mRNA vaccines are suspensions of active (live) or inactivated microorganisms, toxins, or other disease-causing entities. Generally, weakened or inactivated components of pathogens or their antigenic proteins activate the immune system without causing disease. Later, during subsequent infections, this activated immune system recognizes the antigen and fights against it.

mRNA vaccines use messenger RNA (mRNA) to instruct the body’s immune cells to produce a specific antigen. This antigen is detected by the immune system, which creates antibodies. These antibodies are important in targeted immune response in subsequent contact with the same antigen. In comparison to traditional vaccines, which use live or inactivated components of pathogens, mRNA vaccines only provide the genetic instruction to produce antigens rather than the antigens themselves.
History and Development of mRNA Vaccine Technology
- The discovery of mRNA in 1961 as the intermediate between DNA and protein provided the theoretical basis for present prospects in mRNA-based advancements.
- The practical use of mRNA was noted in 1989, when Malone and team demonstrated that mRNA enclosed in a liposomal nanoparticle could be successfully transfected into eukaryotic cells. Soon, the direct intramuscular injection of naked mRNA in mice caused detectable protein expression, providing the first evidence that IVT mRNA could deliver genetic information without the use of a vector.
- In 1993, the immunological basis of mRNA vaccines was established as liposome-encapsulated mRNA encoding the nucleoprotein of the influenza virus induced T-cell responses.
- In 1995, it was confirmed that mRNA could also stimulate humoral immunity.
- In 2005, Kariko and Weissman found that nucleoside modification (uridine with pseudouridine) reduced immune recognition and immunogenicity, and improved translational efficiency. This discovery was awarded the 2023 Nobel Prize in Physiology or Medicine.
- The commercial development of mRNA vaccines then began to grow in the following years. The advances in lipid nanoparticle-based delivery systems further strengthened this technology.
- In 2020, mRNA vaccines encoding the SARS-CoV-2 spike protein were designed, which later showed excellent results in clinical trials and real-world practical applications. This catalyzed even more interest in global research on mRNA vaccines, and now even newer developments are underway.
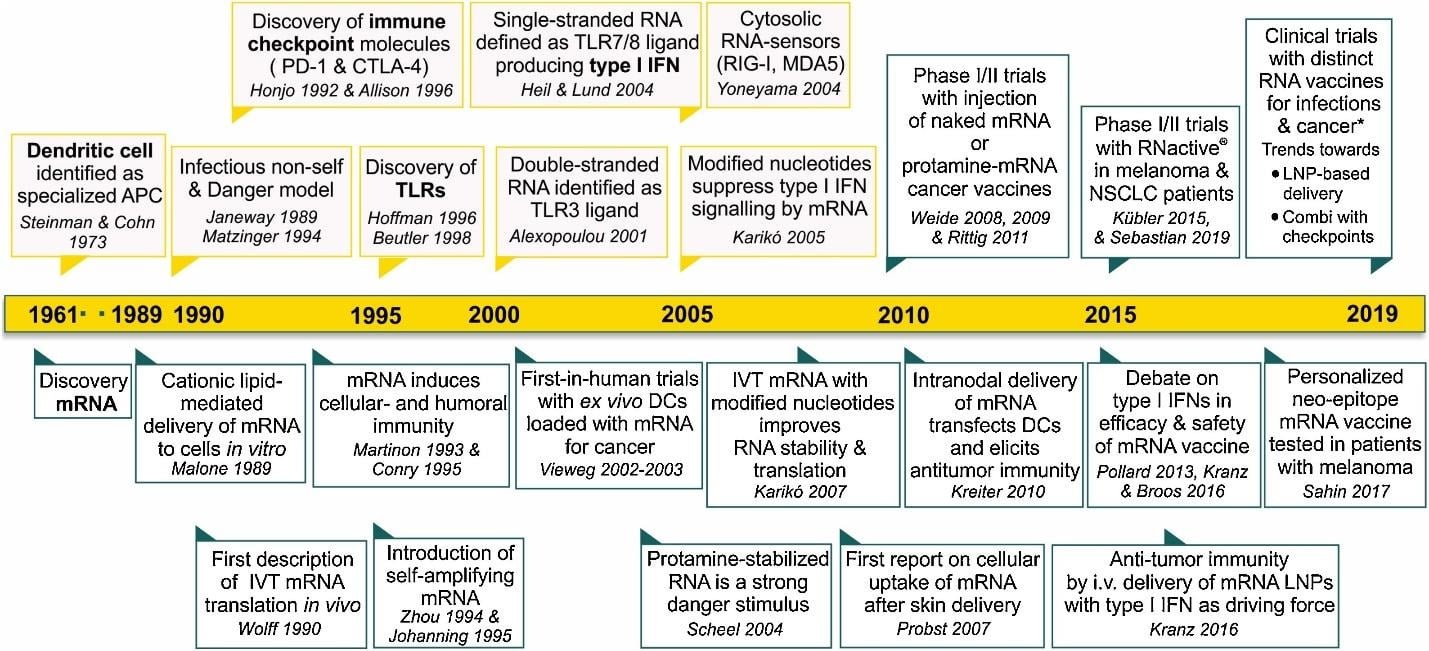
Source: Verbeke, R., Lentacker, I., De Smedt, S., & Dewitte, H. (2019). Three decades of messenger RNA vaccine development. NANO TODAY, 28. https://doi.org/10.1016/j.nantod.2019.100766
Structure and Components of mRNA Vaccines
In an mRNA vaccine, the mRNA is engineered to resemble a fully processed mature mRNA as they naturally occur.
