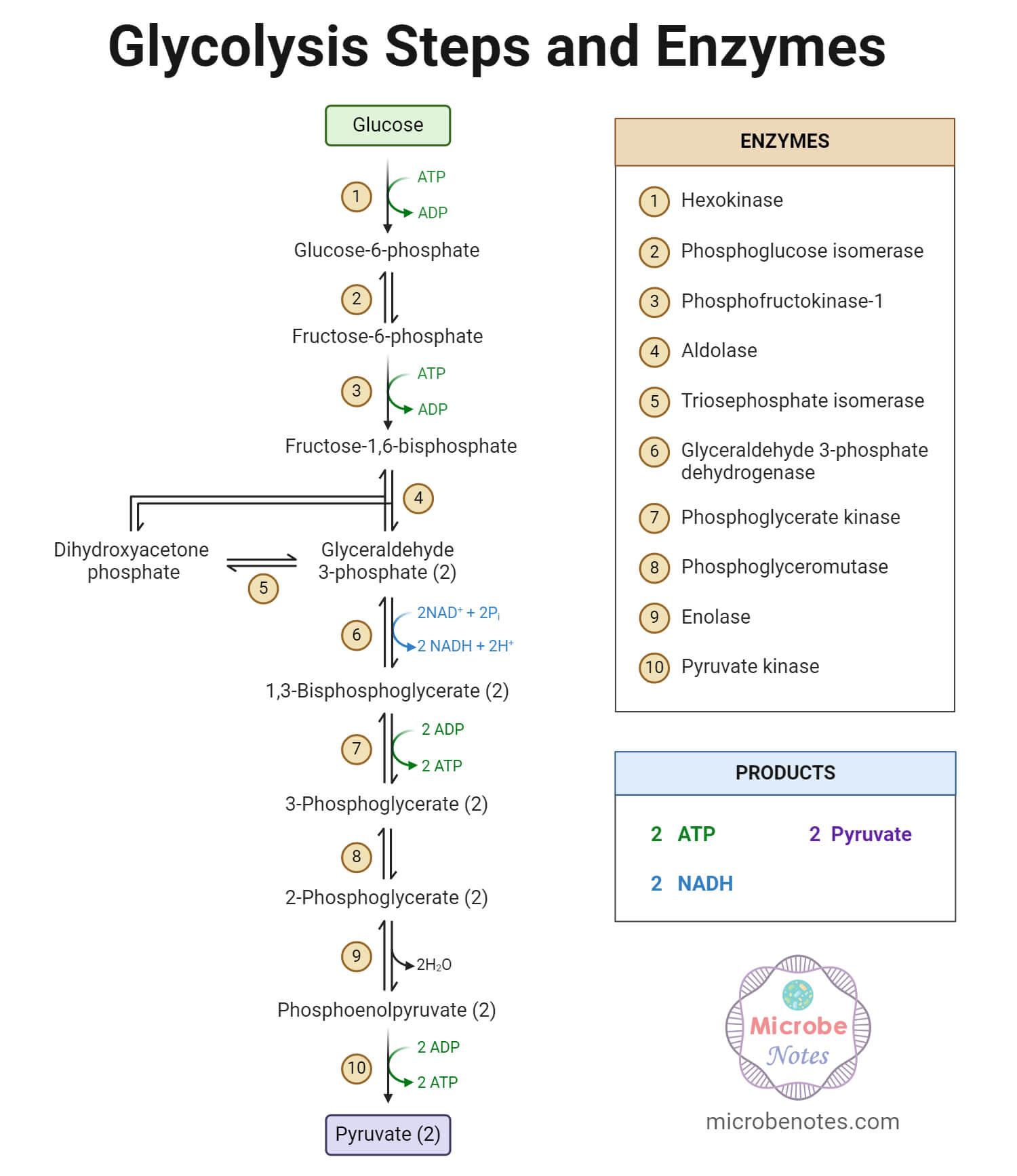
Glycolysis is the central pathway for glucose catabolism in which glucose (6-carbon compound) is converted into pyruvate (3-carbon compound) through a sequence of 10 steps.
- Glycolysis takes place in both aerobic and anaerobic organisms and is the first step toward the metabolism of glucose.
- The glycolytic sequence of reactions differs from one species to the other in the mechanism of its regulation and the subsequent metabolic fate of the pyruvate formed at the end of the process.
- In aerobic organisms, glycolysis is the prelude to the citric acid cycle and the electron transport chain, which together release most of the energy contained in glucose.
- It is also referred to as Embden-Meyerhof-Parnas or EMP pathway in honor of the pioneer workers in the field.
