Fusobacterium nucleatum is a Gram-negative bacterium commonly found in the normal flora of the human oral cavity. It plays a crucial role in the formation of dental plaque.

Although generally commensal, this bacterium is linked to various infections, including periodontal disease and other oral and systemic infections. It is also increasingly associated with conditions like colorectal cancer and adverse pregnancy outcomes.
Taxonomy and Classification of Fusobacterium nucleatum
Domain: Bacteria
Kingdom: Fusobacteriati
Phylum: Fusobacteriota
Class: Fusobacteriia
Order: Fusobacteriales
Family: Fusobacteriaceae
Genus: Fusobacterium
Species: F. nucleatum
F. nucleatum is the principal species of clinical significance within the genus Fusobacterium, being the most frequently studied and commonly isolated in clinical settings. While other species, such as F. necrophorum, F. varium, and F. periodonticum, are also recognized, they are less commonly implicated in human disease. F. nucleatum’s prominence is due to its major role in oral and systemic infections, as well as its association with various pathologies, including periodontal disease and colorectal cancer.
Morphology and Microscopy of Fusobacterium nucleatum
- Gram-negative rod with tapered, pointed ends (spindle-shaped); typically appears in pairs or short chains under the microscope.
- Non-spore-forming
- Non-motile
- Approximately 0.4-0.7µm in width and 3-10µm in length.
- May have a thin capsule in some strains.
They appear as Gram-negative slender rods with pointed ends under the microscope. It is often described as having a “needle-like” or “spindle” appearance.
Cultural and Growth Characteristics of Fusobacterium nucleatum
- Strict anaerobes
- Optimum temperature: 35˚C to 37˚C
- Optimum pH: 7
- Grows mostly on enriched anaerobic media.
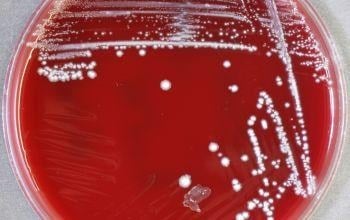
- On Blood Agar: Colonies are 1-2mm, small, round, slightly irregular, translucent, having foul odor, non-hemolytic. It develops distinctive “fried egg” colonies.
- On Brain Heart Infusion (BHI) Agar: Colonies are 1-3 mm, small, circular, irregular or slightly lobate margins, grayish-white to cream colored, smooth to slightly granular bread-crum-like appearance, convex to flat, soft or butyrous, having foul or rancid odor.
- On Thioglycollate Broth: produce diffuse turbidity and sediment at the bottom of the broth.
Source: https://microbe-canvas.com/Bacteria/anaerobic-gram-negative-rods/bbeo-no-growth/fluorescence-yellow/fusobacterium-nucleatum.html
Biochemical and Identification Tests of Fusobacterium nucleatum
| Tests | Results |
| Gram Staining | Negative |
| Catalase | Negative |
| Oxidase | Negative |
| VP (Voges Proskauer) | Negative |
| Indole | Positive |
| Motility | Negative |
| Gas | Positive |
| Gelatin Hydrolysis | Variable |
| Nitrate Reduction | Negative |
| Methylene Blue Reduction | Positive |
| Fermentation of | |
| Arabinose | Negative |
| Glucose | Variable |
| Dnase | Positive |
| Maltose | Negative |
| Mannose | Negative |
| Lactose | Negative |
| Mannitol | Negative |
| Ribose | Negative |
| Fructose | Variable |
| Cellobiose | Negative |
| Sucrose | Negative |
| Glycerol | Negative |
| Sorbitol | Negative |
| Raffinose | Negative |
| Inositol | Negative |
| Enzymatic Reactions | |
| Alkaline Phosphatase | Positive |
| Acid Phosphatase | Positive |
| Phosphatase | Negative |
| Lipase | Positive |
| Lysine Decarboxylase | Negative |
| Esculin Hydrolysis | Negative |
| β-lactamase | Positive |
| β-Glucosidase | Negative |
| Tryptophanase | Positive |
| Casein Hydrolysis | Positive |
| N-Acetyl-glucosaminidase | Negative |
| Lecithinase | Negative |
Pathogenesis and Virulence Factors of Fusobacterium nucleatum
Initial Colonization in the Oral Cavity
- F. nucleatum is generally present in the oral microbiota, which functions as a bridge organism in dental plaque.
- This permits it to merge into complex polymicrobial biofilms.
Biofilm Formation and Plaque Development
- The organisms play an important part in the maturation of dental plaque.
- It increases microbial aggregation via co-adhesion with other bacteria.