- Open Reading Frame (ORF): The ORF (or the coding sequence) is the core active component of the overall mRNA. It encodes the protein of interest, which acts as an antigen for the immune system.
- 5’ and 3’ Untranslated Regions (UTRs): The ORF is typically flanked by 2 untranslated and regulatory regions (5’ and 3’ UTR). These help to increase mRNA stability and translation efficiency.
- 5’ Cap: It is a methylated guanosine nucleotide connected by a triphosphate linkage to the 5’ end of the ORF. It is important in ribosomal recognition of mRNA, translation initiation, and protection from degradation by exonucleases.
- Poly-A tail: A long sequence (50-250bp) of adenine nucleotides is present at the 3’ end. It protects from exonucleolytic degradation and enhances translation efficiency. mRNA half-life and translation initiation efficiency are also dependent upon the length of the poly-A tail.
- Delivery systems: Lipid nanoparticles (LNP) or polymer-based vehicles encapsulate the mRNA to protect it from degradation by extracellular ribonucleases and effectively help in entering the cell.
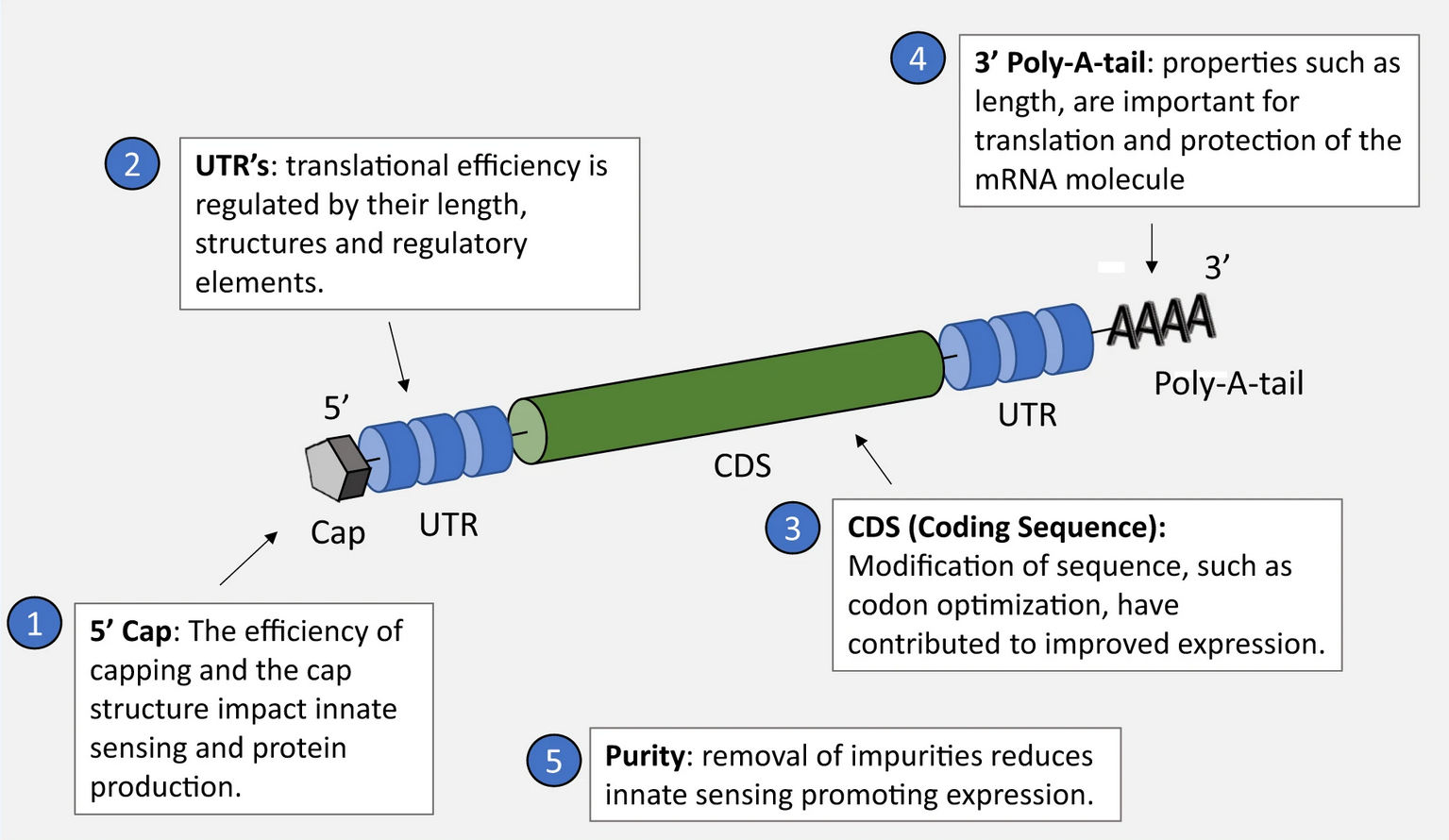
Source: https://www.nature.com/articles/s41541-020-0159-8/figures/2
How mRNA Vaccines Work in the Immune System
The working mechanism of mRNA vaccines involves multiple cellular and immunological activities after the administration of the vaccine. They are:
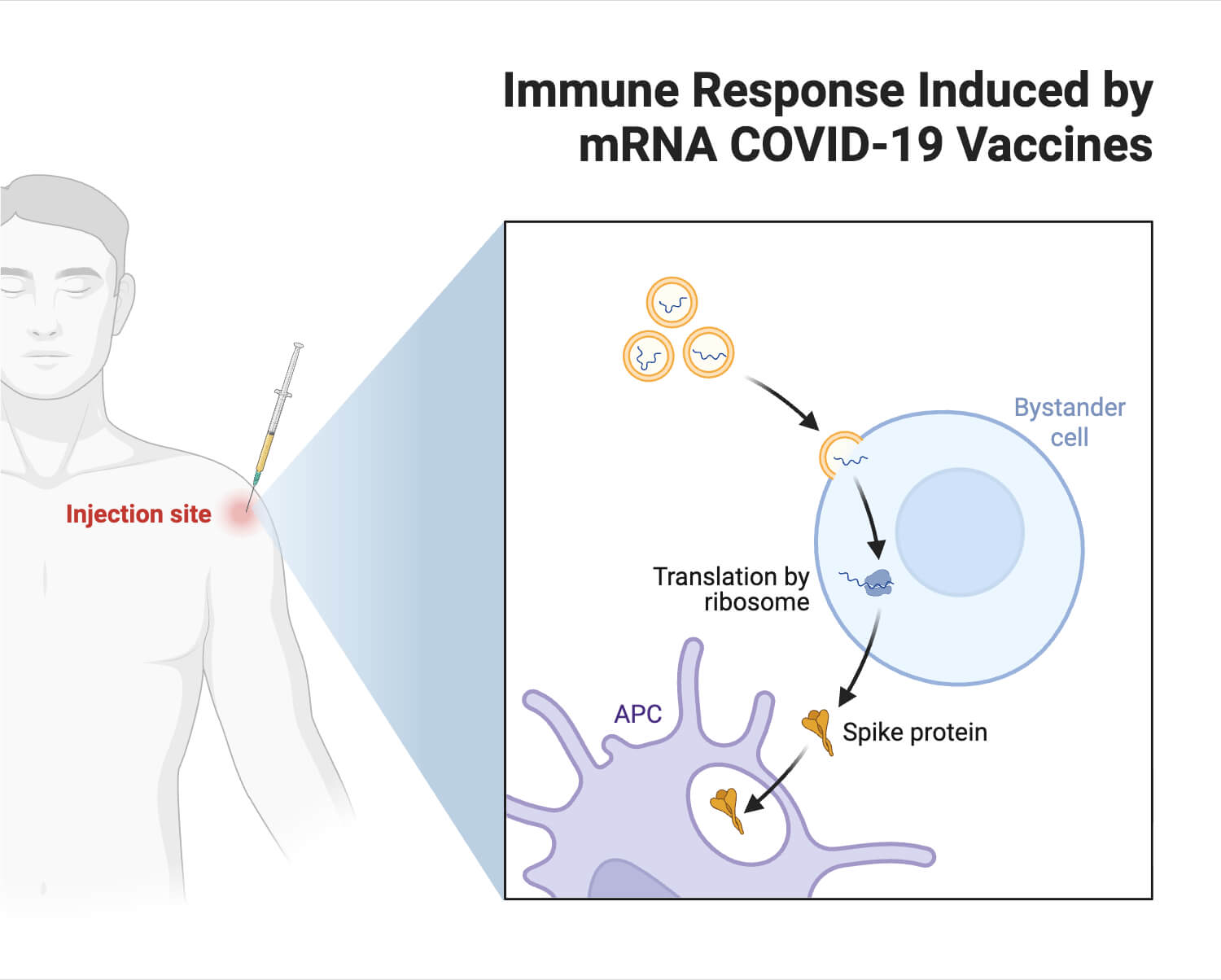
- Delivery and uptake: After intramuscular injection, the plasma membrane of host cells (primarily myocytes or dendritic cells) invaginates and engulfs the encapsulated mRNA through endocytosis to form endosomes.
