Human T-lymphotropic virus type 1 (HTLV-1) is a retrovirus responsible for persistent, lifelong infection in humans. It spreads mainly through breastfeeding, sexual activity, contaminated blood transfusion, and shared needles.

HTLV-1 has worldwide distribution but is particularly prevalent in Latin America and the Caribbean. Two main types exist, HTLV-1 and HTLV-2, with HTLV-1 being dominant and most often implicated in human disease.
Taxonomy and Classification of HTLV-1
Kingdom: Pararnavirae
Phylum: Artverviricota
Class: Retraviricetes
Order: Ortervirales
Family: Retroviridae
Genus: Deltaretrovirus
Species: Primate T-lymphotrophic Virus-1
Common Name: Human T-lymphotrophic Virus -1 (HTLV-1)
Structure and Morphology of HTLV-1
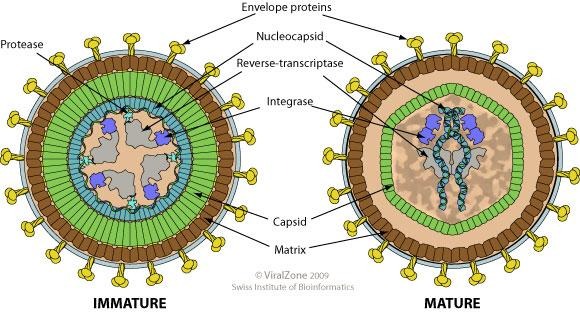
Source: https://viralzone.expasy.org/60?outline=all_by_species
- Virion: spherical to pleomorphic, approximately 80-120nm in diameter.
- Capsid: Icosahedral with helical nucleoprotein.
- Composition: RNA (2%), Protein (60%), Lipid (35%), Carbohydrate (3%)
- Genome: single-stranded RNA, linear, positive-sense, dimeric, 7-11kb
- Proteins: Reverse transcriptase enzyme is contained inside the virions.
- Envelope: present
Genome Organization and Proteins of HTLV-1
- Viral genome is linear, dimeric with a 5’-cap and a 3’ poly-A tail.
- The genome length is approximately 9kb.
- There is the presence of two long terminal repeats (LTRs), which are about 600nt long at the 5’ and 3’ end, which possess the U3, R, and U5 regions.
- At the 5’ end of the genome, there is a primer binding site (PBS), which serves as the initiation point for reverse transcription. At the 3’ end, a polypurine tract (PPT) is present, which acts as a primer for the synthesis of the plus-strand DNA during replication. These elements are essential for the proper replication and integration of the viral genome.
- In addition to the essential genes gag, pol, prt, and env, the HTLV genome also contains the pX region, located between the 3’ end of the genome and the env gene. This pX region encodes several accessory and regulatory genes through multiple open reading frames (ORFs), including tax, rex, and other proteins that play critical roles in viral replication, gene expression, and immune evasion.
- This region possesses four partially ORFs that encode the accessory proteins, the Tax protein (ORF IV), and the Rex post-transcriptional regulator (ORF III).
- The Rex post-transcriptional regulator is responsible for the regulation of viral gene expression post-transcriptionally by facilitating the cytoplasmic expression of the incompletely spliced viral mRNAs.
- Tax proteins start up the initiation of transcription from the viral promoter in the U3 region of the long terminal repeat.
Proteins
-Structural Proteins
From the gag gene
- p19 (matrix protein): Stabilizes viral structure beneath the envelope.
- p24 (Capsid protein): It forms the viral core.
- p15 (Nucleocapsid protein): helps in viral RNA binding.
From the env gene
- gp46 (surface glycoprotein): It enhances the attachment of the virus to the host cell.
- gp 21 (Transmembrane protein): helps in membrane fusion.
-Enzymatic Proteins (from pol gene)
- Reverse Transcriptase (RT): translates RNA into DNA.
- Integrase (IN): helps in the insertion of viral DNA into the genome of the host.
- Protease (PR): breaks precursor protein into functional units.
-Regulatory Proteins (from pX region)
Tax
- Is major transcriptional activator that enhances the viral gene expression.