- Glycolysis occurs in the cell’s cytosol (cytoplasm).
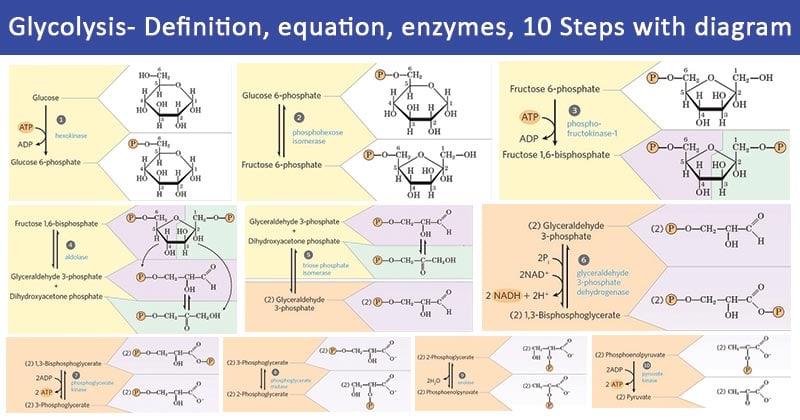
Image Source: Lehninger Principles of Biochemistry.
Glycolysis Equation
A summary of the process of glycolysis cab be written as follows:
C6H12O6 + 2ADP + 2Pi + 2NAD+ → 2C3H4O3 + 2H2O + 2ATP + 2NADH + 2H+
In words, the equation is written as:
Glucose + Adenosine diphosphate + Phosphate + Nicotinamide adenine dinucleotide
↓
Pyruvate + Water + Adenosine triphosphate + Nicotinamide adenine dinucleotide + Hydrogen ions
Glycolysis Enzymes
In most kinds of cells, the enzymes that catalyze glycolytic reactions are present in the cytosol (cytoplasm). One common characteristic in all the enzymes involved in glycolysis is that nearly all of them require Mg2+.
The following are the enzymes that catalyze different steps throughout the process of glycolysis:
- Hexokinase
- Phosphoglucoisomerase
- Phosphofructokinase
- Aldolase
- Phosphotriose isomerase
- Glyceraldehyde 3-phosphate dehydrogenase
- Phosphoglycerate kinase
- Phosphoglycerate mutase
- Enolase
- Pyruvate kinase
Glycolysis 10 Steps
- During glycolysis, a single mole of 6-carbon glucose is broken down into two moles of 3-carbon pyruvate by a sequence of 10 enzyme-catalyzed sequential reactions. These reactions are grouped under 2 phases, phase I and II.
- Stage I comprises “preparatory” reactions which are not redox reactions and do not release energy but instead lead to the production of a critical intermediate of the pathway.
- Stage I consists of the first five steps of the glycolysis process.
- Similarly, in Stage II, redox reactions occur, energy is conserved in the form of ATP, and two molecules of pyruvate are formed.
- The last five reactions of glycolysis constitute phase II.
The ten steps of glycolysis occur in the following sequence:
Step 1- Phosphorylation of glucose
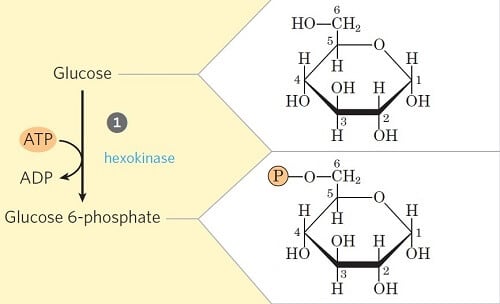
Image Source: Lehninger Principles of Biochemistry.
- In the first step of glycolysis, the glucose is initiated or primed for the subsequent steps by phosphorylation at the C6 carbon.
- The process involves the transfer of phosphate from the ATP to glucose forming Glucose-6-phosphate in the presence of the enzyme hexokinase and glucokinase (in animals and microbes).
- This step is also accompanied by considerable loss of energy as heat.
Step 2- Isomerization of Glucose-6-phosphate
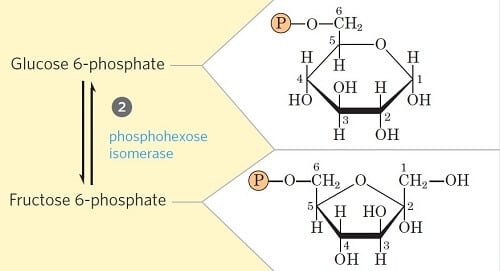
Image Source: Lehninger Principles of Biochemistry.