- Endosomal escape: Once internalized with the endosome, the lipids of the LNP cause endosomal membrane destabilization and the release of mRNA into the cytoplasm.
- Translation: The released mRNA into the cytoplasm is recognized by the cellular ribosomes and is translated into the target protein (antigen). Here, the mRNA neither enters the cell nucleus nor interacts with the host’s genome. The mRNA is generally degraded by the host’s normal physiological processes.
- Antigen presentation: The newly synthesized protein undergoes the proteasomal processing (breakdown of proteins into smaller peptides in an ATP-dependent manner). These smaller peptides (antigens) are presented on the surfaces of dendritic cells and macrophages (antigen-presenting cells) by MHC class I and class II molecules. This antigen presentation activates both the cytotoxic (CD8+) and helper (CD4+) T-cells.
- Activation of humoral and cell-mediated immunity: The activated T-helper cells cause the differentiation of B-lymphocytes into plasma cells. These plasma cells secrete antibodies that are specific to the antigens. Similarly, the cytotoxic T-cells acquire the effector function (by the release of cytotoxic granules, interferons, and interleukins) against the antigen-expressing cells.
- Immunological memory: After the primary immune response, memory B-cells and memory T-cells are generated, which provide immunological memory against subsequent exposure to the pathogen.
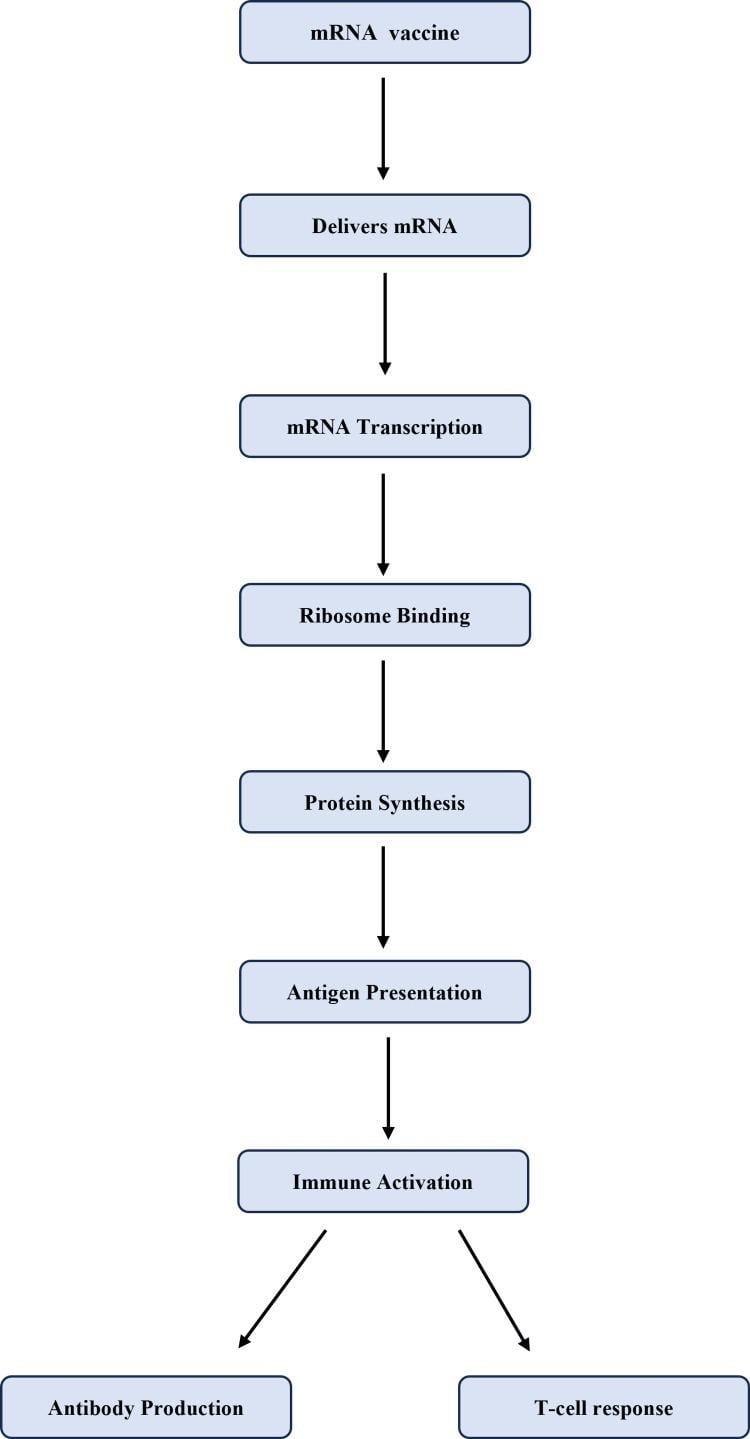
Source: Saxena, S., Mandrah, V., Tariq, W., Das, P., Sambhav, K., & Devi, S. H. (2025). The Future of mRNA Vaccines: Potential Beyond COVID-19. Cureus, 17(5), e84529. https://doi.org/10.7759/cureus.84529
Production and Manufacturing Process of mRNA Vaccines
- Antigen selection and optimization: The target antigen to be produced is first identified. The mRNA coding sequence of the antigen is then optimized using computational tools. It includes epitope selection, codon optimization, and UTRs and nucleoside modifications to obtain maximum stability and protein expression.
