Biological activities of LAM include strong seroreactivity, inhibition of interferon-gamma mediated activation of macrophages, induction of cytokine production and release by macrophages, scavenging reactive oxygen intermediates, and suppression of T-cell proliferation.
- LAM has been identified as a key ligand of dendritic cell (DC)-specific intracellular adhesion molecule-3 grabbing nonintegrin (SIGN).
B. ESX 1 secretion system
- Helps in the secretion of many proteins and enzymes that help the organism to survive in the phagosome.
- Mediates the delivery of bacterial products into the macrophage cytoplasm.
- Prevents the fusion of lysosome to phagosome and hence protects bacteria from phagolysosomal activity.
C. Phagosome maturation factors
- Ure that inhibits the acidification of early phagosome.
- Superoxide dismutase (SOD) that detoxifies host reactive oxygen substances (ROS).
- The nlA gene product might inhibit host defenses by interfering with host cell apoptosis.
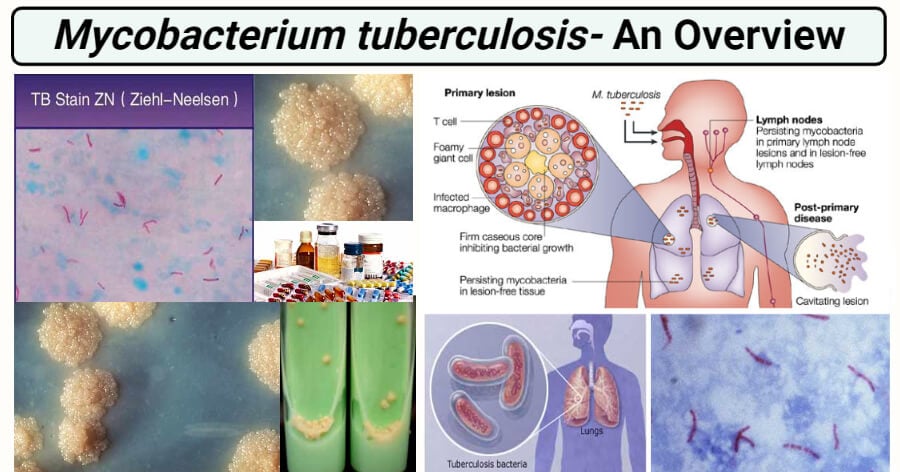
Pathogenesis of Mycobacterium tuberculosis
1. Portal of entry
- The infectious bacilli gain its entry to the human host from inhalation as droplets from the atmosphere.
- The first line of defense of the host tries to ward off the bacteria by tracheal and bronchial epithelium.
- When bacteria survive from the first line of defense, they enter the lungs.
2. Primary tuberculosis
- The bacteria are phagocytosed by alveolar macrophages in the lung, entraps the bacteria in phagosome and action of lysosome along with phagosome collectively called as phagolysosome to kill the bacteria.
- When the bacteria survive the initial host defense mechanism, it attacks alveolar macrophages, gains access to the lung parenchyma, survives in the phagosome, and induces a localized cell-mediated pro-inflammatory response leading to granuloma formation.
- The granuloma protects the bacteria and generally appear after 3 weeks of primary infection.
- When fully developed, this lesion, a chronic granuloma, consists of three zones
- a central area of large, multinucleated giant cells containing tubercle bacilli;
- a mid-zone of pale epithelioid cells often arranged radially; and
- a peripheral zone of fibroblasts, lymphocytes, and monocytes.
- The center of the granuloma contains a mixture of necrotic tissue and dead macrophages.
- Being metabolically very active, the macrophages in the granuloma consume oxygen, and the resulting anoxia and acidosis in the center of the lesion probably kill most of the bacilli.
- Granuloma formation is usually sufficient to limit the primary infection.
- The lesions become quiescent and surrounding fibroblasts produce dense scar tissue, which may become calcified called ghon focus.
- Programmed cell death (apoptosis) of bacteria-laden macrophages by cytotoxic T cells and natural killer (NK) cells contributes to protective immunity by generating a metabolic burst that kills tubercle bacilli.
3. Latent tuberculosis
- In most infected individuals, the primary infection resolves but some residual tubercle bacilli enter a poorly understood stage of latency or dormancy.
- This is mostly observed in immunocompromised cases eg. HIV or aging.