- This formed biofilm saves bacteria from host immune responses and antimicrobial agents.
- Within these newly formed biofilms, bacteria survive in an anaerobic environment.
Adhesion and Tissue Attachment
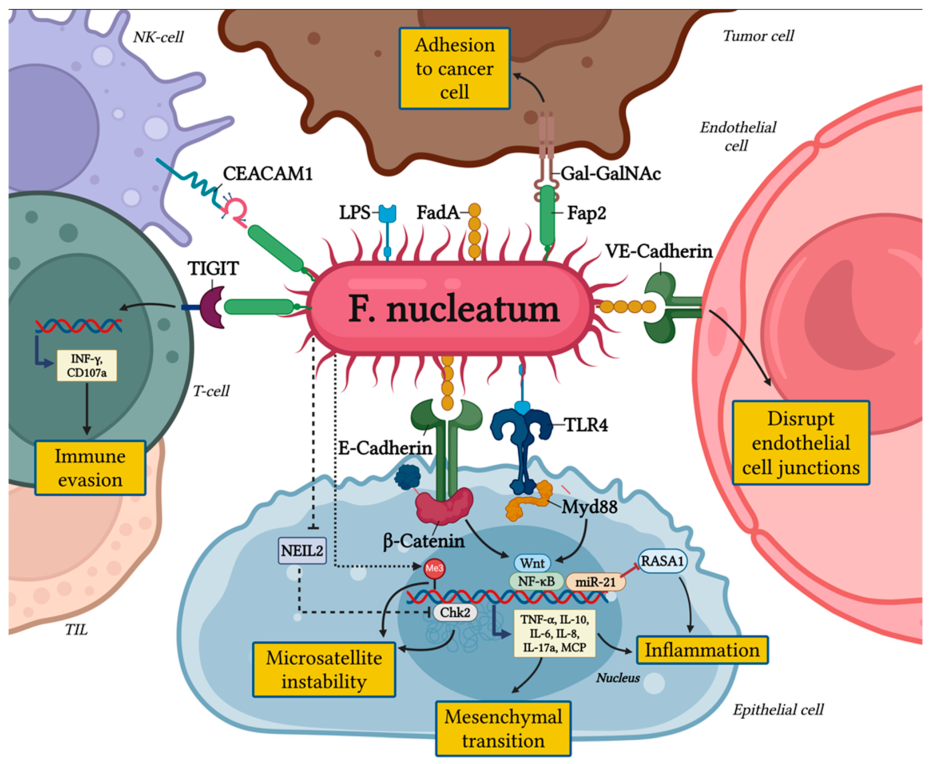
- Bacterium utilizes adhesions like FadA and Fap2 to attach to the host cells.
- FadA adheres to E-cadherin on epithelial cells, promoting strong attachment and cellular entry.
- This process is important for both oral disease and systemic spread.
Invasion of Host Tissues
- After attachment, the bacterium can invade epithelial cells of the gingiva.
- It then perforates deeper tissues, utilizing enzymes and cytoskeletal manipulation of the host cells.
- This can cause inflammation of the gingiva and destruction of tissue.
Induction of Inflammation
- Lipopolysaccharide triggers host immune cells, which activate the release of pro-inflammatory cytokines such as IL-1, IL-6, and TNF-µ.
- It causes swelling of the gingiva, formation of a periodontal pocket, and loss of alveolar bone.
- Chronic inflammation contributes to periodontitis.
Immune Evasion and Persistence
- It then produces factors that repress neutrophil killing and alter macrophage responses.
- The bacterium survives within biofilms and sometimes inside host cells, allowing chronic infection.
Systemic Dissemination
- From periodontal lesions, bacteria can enter the bloodstream.
- The systemic outcomes associated with infection by F. nucleatum include liver and brain abscesses, Lemierre’s syndrome, adverse pregnancy outcomes, and colorectal carcinogenesis.
Source: https://www.mdpi.com/2072-6694/17/3/368
Virulence Factors
-Adhesins
RadD
- It aids in the coaggregation of interspecies and the formation of multispecies biofilm.
- It fights against the human immune system and destroys lymphocytes.
FadA
- It attaches to E-cadherin, facilitating the invasion and inflammation.
Fap2
- It assists in immune evasion and tumor cell interaction.
CmpA
- It contributes further attachment to Streptococcus gordonii.
FomA
- It mediates coaggregation with Porphyromonas gingivalis.
–FAD-I
- It enhances the expression of human β-defensin-2.