- DNA template synthesis: After sequence optimization, a linear plasmid DNA or synthetic double-stranded DNA template that encodes for the optimized target mRNA sequence is produced. This step is crucial because RNA polymerases can only read DNA sequences and convert them into mRNA.
- In vitro transcription (IVT): It is the central process in the manufacturing of mRNA vaccines. The linearized DNA template is incubated with an RNA polymerase, NTPs (along with chemically modified nucleotide, such as N1-methylpseudouridine; m1Ψ), and appropriate buffers for efficient enzymatic activity. The RNA polymerase transcribes the DNA template in a cell-free reaction. Large bioreactors are used to produce a high quantity of mRNA in a short time.
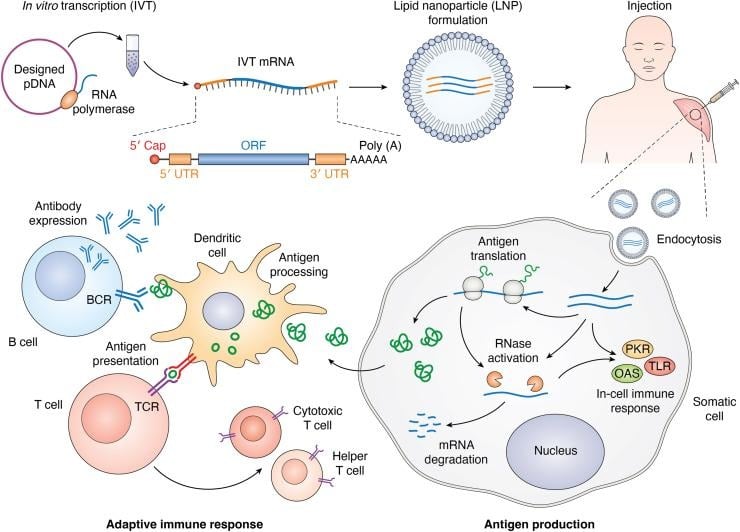
Source: Jin, L., Zhou, Y., Zhang, S., & Chen, S. J. (2025). mRNA vaccine sequence and structure design and optimization: Advances and challenges. The Journal of biological chemistry, 301(1), 108015. https://doi.org/10.1016/j.jbc.2024.108015
- Purification: The transcribed mRNA is then purified to remove residual DNA, enzymes, or proteins, as these products can activate an unintended immune response. Firstly, ds-DNA is removed using DNase treatment. Additionally, HPLC, affinity chromatography, or ion-exchange chromatography are employed to purify mRNA.
- Encapsulation: The purified mRNA is encapsulated with lipid nanoparticles. LNP-mRNA formulations can be prepared by rapidly mixing the aqueous mRNA and the ethanolic solution of lipids through a controlled flow rate. The pH differences between the two flow states cause spontaneous self-assembly of LNPs around the mRNA.
- Filtration: The encapsulated mRNA-LNP undergoes tangential flow filtration (TFF) for purification. This ensures ethanol, exchange buffers, and non-capsulated mRNA are removed.
- Vaccine formulation and quality control: The final product is suspended in a compatible formulation buffer, which commonly contains sucrose (to maintain its integrity during freezing). Before release, the product is then subjected to stringent quality control (QC) testing to confirm integrity, sterility, and potency.
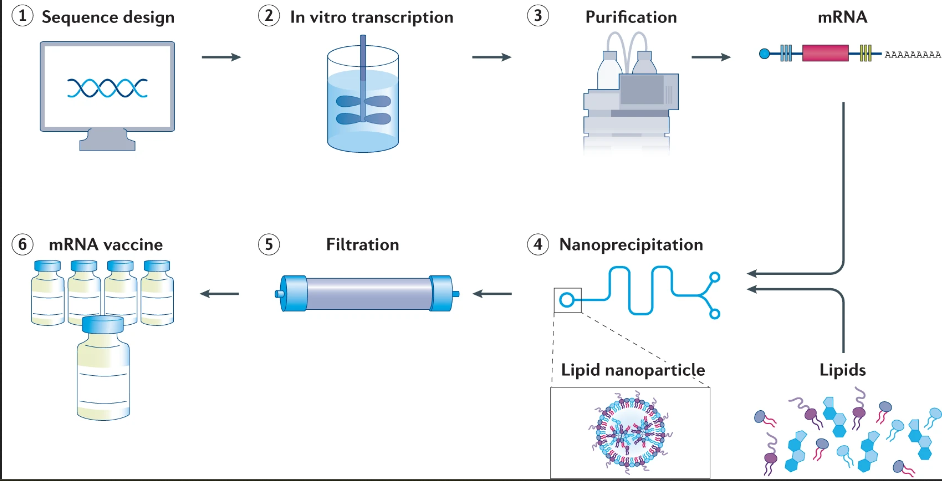
Source: Chaudhary, N., Weissman, D. & Whitehead, K.A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat Rev Drug Discov 20, 817–838 (2021). https://doi.org/10.1038/s41573-021-00283-5
Applications of mRNA Vaccines in Infectious Diseases
mRNA vaccines have been used for a broad range of infectious diseases, particularly for life-threatening viral infections.