4. Post-primary tuberculosis
- Reactivation of dormant foci of tubercle bacilli or exogenous reinfection leads to post-primary tuberculosis, which differs in several respects from the primary disease.
- For unknown reasons, reactivation or reinfection tuberculosis tends to develop in the upper lobes of the lungs.
- Proteases liberated by activated macrophages soften and liquefy the caseous material, and an excess of tumor necrosis factor and other immunological mediators causes the wasting and fevers characteristic of the disease.
- The interior of the tuberculoma is acidic and anoxic and contains few viable tubercle bacilli.
- Eventually, the expanding lesion erodes through the wall of a bronchus, the liquefied contents are discharged and a well-aerated cavity is formed.
- The atmosphere of the lung, with a high carbon dioxide level, is ideal for supporting the growth of the bacilli, and huge numbers of these are found in the cavity walls.
- Once the cavity is formed, large numbers of bacilli gain access to the sputum, and the patient becomes an open or infectious case.
- In post-primary tuberculosis, dissemination of bacilli to lymph nodes and other organs is unusual.
- Instead, the spread of infection occurs through the bronchial tree so that secondary lesions develop in the lower lobes of the lung and, occasionally, in the trachea, larynx, and mouth.
- Bacilli in swallowed sputum cause intestinal lesions.
- Secondary lesions may also develop in the bladder and epididymis in cases of renal tuberculosis.
- Post-primary cutaneous tuberculosis (lupus vulgaris) usually affects the face and neck.
Clinical manifestation of tuberculosis
1. Primary or pulmonary tuberculosis
- Primary tuberculosis typically is considered a disease of the respiratory tract.
- Common presenting symptoms include:
- low-grade fever
- night sweats
- fatigue
- anorexia(loss of appetite)
- weight loss.
- A patient who presents with pulmonary tuberculosis usually has a productive cough, along with low-grade fever, chills, myalgias (aches), and sweating; however, these signs and symptoms are similar for influenza, acute bronchitis, and pneumonia.
2. Disseminated tuberculosis
- Organs besides the lungs can become involved after infection with M. tuberculosis complex organisms.
- These organs include the following:
- Genitourinary tract
- Lymph nodes (cervical lymphadenitis)
- The central nervous system (meningitis)
- Bone and joint (arthritis and osteomyelitis)
- Peritoneum
- Pericardium
- Larynx
- Pleural lining (pleuritis)
Laboratory diagnosis of tuberculosis caused by Mycobacterium tuberculosis
Specimen and processing
- sputum, bronchial washings, brushings or biopsies or early morning gastric aspirates, Cerebrospinal Fluid (CSF), urine
- Specimens from sputum and other nonsterile sites should be liquefied with N-acetyl-L-cysteine decontaminated with NaOH (kills many other bacteria and fungi), neutralized with buffer, and concentrated by centrifugation.
- Specimens from sterile sites, such as cerebrospinal fluid, do not need the decontamination procedure but can be directly centrifuged, examined, and cultured.
Direct detection methods
1. Microscopy
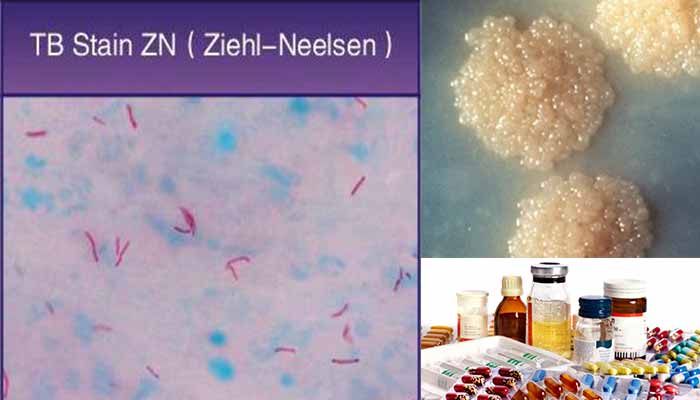
- Detection of the acid-fast property of mycobacteria to detect them in sputum and other clinical material by the Ziehl–Neelsen (ZN) staining technique.
- Red bacilli are seen against the contrasting background color.
- Slender, straight, or slightly curved rods with a barrel or beaded appearance.
- Fluorescence microscopy, based on the same principle of acid-fastness, is increasingly used and is much less tiring.
2. Culture
- As sputum and certain other specimens frequently contain many bacteria and fungi that would rapidly overgrow any mycobacteria on the culture media, decontamination methods make use of the relatively high resistance of mycobacteria to acids and certain disinfectants.