-Outer Membrane Lectin
- It promotes interspecific accumulation of Treponema denticulatum and Porphyromonas gingivalis.
-Endotoxins
- It activates a strong inflammatory response through the release of cytokines like IL-1 and TNF-α.
-Proteolytic Enzymes
- It destroys host tissues and proteins, helping in the spread and nutrient acquisition.
-Immune Modulation
- It alters immune responses, assisting in chronic persistence.
-Biofilm Formation
- It increases survival and resistance to antibiotics and host defenses.
Epidemiology and Transmission of Fusobacterium nucleatum
F. nucleatum is worldwide in distribution, found in populations across all geographic regions as a part of the normal flora of the mouth, especially in dental plaque and gingival cervices. The prevalence of F. nucleatum in Asia, especially in Japan, Iran, and China, has been associated with colorectal cancer and other diseases.
Studies employ a case-control process and analysis of tissue samples, recommending a possible role of this bacterium in the pathogenesis of disease. A meta-analysis conducted in the same region revealed an elevated prevalence of this bacterium in patients with colorectal cancer, in contrast to healthy individuals.
Similarly, a study conducted in Brazil showed an elevated concentration of potentially oncogenic bacteria in colorectal cancer, with Fusobacterium being the important genus in cancerous tissues, suggesting notable clinical and molecular characteristics in a Brazilian cohort of colorectal cancer patients. Likewise, another study showed F. nucleatum was responsible for promoting the multiplication of oral squamous cell carcinoma cells via the CDH1/β-catenin pathway, suggesting a new insight into its role in the progression of oral cancer. A cellular model of human immortalized oral epithelial cells with F. nucleatum was confirmed. This study revealed that F. nucleatum assisted the functional reduction of E-cadherin, cell migration, and the up-regulation of Snail family transcriptional repressor1 among cancerous and non-cancerous oral epithelial cells, which is considered an indicator for epithelial-mesenchymal transition.
In addition, a study conducted in China showed that the rate of infection and number of F. nucleatum were higher in carcinoma tissues compared to the adjacent normal tissues of the CRC patients. The bacterium is related to the incidence and development of colon carcinogenesis, since it is spotted in higher numbers in high-grade dysplasia lesions, in comparison to initial neoplastic transformation.
Additionally, no connection has been witnessed with dietary habits, lifestyle, or other conditions of the colon, like ulcerative colitis, even though its existence may provoke ulcerative colitis by developing gut microbiota dysbiosis and dysmetabolism. Transmission of bacteria can occur through various methods, such as:
Endogenous
- It is the most common mode of transmission.
- Infection generally takes place after the disruption of mucosal barriers, such as gum disease, dental procedures, and trauma.
- It then spreads from the oral cavity to adjacent tissues or the bloodstream.
Person- to-Person Transmission
- Bacteria can be transmitted via saliva, such as while kissing, sharing utensils, or toothbrushes.
- It plays a crucial part in the colonization of oral microbiota, especially in close contact.
Vertical Transmission
- Bacteria can spread from mother to infant during birth or through close contact after birth.
- It has been associated with adverse pregnancy outcomes, suggesting possible placental transmission.
Hematogenous Transmission
- Bacteria can enter the bloodstream from oral infections and spread to various organs such as the liver, brain, and lungs.
- This can cause systemic infections and the formation of abscesses.
Clinical Manifestations of Fusobacterium nucleatum
Oral and Dental Infections
-Gingivitis
- Swelling, Redness, and bleeding of gums.
-Periodontitis
- Destruction of the supporting tissues of teeth.
- Formation of the periodontal pocket and mobility of the tooth.
-Dental Plaque Formation
- Formation of biofilm in teeth.
-Periapical Abscesses
- Formation of painful pus around the tooth roots.
Head and Neck Infections
-Lemierre’s Syndrome
Causes painful inflammation and septic thrombophlebitis in the jugular vein.
- Sore throat
- Fatigue
- Headache
- Fever
- Nausea
- Vomiting
- Tonsil infection
- Light sensitivity
- A cough that produces blood and bloody mucus
- Muscle stiffness or soreness
- Swollen lymph nodes in the neck
- Breathing and swallowing trouble
Systemic Infections
-Abscess formation
- Abscesses are formed in the brain, liver, and lungs.
-Bacteremia and Sepsis
- It occurs mainly in immunocompromised persons.
Gastrointestinal Infections
-Colorectal cancer
- It assists in the development of tumors and inflammation.
-It causes appendicitis and intra-abdominal infections.