- Influenza: mRNA-based influenza vaccine candidates are developed to target the surface proteins of the influenza virus, with the ability to rapidly update according to the strain type (in case of antigenic shift and drift in Influenza).
- Respiratory Syncytial Virus (RSV): mRNA vaccines for RSV, which causes lower respiratory tract disease (LRTD), demonstrate high efficacy in lowering RSV-associated LRTD.
- Human Immunodeficiency Virus (HIV): The development of early-stage trials of mRNA vaccines such as IAVI’s G001 and G002, and Moderna’s mRNA-1644, has demonstrated a proof of concept for targeted antibodies for HIV.
- Rabies: mRNA-based rabies vaccines, encoding the rabies glycoprotein RABV-G, induce both strong antibody and cell-mediated immune response as compared to traditional vaccines.
Additional mRNA vaccines are also applied for the treatment and prevention of Zika Virus, Cytomegalovirus (CMV), and Epstein-Barr Virus (EBV).
Role of mRNA Vaccines in COVID-19 Prevention
mRNA vaccines played an important role in the prevention and treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which caused the COVID-19 pandemic. SARS-CoV-2 utilizes its spike protein (S) for the interaction, fusion, and entry with the host cell; therefore, the S-protein was identified as the primary target antigen against this pathogen.
mRNA vaccines developed against SARS-CoV-2 encode the target spike protein. The Pfizer-BioNTech and Moderna vaccines were among the first to receive approval for widespread use. Within a year, these vaccines underwent clinical trials with thousands of participants, demonstrating high efficacy (~95%) in preventing symptomatic COVID-19 in healthy adults.
These results greatly exceeded the efficacy of most traditional vaccines by showing prevention of severe disease and hospitalization. During the global vaccination campaign, over 13 billion doses were administered by 2022. Epidemiological data revealed that these vaccines saved millions of lives by preventing infection worldwide.
Advantages of mRNA Vaccines
- Rapid development: mRNA vaccines can be synthesized rapidly in the laboratory and on a large scale once the target antigen sequence is known. This allows a rapid public health response to emerging pathogens.
- Cell-free production: mRNA vaccines are produced through in vitro transcription, which is highly scalable and does not require the use of live pathogens or complicated cell culture techniques.
- Non-interacting: mRNA works only in the cytoplasm of the cell, and after its function, it is completely degraded by the cell’s general physiology. Therefore, it doesn’t integrate into the host genome, with no risk of insertional mutagenesis.
- Flexibility: Similar manufacturing platforms or infrastructures can be applied to produce vaccines against different pathogens and their different target antigens. Only the encoding mRNA sequence needs to be altered for this flexibility.
Limitations of mRNA Vaccines
- Thermostability: mRNA vaccines are highly susceptible to degradation and require storage at very low temperatures (-80°C). This poses logistical challenges in areas with limited cold chain infrastructures.
- Reactogenicity: The efficient delivery of mRNA vaccine is dependent upon LNP formulations, which are associated with both local (injection site) and systemic reactogenicity in many recipients.
- Reduced antiviral titers: As compared to live vaccines, the antiviral antibody titers induced by mRNA-based vaccines are known to rapidly decrease within 2 to 3 months after vaccination, requiring continuous booster doses for effective action.
- Limited long-term data: As a relatively new vaccine technology, the long-term immunological efficiency, durability, and safety profiles of mRNA vaccines require more evaluation.
Safety, Side Effects, and Effectiveness of mRNA Vaccines
- Safety Profile: mRNA vaccines are extensively evaluated through clinical trials and post-application pharmacovigilance programs. Large-scale clinical trials establish an acceptable safety standard for mRNA vaccines. Serious vaccine-related adverse effects during clinical trials could disqualify their authorization.
- Common side effects: Local effects, such as pain, rashes, lesions, redness, or swelling in the injection site; and systemic effects, such as fatigue, headache, myalgia, fever, and chills, are some of the side effects of mRNA vaccines. These are temporary and self-resolving.
- Rare adverse events: Myocarditis and pericarditis are mostly observed in younger males after the second vaccine dose (in case of the COVID-19 vaccine).