- The deposit is used to inoculate the Lowenstein Jensen (LJ) medium.
- Specimens such as cerebrospinal fluid and tissue biopsies, which are unlikely to be contaminated, are inoculated directly onto culture media.
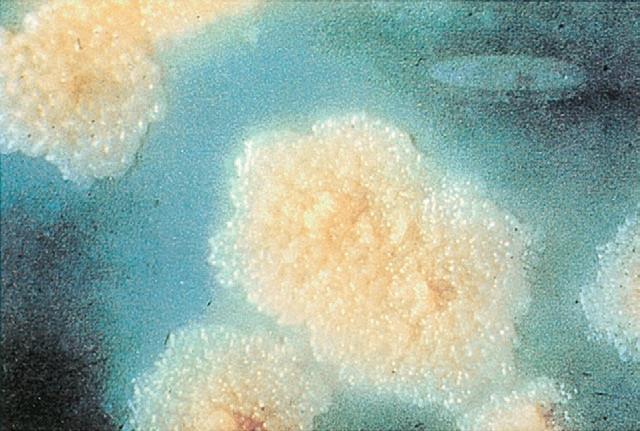
- Dry, rough, raised, wrinkled, off white to buff-colored colonies on LJ medium, commonly called as rough, tough, and buff colonies.
- The use of the liquid medium with radiometric growth detection such as BACTEC 460 TB has simplified the culture method.
Figure: Colony morphology of Mycobacterium tuberculosis on Lowenstein Jensen (LJ) agar after 8 weeks of medium
3. Biochemical analysis
- Niacin accumulation test: positive
- Arylsulphatase test: positive
- Neutral red test: positive
- Catalase peroxidase test: peroxidase positive and weakly catalase-positive
- Amidase test: positive
- Nitrate reduction test: positive
- Tween 80 hydrolysis: variable result
- Susceptibility to pyrazinamide: sensitive
- Susceptible to thiophene -2-carboxylic acid hydrazide (TCH): not susceptible
4. Animal inoculation
- For many years, guinea pig inoculation had been a popular method of diagnosis of tuberculosis, but it is now regarded as obsolete.
5. Serodiagnosis
- ELISA techniques have been attempted for rapid diagnosis of different clinical forms of tuberculosis by estimating specific IgM, IgA, and IgG antibody titers employing the use of various mycobacterial antigens.
- Latex agglutination test with latex particle coated with rabbit antibody to M tuberculosis has a sensitivity of 94.4% and specificity of 2%.
6. Antigen- protein detection
- The detection of microbial products or components has been used in recent years to diagnose infections caused by M. tuberculosis.
- Tuberculostearic acid is a fatty acid that can be extracted from the cell wall of mycobacteria and detected by gas chromatography or mass spectrometry in clinical samples containing few mycobacteria.
7. Molecular techniques
- Subsequent to the introduction of commercially available hybridization assays, commercially available and inhouse– developed nucleic acid amplification tests were used successfully for early identification of M. tuberculosis complex grown in liquid cultures.
- PCR-based sequencing for mycobacterial identification consists of PCR amplification of mycobacterial DNA with genus-specific primers and sequencing of the amplicons.
- The organism is identified by comparison of the nucleotide sequence with reference sequences.
- The most reliable sequence for the identification of mycobacteria is the approximately 1500 bp 16S rRNA gene.
Indirect detection methods
1. Tuberculin test
- A purified protein derivative (PPD) is obtained by chemical fractionation of old tuberculin.
- PPD is standardized in terms of its biologic reactivity as tuberculin units (TU).
- A large amount of tuberculin injected into a hypersensitive host may give rise to severe local reactions and a flare-up of inflammation and necrosis at the main sites of infection (focal reactions).
- For this reason, tuberculin tests in surveys use 5 TU in 0.1 mL solution; in persons suspected of extreme hypersensitivity.
- After the tuberculin skin test is placed, the area is examined for the presence of induration no later than 72 hours after placement.
The result interpretation is given as:
- For patients, at the highest risk of developing the active disease (eg, HIV-infected persons, people who have had exposure to persons with active tuberculosis) 5 mm or larger of induration is considered positive.
- Larger than 10 mm is considered positive for persons with an increased probability of recent infection. This category might include individuals such as recent immigrants from high-prevalence countries, injection drug users, and health care workers with exposure to tuberculosis.
