Aspergillus flavus is a prevalent fungus that causes human diseases and is known for producing mycotoxins, particularly aflatoxins.
- It is a saprophytic soil fungus that infects and contaminates seed crops both preharvest and postharvest. Over 100 species of Aspergillus have been found, with Aspergillus fumigatus and Aspergillus niger as the primary pathogenic agents, followed by Aspergillus flavus.
- They also induce opportunistic infections, such as aspergillosis, in immunocompromised humans and animals.
- They infect the plants before and after harvesting when they are in storage rooms. In the pre-harvesting stage, the infection remains dormant until harvest time when it starts to cause yellowing in the infected parts of the plants.
- They are also known to produce mycotoxins which can cause poisoning in humans and animals.
- They also cause opportunistic infections such as aspergillosis in immunocompromised humans and animals.
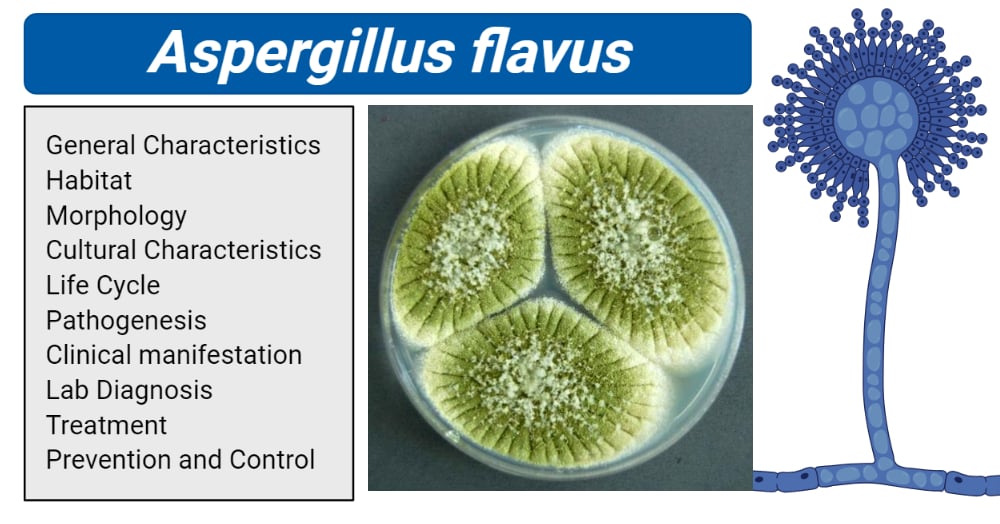
Taxonomy and Classification of Aspergillus flavus
Domain: Eukaryota
Kingdom: Fungi
Phylum: Ascomycota
Subphylum: Pezizomycotina
Class: Eurotiomycetes
Order: Eurotiales
Family: Trichocomaceae
Genus: Aspergillus
Species: flavus
Habitat of Aspergillus flavus
- It is widespread globally and commonly found in soil.
- The fungus is present in the soil as conidia or sclerotia and in plant tissues as mycelia
- They are hosted by cereal grains, legumes, and tree nuts.
- They are thermotolerant fungi hence can survive in a wide range of surfaces than other fungi.
- They thrive in high moisture (hot and humid) environments
- They grow in a minimum temperature of 12 °C (54 °F) and a maximum temperature of 48 °C (118 °F), and an optimal growth temperature is 37 °C (98.6 °F).
- The optimum moisture level for the growth of Aspergillus flavus is 14%, however, these levels vary depending on the crops. Such as, for starchy cereals, they grow at moisture levels of 13-13.2% while for soybeans, at 11.5-11.9%.
- Sclerotia survive in the soil under severe environmental conditions and produce conidia and possibly ascospores based on recent data, leading to a population increase under hot and drought weather conditions.
Morphology of Aspergillus flavus
- The Aspergillus flavus group of fungi has a complex morphology that is classified based on the formation of sclerotia. Group I has L-strains whose sclerotia are greater than 400 μm in diameter and Group II has S strains with sclerotia less than 400 μm in diameter.
- They also have both sexual and asexual forms of reproduction.
- Asexual reproduction produces conidia spore and sclerotia while sexual reproduction produces sclerotia.
- Aspergillus flavus produces asexual spores known as conidia.
- The conidial spores are produced from the phialides on the conidiophore vesicles.
- The conidia spores have a thick mycelial mat that can be seen with unaided eyes and have a size of 3 to 6 µm.
- The conidiophore originates from the hyphal threads.
- The conidiophores are colorless and rough-textured.
- The phialides originating from the conidiophore are both uniseriate and biseriate
- Hyphae occur as thread-like septate branches that form mycelium.
- They branched hyphae have hyaline in each septae
- The hyphae are very tiny and can not be seen with naked eyes.
- Aspergillus flavus can also reproduce sexually producing ascospores contained within the sclerotia.
- Sexual reproduction occurs between two compatible strains with different vegetative forms that are cultured together.
Cultural Growth and Characteristics of Aspergillus flavus
- Under laboratory conditions, A. flavus exhibits rapid growth and distinctive cultural morphology. When grown on common fungal media such as Potato Dextrose Agar (PDA), Czapek Yeast Extract Agar (CYA), and Malt Extract Agar (MEA), colonies typically mature within 5–7 days at temperatures between 25–30 °C.
- The colony diameter usually reaches 3–7 cm within seven days, depending on environmental conditions and medium composition
- The surface of the colony appears yellow-green due to abundant conidial production. The texture is generally velvety or powdery.