- It plays a crucial role in cell transformation and oncogenesis.
Rex
- Helps in the regulation of RNA splicing and transport.
- Helps in balancing between the structural and regulatory protein production.
- Accessory Proteins (pX region)
- p12: is involved in immune evasion and activation of T-cells
- p13: influence the function of mitochondria
- p30: helps in the regulation of viral gene expression
Replication Cycle of HTLV-1
The replication cycle of HTLV consists of the following steps:
Attachment and Entry
- The viral surface glycoprotein gp46 binds to the receptors present on CD4+ T-lymphocytes.
- Host receptors possess molecules such as GLUT1, heparan sulfate proteoglycans, and neuropilin-1.
- Then the surface glycoprotein gp21 mediates the fusion of the envelope of the virus with the membrane of the host cell.
- After that, the viral core enters the cytoplasm.
Uncoating
- Then the capsid is detached, releasing RNA and enzymes into the host cytoplasm.
Reverse Transcription
- Viral enzyme reverse transcriptase translates RNA into double-stranded DNA, which takes place in the cytoplasm.
- It then produces proviral DNA copy.
Integration
- After that, the viral DNA moves into the nucleus.
- Then, the enzyme integrase inserts viral DNA into the host genome.
- This integrated form of DNA is known as a provirus.
Transcription
- Then the RNA polymerase of the host cell transcribes the proviral DNA into viral DNA.
- This step is controlled by LTR regions and enhanced by the tax protein.
Translation
- The viral mRNA is then translated into proteins using ribosomes present in the host cell, such as:
- Gag: structural proteins (p12, p24, p15)
- Pol: enzymes (RT, Integrase, Protease)
- Env: gp46, gp21
- Regulatory proteins such as Tax and Rex are also produced in this step.
Assembly
- Assembly of viral RNA and proteins occurs near the host cell membrane.
- Gag proteins are responsible for the formation of immature viral proteins.
Budding and Release
- After that, the new virions bud from the membrane of the host cell.
- It acquires the lipid envelope with embedded glycoproteins.
- Then the protease breaks the precursor proteins into mature infectious virus.
- This mature virus is then released from the cell by budding.
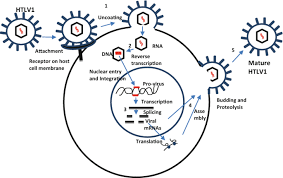
Source: https://link.springer.com/chapter/10.1007/978-981-97-4480-0_27
Pathogenesis and Host Immune Response to HTLV-1
Initial Infection and Proviral Integration
- The virus enters target cells by binding to the receptors of the host cell, such as neuropilin-1, GLUT-1, and heparan sulfate proteoglycans.
- After entry, the RNA genome undergoes reverse transcription following incorporation into the host genome in the form of a provirus.
- This incorporation permits the lifelong persistence and upright transfer of the genome of the virus throughout the cell division of the host cell.
Viral Gene Expression and Immune Evasion
- HTLV demonstrates key regulatory proteins that maintain its pathogenesis.
- The Tax protein functions as a strong transactivator of host and virus genes, enhancing the increase of infected T-cells, improving NF-Kb signaling.
- Tax also disturbs the repair mechanisms of DNA and obstructs the regulation of the cell cycle, influencing genomic instability.
- In contrast, HBZ (HTLV-1 basic leucine zipper factor) reduces the expression of viral gene, aiding in immune evasion, while simultaneously promoting survival and increase of infected cells.
Oncogenesis and Clonal Expansion
- Tax enhances cell division, whereas HBZ sustains the long-term presence of clones.
- Over time, aggregation of mutations and instability of the chromosome cause transformation of malignant cells, leading to ATL.
- This process is usually long, taking about decades before the occurrence of clinical manifestation.
Immunopathogenesis and HAM/TSP
- Along with malignancy, HTLV is responsible for neuroinflammatory disease, most commonly HAM/TSP.
- In contrast to ATL, which develops from clonal expansion, HAM/TSP results from chronic immune activation against HTLV infected cells.