- For persons at low risk for tuberculosis, 15 mm or larger of induration is considered a positive test result.
- Positive test results tend to persist for several days. Weak reactions may disappear more rapidly.
2. Interferon-gamma release assay (IGRA)
- Sometimes the results of the tuberculin skin test are equivocal, particularly in persons who have been vaccinated with BCG or who live in areas where NTM are highly prevalent in the environment.
- In an effort to improve diagnostic accuracy, whole-blood interferon release assays (IGRAs) have been commercially developed.
- These assays are based on the host’s immune responses to specific M tuberculosis antigens ESAT-6 (early secretory antigenic target-6), CFP-10 (culture filtrate protein-10), and TB7.7.
- The tests detect interferon-gamma that is released by sensitized CD4 T cells in response to these antigens.
Two commercial assays generally used to detect the interferon are:
- The Quantiferon-Gold In-Tube test (QFT-GIT) is an enzyme-linked immunosorbent assay (ELISA) that detects interferon-gamma in whole blood.
- The T-SPOT-TB is an ELISA ImmunoSpot assay that uses purified peripheral blood mononuclear cells.
Treatment of tuberculosis
The first line of anti-TB agents that form the core of treatment regimens are
- Isoniazid (INH)
- Rifampin (RIF)
- Pyrazinamide (PZA)
- Ethambutol (EMB)
- The current World Health Organization recommendations are that all new patients with tuberculosis, irrespective of site or severity of the disease, and in the absence of evidence of drug resistance, should receive a 6-month course of therapy, consisting of a 2-month intensive phase of rifampicin, isoniazid, pyrazinamide, and ethambutol followed by a 4-month phase of rifampicin and isoniazid.
- Isoniazid may cause mild psychiatric disturbances and peripheral neuropathy, particularly in alcoholics, but these are usually preventable by prescribing pyridoxine (vitamin B6).
- Resistance may develop during therapy (acquired resistance) with poor-quality drugs or inadequate supervision, or patients may be infected with resistant strains (initial or primary resistance).
- Multidrug resistant strains are defined as those resistant to isoniazid and rifampicin, with or without resistance to additional drugs.
- The newer category of extensively drug-resistant tuberculosis (XDR-TB) is defined as resistance to, at least, isoniazid, rifampicin, fluoroquinolone, and any injectable agent.
The second line drugs include
- A later generation of fluoroquinolones such as moxifloxacin, levofloxacin, or gatifloxacin.
- An injectable agent such as amikacin, kanamycin, or capreomycin.
- Two or more core second-line agents include ethionamide, prothionamide, cycloserine, terizidone, clofazimine, or linezolid.
Prevention of tuberculosis
Human tuberculosis is preventable:
- by the early detection and effective therapy of the open or infectious individuals in a community
- by lowering the chance of infection by reducing overcrowding as the most important factors affecting the incidence of tuberculosis are socio-economic ones, particularly those leading to a reduction of overcrowding in homes and workplaces.
- to a limited extent, by vaccination which includes BCG vaccination that is given by intracutaneous injection after the birth.
- neonatal vaccination is recommended as prior exposure of the human population to environmental mycobacteria confer some protection, but in others induce inappropriate immune reactions that antagonize protection.
References and Sources
- http://textbookofbacteriology.net/tuberculosis.html
- http://web.uconn.edu/mcbstaff/graf/Student%20presentations/Mtuberculosis/Mtuberculosis.html
- http://www.austincc.edu/microbio/2421a/tb
- http://www.meddean.luc.edu/lumen/MedEd/medicine/pulmonar/diseases/tbthc1.htm
- http://www.microbeworld.org/component/jlibrary/?view=article&id=6641
- http://www.microbiologyinpictures.com/mycobacterium%20tuberculosis.html
- http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3865090/
- http://www.sigmaaldrich.com/technical-documents/articles/microbiology-focus/mycobacteria-identification.html
- http://www.slideshare.net/deshkar/mycobacterium-tuberculosis-seminar
- https://en.wikipedia.org/wiki/Löwenstein–Jensen_medium
- https://labtestsonline.org/understanding/analytes/afb-culture/tab/test/
- https://microbewiki.kenyon.edu/index.php/Mycobacterium_tuberculosis

Good work. Keep it up
A scholarly comprehensive article on TB with beautiful diagram/illustrations.
Well done
Good job