- Glucose 6-phosphate is reversibly isomerized to fructose 6-phosphate by the enzyme phosphohexoisomerase/phosphoglucoisomerase.
- This reaction involves a shift of the carbonyl oxygen from C1 to C2, thus converting an aldose into a ketose.
Step 3- Phosphorylation of fructose-6-phosphate
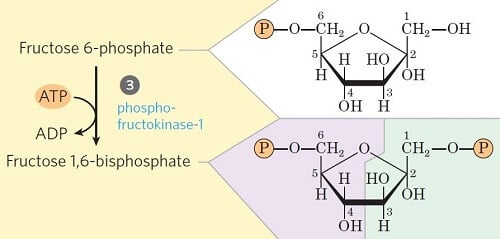
Image Source: Lehninger Principles of Biochemistry.
- This step is the second priming step of glycolysis, where fructose-6-phosphate is converted into fructose-1,6-bisphosphate in the presence of the enzyme phosphofructokinase.
- Like in Step 1, the phosphate is transferred from ATP while some amount of energy is lost in the form of heat as well.
Step 4- Cleavage of fructose 1, 6-diphosphate
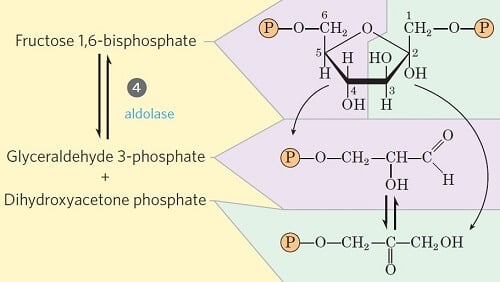
Image Source: Lehninger Principles of Biochemistry.
- This step involves the unique cleavage of the C-C bond in the fructose 1, 6-bisphosphate.
- The enzyme fructose diphosphate aldolase catalyzes the cleavage of fructose 1,6-bisphosphate between C3 and C4 resulting in two different triose phosphates: glyceraldehyde 3-phosphate (an aldose) and dihydroxyacetone phosphate (a ketose).
- The remaining steps in glycolysis involve three-carbon units, rather than six carbon units.
Step 5- Isomerization of dihydroxyacetone phosphate
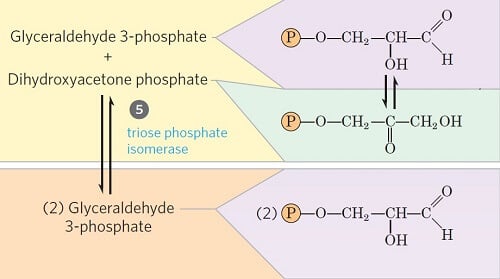
Image Source: Lehninger Principles of Biochemistry.
- Glyceraldehyde 3-phosphate can be readily degraded in the subsequent steps of glycolysis, but dihydroxyacetone phosphate cannot be. Thus, it is isomerized into glyceraldehyde 3-phosphate instead.
- In this step, dihydroxyacetone phosphate is isomerized into glyceraldehyde 3-phosphate in the presence of the enzyme triose phosphate isomerase.
- This reaction completes the first phase of glycolysis.
Step 6- Oxidative Phosphorylation of Glyceraldehyde 3-phosphate
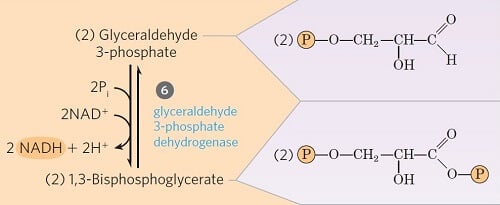
Image Source: Lehninger Principles of Biochemistry.
- Step 6 is one of the three energy-conserving or forming steps of glycolysis.
- The glyceraldehyde 3-phosphate is converted into 1,3-bisphosphoglycerate by the enzyme glyceraldehyde 3-phosphate dehydrogenase (phosphoglyceraldehyde dehydrogenase).
- In this process, NAD+ is reduced to coenzyme NADH by the H– from glyceraldehydes 3-phosphate.
- Since two moles of glyceraldehyde 3-phosphate are formed from one mole of glucose, two NADH are generated in this step.
Step 7- Transfer of phosphate from 1, 3-diphosphoglycerate to ADP
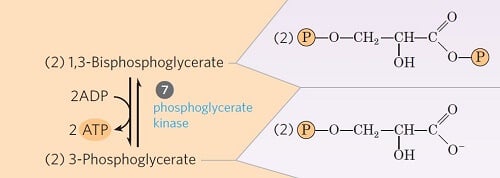
Image Source: Lehninger Principles of Biochemistry.