- These infected cells then invade the central nervous system, where they are responsible for causing inflammation and bystander damage, resulting in demyelination and progressive neurological dysfunction.
Host Immune Response and Viral Persistence
- The host immune system exhibits powerful cytotoxic T lymphocyte responses against HTLV, mainly against Tax.
- However, the decrease in expression of Tax and persistence of HBZ allows the virus to evade immune clearance.
- The equilibrium between this immune control and viral evasion leads to the long-term asymptomatic carrier state in most infected persons.
Epidemiology and Transmission of HTLV-1
It is estimated that approximately 15 to 20 million people are infected with HTLV-1 worldwide. This virus is endemic in Japan, Central Australia, parts of Africa, South America, and the Caribbean, and is frequently present in Melanesia, Papua New Guinea, and the Solomon Islands. Even though the global endemics of HTLV-1 have been recognized long ago, an emerging epidemic has been recorded among intravenous drug users in the United States. Japan is traditionally considered the most endemic for HTLV-1. Incidence rates have been reported at 3.8 per 100,000 persons per year, among which a higher rate is observed in women, i.e., 6.9 per 100,000 persons per year, compared to that of men, i.e., 2.3 per 100,000 persons per year. Among patients infected with HTLV-1, approximately 6.6% of men and 2.1% women develop ATL. The epidemiological patterns also show sex-based differences in the expression of disease, which means men are more susceptible to the ATL, while women are most frequently infected by HAM/TSP. According to data from 2012, about 1 to 2.5 million people are living with HTLV-1 in the Pan American region.
There are mainly 3 routes of transmission of the HTLV-1 virus, which are explained below:
Vertical Transmission (from mother to child)
- Breastfeeding is the primary route for the transmission of HTLV-1.
- The virus is present in infected lymphocytes in breast milk.
- The risk of transmission increases with prolonged breastfeeding for more than 6 months.
Sexual Transmission
- It occurs mainly from male to female.
- Transmission occurs via infected lymphocytes, which are present in semen or vaginal secretions.
- Risk factors for this transmission are unprotected sex and multiple sexual partners.
Parenteral Transmission
- Transmitted via blood transfusion or organ transplantation.
- Risk from HTLV-1-infected lymphocytes in donor blood.
- Transmits rarely by shared needles among intravenous drug users.
Clinical Manifestations of HTLV-1
There are various clinical manifestations that are described below:
Asymptomatic Infection
- Virus persists in CD4+ T cells for life-long without any symptoms, which is found in the majority of infected individuals.
Adult T-cell Leukemia / Lymphoma
- Generalized lymphadenopathy-
- Hepatosplenomegaly
- Skin lesions (nodules, plaques, rash)
- Hypercalcemia
HTLV-1 Associated Myelopathy/Tropical Spastic Paraparesis (HAM/TSP)
- Progressive weakness in the lower limbs
- Spastic paraparesis is characterized by stiff and weak legs.
- Hyperreflexia
- Urinary incontinence
- Mild sensory disturbances
HTLV-1 Associated Inflammatory Conditions
- Eye inflammation with blurred vision, pain in the eye, and redness.
- Chronic, recurrent skin infections, especially in children.
- Eczematous lesions mainly on the scalp and face.
- Inflammation and pain in the joint.
Immunological Effects
- Increased susceptibility to opportunistic infections.
- Altered immune regulation due to infected T cells.
Laboratory Diagnosis of HTLV-1
Sample Collection
- Blood (serum or plasma) – antibody detection
- Peripheral blood mononuclear cells (PBMCs) – for molecular tests
- CSF (Cerebrospinal fluid) – for suspected neurological disease (HAM/TSP)
Molecular Tests
PCR (Polymerase Chain Reaction)
- It detects proviral DNA present in the lymphocytes of an infected person.
- It is useful in early infection and neonatal diagnosis.
qPCR (Quantitative PCR)
- It measures the proviral load.
- It is useful in monitoring the progress of the disease and for risk assessment.
Serological Tests
ELISA (Enzyme-Linked Immunosorbent Assay)
- It is useful in the detection of antibodies against HTLV-1 antigens.
- It is used as a screening test and has high sensitivity.