- Effectiveness: Data from clinical trials and real-world applications have demonstrated a high protective and preventive efficiency of mRNA vaccines, evidenced by their activity against emerging pathogens across multiple demographic groups and populations.
mRNA Vaccines vs. Traditional Vaccines
Traditional vaccines include live-attenuated, inactivated, and protein subunit vaccines. These vaccines provide immunity through direct introduction of the pathogen, or pathogenic material, or their isolated proteins. Production of these vaccines requires pathogen culture, removal of their pathogenicity (attenuation), and protein isolation. Also, these vaccines possess a risk of virulence, and are time-consuming for large-scale manufacturing. Inactivated and protein subunit vaccines are stable at refrigeration temperature and do not generally require multiple boosters for long-term effectiveness.
mRNA vaccines, however, provide genetic instruction for antigen production inside the cell without involving a live pathogen at any stage of manufacturing or application. The antigen sequence is first identified, which is then modified to produce the vaccine. These vaccines also possess no risk of insertional mutagenesis in the host and are degraded following translation of target antigens. Despite these advantages, mRNA vaccines require ultra-low temperatures for storage and shipment, and require continuous multiple booster doses for long-term effectiveness.
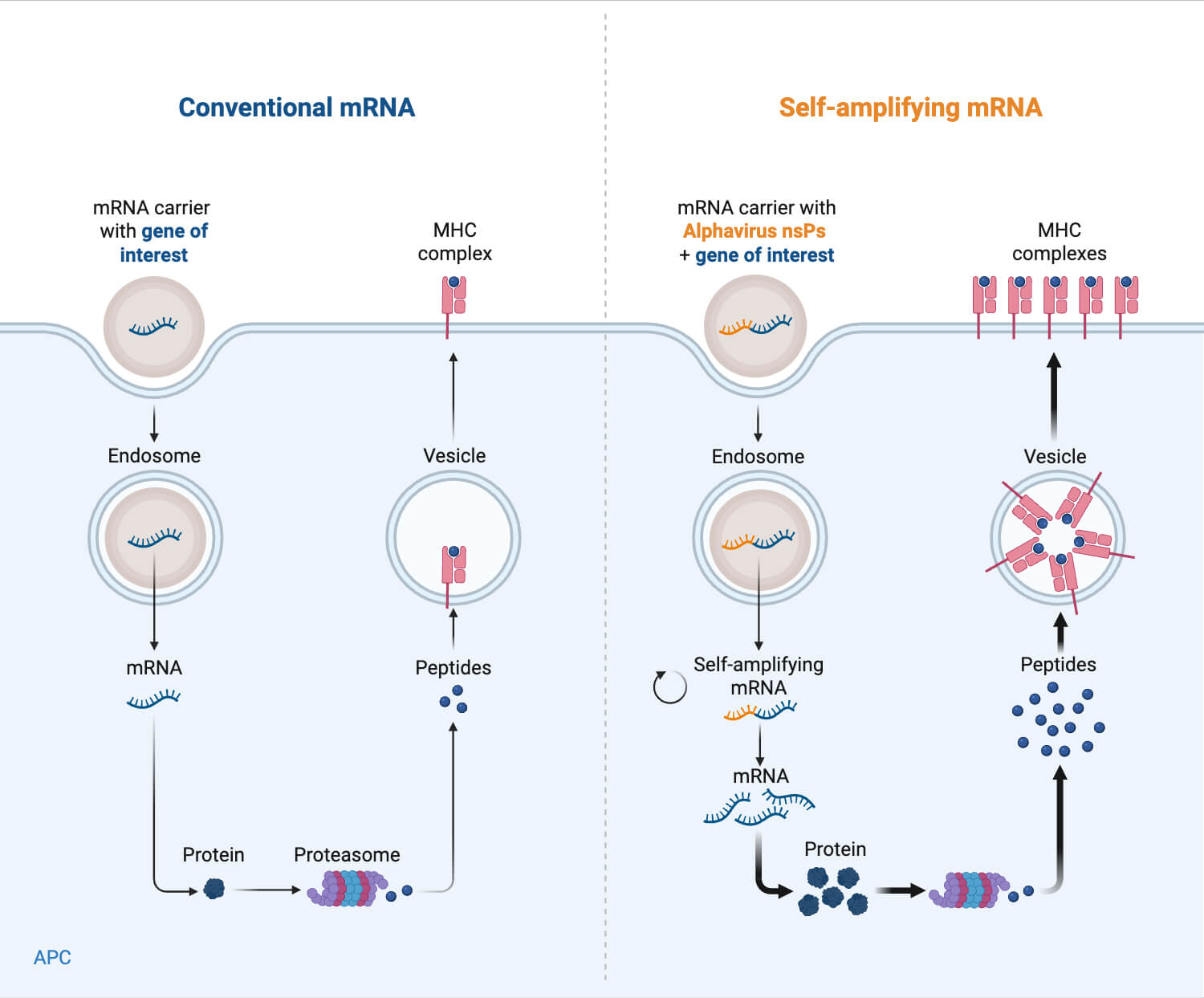
Future Prospects of mRNA Vaccine Technology
- Broader applications of mRNA vaccines as personalized cancer vaccines with patient-specific tumor antigens to activate immune responses directed directly towards tumors.