- This step is the ATP-generating step of glycolysis.
- It involves the transfer of phosphate group from the 1, 3-bisphosphoglycerate to ADP by the enzyme phosphoglycerate kinase, thus producing ATP and 3-phosphoglycerate.
- Since two moles of 1, 3-bisphosphoglycerate are formed from one mole of glucose, two ATPs are generated in this step.
Step 8- Isomerization of 3-phosphoglycerate
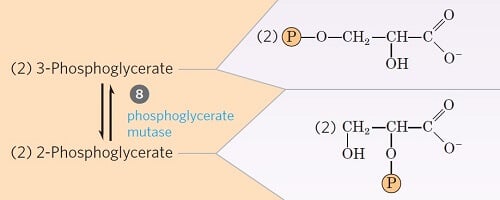
Image Source: Lehninger Principles of Biochemistry.
- The 3-phosphoglycerate is converted into 2-phosphoglycerate due to the shift of phosphoryl group from C3 to C2, by the enzyme phosphoglycerate mutase.
- This is a reversible isomerization reaction.
Step 9- Dehydration 2-phosphoglycerate
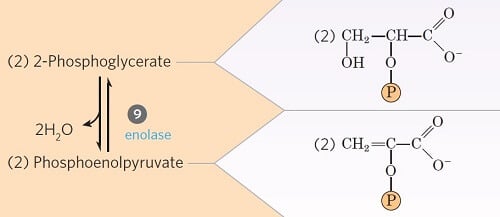
Image Source: Lehninger Principles of Biochemistry.
- In this step, the 2-phosphoglycerate is dehydrated by the action of enolase (phosphopyruvate hydratase) to phosphoenolpyruvate.
- This is also an irreversible reaction where two moles of water are lost.
Step 10- Transfer of phosphate from phosphoenolpyruvate
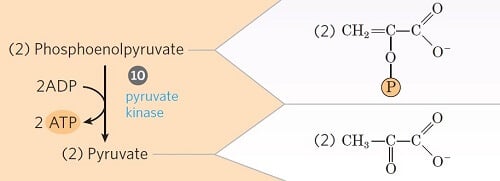
Image Source: Lehninger Principles of Biochemistry.
- This is the second energy-generating step of glycolysis.
- Phosphoenolpyruvate is converted into an enol form of pyruvate by the enzyme pyruvate kinase.
- The enol pyruvate, however, rearranges rapidly and non-enzymatically to yield the keto form of pyruvate (i.e. ketopyruvate). The keto form predominates at pH 7.0.
- The enzyme catalyzes the transfer of a phosphoryl group from phosphoenolpyruvate to ADP, thus forming ATP.
Result of Glycolysis
The overall process of glycolysis results in the following events:
- Glucose is oxidized into pyruvate.
- NAD+ is reduced to NADH.
- ADP is phosphorylated into ATP.
Glycolysis Steps Video
Fates of Pyruvate
Depending on the organism and the metabolic conditions, the pyruvate takes one of the following three essential routes:
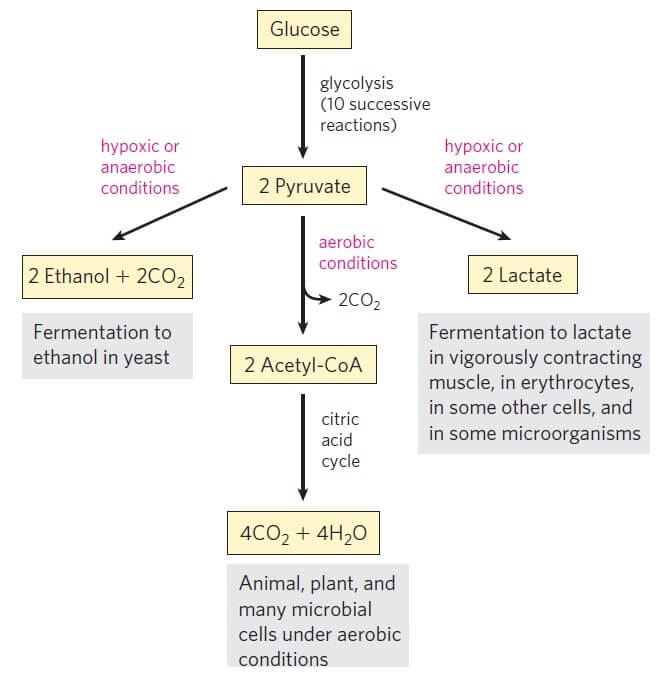
1. Oxidation of pyruvate
- In aerobic organisms, the pyruvate is then moved to the mitochondria where it is oxidized into the acetyl group of acetyl-coenzyme A (acetyl Co-A).