Confirmatory Tests
Western Blot
- It detects antibodies to specific viral proteins such as gag and env.
- It is used for the confirmation of infection after screening.
Line Immunoassay (LIA)
- It is more specific than Western Blot and is used to differentiate between HTLV-1 and HTLV-2.
Treatment and Antiviral Therapy of HTLV-1
There is no cure for infection by the HTLV-1 virus; however, ATL is treated by the following methods:
- Chemotherapy is done as in other lymphoma cases.
- Antiviral drugs such as Zidovudine (Retrovir®) and interferon α are used. However, not all ATL respond to antiviral treatment.
- Stem cell transplant is done; however, many patients with ATL do not receive a stem cell transplant.
For patients with HAM/TSP, the following treatments are given:
- Interferon α: It can lower inflammation and hinder nerve damage.
- Corticosteroids help to lower inflammation.
- For muscle stiffness and spasticity, Baclofen (Lioresal®) or tizanidine is used.
- For urinary discrepancy, Oxybutynin is used.
Prevention and Control of HTLV-1
-Blood Safety Measures
- Prior screening of blood donors for HTLV-1 antibodies.
- Avoid transfusion of infected blood.
- Proper sterilization of medical equipment before use.
-Prevention of Mother-to-Child Transmission
- Limiting or avoiding breastfeeding a child if the mother is diagnosed with the HTLV-1 virus.
- Use of formula milk where feasible and safe.
- Short-term breastfeeding is recommended as it reduces risk compared to prolonged breastfeeding.
-Sexual Transmission Prevention
- Use of a condom during sex.
- Avoiding multiple sexual partners.
- Screening of high-risk persons.
-Prevention among High-Risk Groups
- Avoid sharing needles.
- Screening in endemic areas.
-Prevention in Healthcare Settings
- Proper sterilization of needles and surgical instruments.
- Avoid reuse of contaminated needles or syringes.
-Screening and Surveillance
- Screening of blood donors, organ donors, and pregnant women, especially in endemic regions.
- Monitoring prevalence in communities.
-Health Education
- Public awareness about modes of transmission and safe practices.
- Education and awareness program.
- Counseling infected patients to prevent the spread.
Conclusion
HTLV-1 is a retrovirus of the Human T-cell Lymphotropic virus family, which is found mainly in the Pacific countries, including Australia and Melanesia, as well as in North, Central, and South America. It is transmitted through breastfeeding, needle sharing, unsafe blood transfusion, and sexual contact and causes a spectrum of inflammatory and immunosuppressive diseases. Antiviral medications such as Zidovudine (Retrovir®) and Interferon α are used for the treatment of infections caused by the HTLV-1 virus.
References
- National Center for Biotechnology Information. (2016). Human T-cell lymphotropic virus type 1. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK304341/
- World Health Organization. (2025, December 24). Human T-lymphotropic virus type 1. https://www.who.int/news-room/fact-sheets/detail/human-t-lymphotropic-virus-type-1
- Forouzanfar, E. S., & Nguyen, A. D. (2025). Human T-cell lymphotropic virus. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK560825/
- Iheukwumere, I. H., & Iheukwumere, C. M. (2023). Human T-lymphotropic virus (HTLV): Virology, pathogenesis, epidemiology, and clinical management. ResearchGate. https://www.researchgate.net/publication/396305308_Human_T-Lymphotropic_Virus_HTLV_Virology_Pathogenesis_Epidemiology_and_Clinical_Management
- Kannian, P., & Green, P. L. (2010). Human T-lymphotropic virus type 1 (HTLV-1): Molecular biology and oncogenesis. Viruses, 2(9), 2037–2077. https://doi.org/10.3390/v2092037
- Rosadas, C., Brites, C., Arakaki-Sanchez, D., Casseb, J., & Ishak, R. (2021). Brazilian protocol for sexually transmitted infections 2020: Human T-cell lymphotropic virus (HTLV) infection. Revista da Sociedade Brasileira de Medicina Tropical, 54(Suppl 1), e2020605. https://doi.org/10.1590/0037-8682-605-2020