Pregnancy-related Infection
-Premature Birth
-Stillbirth
-Chorioamnionitis
Complications
Complications of Lemierre’s syndrome consist of the following:
- Septic emboli
- Pleural effusion (presence of fluid around the lungs)
- Arthritis
- Lung abscess
- Pneumonia
- Empyema (formation of pus between the lungs and underneath the chest wall)
Laboratory Diagnosis of Fusobacterium nucleatum
Sample Collection
Specimen depends on the site of infection:
| Site of Infection | Samples |
| Oral and Dental Infections | Dental plaque scraping, gingival swab, periodontal pocket sample, saliva |
| Head and Neck Infections | Throat swab, pus aspirate from neck abscess, tissue biopsy |
| Bloodstream Infection | Blood, serum |
| Systemic Infections | Pus from abscess, tissue biopsy, sterile body fluids |
| Pregnancy-related Infection | Amniotic fluid, placental tissue |
Microscopy
- Gram staining, it appears as Gram-negative, slender rods with pointed or tapered ends under the microscope.
- It is helpful in the preliminary identification of bacteria.
Culture
- Organism is inoculated in suitable media under strict anaerobic conditions and incubated at 37˚C for 24-48 hours.
- On Blood Agar: Colonies are 1-2mm, small, round, slightly irregular, translucent, having foul odor, non-hemolytic. It develops distinctive “fried egg” colonies.
- On Brain Heart Infusion (BHI) Agar: Colonies are1-3 mm, small, circular, irregular or slightly lobate margins, grayish-white to cream colored, smooth to slightly granular bread-crum-like appearance, convex to flat, soft or butyrous, having foul or rancid odor.
- On Thioglycollate Broth: produce diffuse turbidity and sediment at the bottom of the broth.
Biochemical Tests
- After culture, colonies from incubated plates are tested for biochemical tests and identified as F. nucleatum based on the following results:
| Tests | Results |
| Gram Staining | Negative |
| Catalase | Negative |
| Oxidase | Negative |
| VP (Voges Proskauer) | Negative |
| Indole | Positive |
| Motility | Negative |
| Gas | Positive |
| Gelatin Hydrolysis | Variable |
| Nitrate Reduction | Negative |
| Methylene Blue Reduction | Positive |
Serological Test
- It can detect F. nucleatum-specific antibodies in saliva and serum.
- It is generally a non-invasive, inexpensive, and convenient method to detect IgG antibodies.
- In blood, antibodies can remain for several weeks, so this test cannot be used for ongoing infection, but can be helpful when used in combination with other methods.
Molecular Methods
PCR
- It detects bacterial DNA and has high sensitivity and specificity.
16S rRNA gene sequencing
- This method is used for accurate identification and is used especially in research and in other infections.
Fecal Immunochemical Test (FIT)
- This method is rapid, non-invasive, and convenient.
- It is suitable for the screening of CRC as it stably detects bacterial biomarkers.
- It works well in the detection of colonic lesions in symptomatic patients but has restricted diagnostic efficacy.
Fluorescence in situ hybridization
- Fluorescence in situ hybridization and immunohistochemistry are used to find out the location of bacteria in CRC tissues.
- Fluorescence in situ hybridization identifies F. nucleatum, visualizes the interaction between bacteria and tumor cells, and reveals if bacteria are attached to or have invaded cells.
Treatments of Fusobacterium nucleatum Infections
Antibiotic Therapy
-First-line antibiotics
- Metronidazole
- Amoxicillin
-Alternative antibiotics
- Clindamycin: It is the best option for a penicillin-allergic person.
- Amoxicillin-clavulanate: It is the best choice for broad coverage for mixed infections.
- Carbapenems such as meropenem. It is used in severe cases.
- Combination of Cephalosporins and metronidazole for systemic infections.
-Surgical Procedures
In severe cases, surgery is done to:
- Remove an abscess present in the brain, head, neck, and lungs.
- Remove fluid present around the lungs.
- Drain the blood clot formed in the jugular vein.
- Extract your tonsils.
- Remove infected tissue.
-Dental Procedures
- Dental procedures like scaling, root canaling, or the extraction of a tooth are done in periodontal disease.
-Supportive Care
- Non-Steroidal Anti-Inflammatory Drugs are used for the control of pain and fever.
- Proper and timely hydration and nutritional support in severe cases.
- May require hospitalization in cases of sepsis and deep organ abscesses.
-For Lemierre’s Syndrome
- Amoxicillin/Clavulanic acid
- Clindamycin
- Imipenem
- Metronidazole
– For Pregnancy-Related Infections
- Safe antibiotics such as Amoxicillin and Clindamycin are used in pregnant women.