- This process involves the release of one mole of CO2.
- Later, the acetyl CoA is completely oxidized into CO2 and H2O by entering the citric acid cycle.
- This pathway follows glycolysis in aerobic organisms and plants.
2. Lactic acid fermentation
- In conditions where the oxygen is insufficient, like in the skeletal muscle cells, the pyruvate cannot be oxidized due to lack of oxygen.
- Under such conditions, the pyruvate is reduced to lactate by the process of anaerobic glycolysis.
- Lactate production from glucose also occurs in other anaerobic organisms by the process of lactic acid fermentation.
3. Alcoholic Fermentation
- In some microbes like brewer’s yeast, the pyruvate formed from glucose is converted anaerobically into ethanol and CO2.
- This is considered the most ancient form of the metabolism of glucose, as observed in conditions where the oxygen concentration is low.
Significance of Glycolysis Pathway
- All tissues employ the glycolytic pathway for the breakdown of glucose to provide energy in the form of ATP.
- An important pathway for the production of energy, especially under anaerobic conditions.
- It is crucial for the generation of energy in cells without mitochondria.
- It forms products that are intermediates for other metabolic pathways.
- Glycolysis interfaces with glycogen metabolism, the pentose phosphate pathway, the formation of amino sugars, triglyceride synthesis (by means of glycerol 3-phosphate), the production of lactate (a dead-end reaction), and transamination with alanine.
Glycolysis Associated Diseases
Deficiency in any of the glycolytic enzymes leads to hemolytic anemia because RBCs depend on glycolysis for energy production and will lyse if their energy demands are not met as a result of faulty glycolysis.
Glycolysis Frequently Asked Questions (FAQs)
What is aerobic glycolysis?
Aerobic glycolysis is the process of oxidation of glucose into pyruvate followed by the oxidation of pyruvate into CO2 and H2O in the presence of a sufficient amount of oxygen.
What is anaerobic glycolysis?
Anaerobic glycolysis is the process that takes place in the absence of enough oxygen resulting in the reduction of pyruvate into lactate and reoxidation of NADH into NAD+.
Where does glycolysis occur?
Glycolysis occurs in the cell’s cytosol (cytoplasm).
What are the products of glycolysis?
The products of glycolysis are two moles of pyruvate, four moles of ATPs (net gain of 2 ATPs), and one mole of NADH.
How many NADH are produced by glycolysis?
Two moles of NADH are produced by glycolysis.
How many ATPs are formed in glycolysis?
A total of four moles of ATPs are formed in glycolysis. The net gain of ATP in glycolysis is just 2 ATPs as two ATPs are utilized during the preparatory phase of glycolysis.
What are the functions of glycolysis?
The primary function of glycolysis is to produce energy in the form of ATP. Similarly, glycolysis also produces pyruvate, which is then oxidized further to create more ATPs.
References
- Jain JL, Jain S, and Jain N (2005). Fundamentals of Biochemistry. S. Chand and Company.
- Nelson DL and Cox MM. Lehninger Principles of Biochemistry. Fourth Edition.
- Berg JM et al. (2012) Biochemistry. Seventh Edition. W. H Freeman and Company.
- Madigan MT et al. (2012). Brock Biology of Microorganisms. Thirteenth Edition. Pearson Education, Inc.
- https://www.khanacademy.org/science/biology/cellular-respiration-and-fermentation/glycolysis/a/glycolysis

the note nice and well explained but they refuse to be downloaded
thank you so much my assignment is done here
Thank you.
thanks, this note was very helpful in preparation of my exams
Thank you very much for this wonderful presentation of the basic pathways of the glycolysis cycle
Good writing!
Glad to have found this site before semester ends. Learning is a lot easier now.
Same here
It is very useful ☺️
So interesting subject, keep it up with that technology.
But could there be an easier way to remember the steps and the enzymes .applied to each?
Finally my assignment is done????
the pathway you are taking is good one
Learned alot ????????
Nice one, noted.
Is it necessary to remember structures in cycles???
Is it necessary to remember structure in cycle?
I am also microbiologist 1styear student so plz give me help in microbiology interested subject I like it
The notes are very useful indeed. More especially the illustrations.
The note is very good and helpful for expecially undergraduate like us
amazing notes thanks alot
Fabulous!!!????
THANKS SO MUCH, MIND IF WE ASK QUESTIONS? FOR FREE ANSWERS