- Advancements in the formulation of LNPs and heat stability are underway to improve storage stability and cold chain accessibility for mRNA-based vaccines.
- Development of vaccines for organisms or pathogens with high antigenic variability, for example, Influenza, while simultaneously targeting multiple strains for broader immunity.
- Self-amplifying RNA (saRNA) based vaccines can utilize viral replication machinery to amplify antigen production inside the cell, requiring lower doses with more durable immune responses.
Conclusion
mRNA vaccines have changed the field of immunization by demonstrating high efficacy and safety in preventing disease, as evidenced by the success of mRNA COVID-19 vaccines. The rapid development ability of these vaccines makes them an ideal response to global emerging pathogens. However, the limitations in stability and global accessibility should be considered as well.
References
- https://www.who.int/news-room/questions-and-answers/item/vaccines-and-immunization-what-is-vaccination
- Pardi, N., Hogan, M., Porter, F. et al. mRNA vaccines — a new era in vaccinology. Nat Rev Drug Discov 17, 261–279 (2018). https://doi.org/10.1038/nrd.2017.243
- Li, J., Liu, Y., Dai, J., Yang, L., Xiong, F., Xia, J., Jin, J., Wu, Y., & Peng, X. (2025). mRNA Vaccines: Current Applications and Future Directions. MedComm, 6(11), e70434. https://doi.org/10.1002/mco2.70434
- Verbeke, R., Lentacker, I., De Smedt, S., & Dewitte, H. (2019). Three decades of messenger RNA vaccine development. NANO TODAY, 28. https://doi.org/10.1016/j.nantod.2019.100766
- Chaudhary, N., Weissman, D. & Whitehead, K.A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat Rev Drug Discov 20, 817–838 (2021). https://doi.org/10.1038/s41573-021-00283-5
- Hou, X., Zaks, T., Langer, R. et al. Lipid nanoparticles for mRNA delivery. Nat Rev Mater 6, 1078–1094 (2021). https://doi.org/10.1038/s41578-021-00358-0
- Jin, L., Zhou, Y., Zhang, S., & Chen, S. J. (2025). mRNA vaccine sequence and structure design and optimization: Advances and challenges. The Journal of biological chemistry, 301(1), 108015. https://doi.org/10.1016/j.jbc.2024.108015
- Wilson, E., Goswami, J., Baqui, A. H., et al. (2023). Efficacy and Safety of an mRNA-Based RSV PreF Vaccine in Older Adults. New England Journal of Medicine, 389(24), 2233–2244. DOI: 10.1056/NEJMoa2307079
- https://www.iavi.org/press-release/two-hiv-vaccine-trials-show-proof-of-concept-for-pathway-to-broadly-neutralizing-antibodies/
- Li, J., Liu, Q., Liu, J., Wu, X., Lei, Y., Li, S., Zhao, D., Li, Z., Luo, L., Peng, S., Ou, Y., Yang, H., Jin, J., Li, Y., & Peng, Y. (2022). An mRNA-based rabies vaccine induces strong protective immune responses in mice and dogs. Virology journal, 19(1), 184. https://doi.org/10.1186/s12985-022-01919-7
- Fang, Z., Yu, P., & Zhu, W. (2024). Development of mRNA rabies vaccines. Human vaccines & immunotherapeutics, 20(1), 2382499. https://doi.org/10.1080/21645515.2024.2382499
- Korzun, T., Moses, A. S., Diba, P., Sattler, A. L., Taratula, O. R., Sahay, G., Taratula, O., & Marks, D. L. (2023). From Bench to Bedside: Implications of Lipid Nanoparticle Carrier Reactogenicity for Advancing Nucleic Acid Therapeutics. Pharmaceuticals (Basel, Switzerland), 16(8), 1088. https://doi.org/10.3390/ph16081088
- https://www.bmj.com/content/387/bmj.q2859/rr
- https://www.who.int/teams/regulation-prequalification/regulation-and-safety/pharmacovigilance
- https://www.cdc.gov/vaccines/covid-19/clinical-considerations/myocarditis.html
- Yaremenko, A. V., Khan, M. M., Zhen, X., Tang, Y., & Tao, W. (2025). Clinical advances of mRNA vaccines for cancer immunotherapy. Med (New York, N.Y.), 6(1), 100562. https://doi.org/10.1016/j.medj.2024.11.015
- Saxena, S., Mandrah, V., Tariq, W., Das, P., Sambhav, K., & Devi, S. H. (2025). The Future of mRNA Vaccines: Potential Beyond COVID-19. Cureus, 17(5), e84529. https://doi.org/10.7759/cureus.84529