Prevention and Control of Fusobacterium nucleatum Infections
- Regular tooth brushing and daily flossing using antiseptic mouthwashes to remove interdental plaque.
- Routine dental check-ups and scaling and root canaling to remove dental plaque and tartar.
- Avoid direct contact with a person infected with F. nucleatum.
- Ignore food and utensils shared with a sick person.
Conclusion
F. nucleatum is a Gram-negative, obligate anaerobic, spindle-shaped bacterium that is a normal inhabitant of the human oral cavity but can act as an opportunistic pathogen. It plays a crucial role in the formation of dental plaque and periodontal diseases like gingivitis and periodontitis.
It also functions as a bridge organism in biofilm formation and, in some cases, can spread beyond the oral cavity and cause serious systemic infections such as abscesses, Lemierre’s syndrome, and is sometimes related to colorectal cancer and adverse pregnancy outcomes.
Its pathogenicity is mainly due to its ability to attach, invade, form biofilms, and modulate host immune responses. Various antibiotics are used for the treatment. While preventive measures are mainly maintained by regular dental care and proper oral hygiene.
References
- Global Biodiversity Information Facility (GBIF). (n.d.). GBIF species page (ID: 165671650). Retrieved April 10, 2026, from https://www.gbif.org/species/165671650
- Muchová, M. (2020). Characterisation of Fusobacterium nucleatum biofilms and the role of cyclic di-nucleotides (Master’s thesis, University of Birmingham). University of Birmingham eTheses Repository. https://etheses.bham.ac.uk/id/eprint/11013/7/Muchova2020MScbyRes.pdf
- Leibniz Institute DSMZ—German Collection of Microorganisms and Cell Cultures GmbH. (n.d.). BacDive: The bacterial diversity metadatabase—Strain 5765. Retrieved April 10, 2026, from https://bacdive.dsmz.de/strain/5765
- Aryal, S. (2022). Biochemical test of Fusobacterium necrophorum. Microbe Notes. https://microbenotes.com/biochemical-test-of-fusobacterium-necrophorum/
- Groeger, S., Zhou, Y., Ruf, S., & Meyle, J. (2022). Pathogenic mechanisms of Fusobacterium nucleatum on oral epithelial cells. Frontiers in Oral Health, 3, 831607. https://doi.org/10.3389/froh.2022.831607
- Chen, Y., Shi, T., Li, Y., Huang, L., & Yin, D. (2022). Fusobacterium nucleatum: The opportunistic pathogen of periodontal and peri-implant diseases. Frontiers in Microbiology, 13, 860149. https://doi.org/10.3389/fmicb.2022.860149
- Hong, B.-Y., Chhaya, A., Robles, A., Cervantes, J., & Tiwari, S. (2024). The role of Fusobacterium nucleatum in the pathogenesis of colon cancer. Gastrointestinal Endoscopy Clinics of North America, 72(8), 819–827. https://doi.org/10.1177/10815589241277829
https://pubmed.ncbi.nlm.nih.gov/39175147/ - Nayak, S., Shetty, N. D., & Kamath, D. G. (2024). Commensalism of Fusobacterium nucleatum: The dilemma. Journal of Indian Society of Periodontology, 28(4), 427–430. https://doi.org/10.4103/jisp.jisp_286_23
- Yang, X., Zhang, S., Ning, T., & Wu, J. (2025). Fusobacterium nucleatum in health and disease. MedComm, 6(11), e70465. https://doi.org/10.1002/mco2.70465
- Plata, A. F., Sanchez Najera, R. I., Lanas Ortiz, J. U., Chang, S. S., Sifuentes Casas, J. M., Muñuzuri Arana, H. L., Giles Lopez, J. F., Moreno Alvarado, Y., & Solis Soto, J. M. (2025). Fusobacterium nucleatum: Update on epidemiology, clinical manifestations, diagnosis and treatment. International Journal of Applied Dental Sciences, 11(2), 72–76. https://doi.org/10.22271/oral.2025.v11.i2b.2139
- Cleveland Clinic. (2025, August 15). Lemierre’s syndrome: Causes, symptoms & treatment. https://my.clevelandclinic.org/health/diseases/lemierres-syndrome
- He, X., Zhao, Q., Zhang, J., Shi, J., Wan, N., Tang, B., Tian, B., & Li, P. (2025). Potential and application of Fusobacterium nucleatum in the diagnosis and treatment of colorectal cancer. Frontiers in Microbiology, 16, 1652702. https://doi.org/10.3389/fmicb.2025.1652702