- The reverse side of the colony may appear cream, golden, or tan.
- The fungus demonstrates considerable environmental adaptability. It can grow within a temperature range of approximately 12–48 °C, with optimal growth occurring between 28–35 °C.
- It tolerates a broad pH range (approximately pH 2–11) and can grow at relatively low water activity levels (around 0.78), which explains its frequent occurrence in stored dry commodities.
- In conclusion, Aspergillus flavus is a rapidly growing mold with distinct cultural and microscopic features. Its ability to adapt to diverse environmental conditions and produce aflatoxins makes it a significant organism in food safety, agriculture, and medical microbiology.
The life cycle of Aspergillus flavus
- Aspergillus flavus lives through the winter season in soil, appearing as propagules on decaying matter as mycelia or a thick hard mass of mycelia know as sclerotia.
- The sclerotia germinate producing hyphae and asexual spores known as conidia.
- The conidia disperse into the air and the environment via insect (bugs)and wind type of pollinations.
- When the conidia land on grains and legumes, they infect them, through the silks of corn into the corn kernel.
- They grow producing conidiophore and conidia from the sclerotial surface.
- Some conidia may land on the surface of leaves which have been fed on by insects causing damage to the leaves, this is known as secondary inoculation.
- While, some spores may land in the soil via dispersing by rainwater, which then infects the oily plants such as peanuts and cotton seeds.
Identification Tests of Aspergillus flavus
- The principal method for identification is the Lactophenol Cotton Blue (LPCB) mount, which shows septate hyaline hyphae, rough conidiophores, uniseriate or biseriate phialides, and rough globose conidia after the colony observation.
- Toxin production can be tested using Coconut Agar Medium under UV light and verified via TLC, HPLC, or ELISA.
- PCR can also be used to detect various genes which can be produced for the species.
Virulence factors of Aspergillus flavus
The various virulent factors that cause pathogenesis in humans are:
Aflatoxin
- Aflatoxins, especially aflatoxin B₁, are secondary metabolites that are hepatotoxic, immunosuppressive, and carcinogenic.
- These toxins contribute indirectly to virulence by harming host tissues, decreasing immune response, and increasing colonization in the host cell.
Hydrolytic enzymes
- Proteases, lipases, and cellulases secreted by A. flavus break down elements of host tissue.
- These enzymes make it possible for the fungus to enter host tissues and use nutrients, which makes it easier for both plants and animals to become infected.
Adhesion
- A. flavus can adhere to surfaces, which protects the septate hyphae from any antifungal agents consumed by hosts.
- This process i.e., adhesion is crucial for the colonization and establishment of infection in the host.
Adaptation of Aspergillus flavus in the environment
- The ability of A. flavus to grow at a wide temperature (12-48 °C) and the tolerance of low water activity, enables the survival at wide range of hosts.
- This thermotolerance allows the organism to invade in the tissue of the human hosts such as lungs or alveolar macrophages.
Regulatory genes
- The various genes such as aflR play a major role in virulence. This causes aflatoxin biosynthesis, stress response which coordinates in toxin production.
Pathogenesis of Aspergillus flavus
The pathogenesis of Aspergillus flavus begins when airborne conidiospores are inhaled and deposited in the alveoli of the lungs. In healthy individuals, these type of airborne conidiospores are usually phagocytosed and cleared by macrophages and neutrophils present in the alveoli. However, in immunocompromised host for instance, those hosts with neutropenia, the spores can invade the alveoli suppressing the immune system and germinating into septate hyphae in the site of invasion.
The hyphal form is invasive and capable of penetrating lungs tissue by secreting enzymes like proteases, lipases and cellulases. As the infection progresses the hyphae invades nearby the blood vessel and hematogenous dissemination to other organs like kidney, brain and heart.
Additionally, Aspergillus flavus produces their toxins like aflatoxins which suppresses immune response by inhibiting phagocytosis, inducing apoptosis of immune cells.
Epidemiology of Aspergillus flavus
Aspergillus flavus is an opportunistic pathogen that causes invasive pulmonary infections, keratitis, and sinusitis, particularly in immunocompromised people. Fungal proliferation and toxin production are increased by environmental stressors such as drought, high temperatures, and unfavorable storage conditions, which makes them a problem for both agriculture and public health.
The most common species causing invasive sinusitis in immunocompetent patients in the area of Sudan and in the Middle East. It is the most commonly reported food-borne fungus; many of the isolates produce aflatoxins. Aflatoxins constitute a group of highly potent carcinogens and hepatotoxins that are formed in stored grains, corn, and legumes that are subsequently ingested by humans and farm animals. There are various clinical manifestations of Aspergillus flavus infection that have been reported worldwide.
Transmission of Aspergillus flavus
- Conidiospores are spread by wind, dust, and mechanical crop handling, which can result in inhalation exposure.
- When consumed, contaminated crops like tree nuts, cottonseed, peanuts, and maize can spread the fungus or its aflatoxins.
- Workers who handle animal feed, stored grains, or decomposing vegetation run a greater risk of exposure through inhalation and contact.
- Immunocompromised people are susceptible to opportunistic infections, which typically start with spore inhalation and result in sinusitis, keratitis, or invasive pulmonary infections.
Clinical Manifestations of Aspergillus flavus
In immunocompromised host, Aspergillus flavus may primarily affect the lungs, paranasal sinus, ear, eye, skin and multiple systemic sites. A. flavus can show following manifestations:
- Invasive aspergillosis
It is an infection that spreads quickly in patients with compromised immune systems. The illness is primarily lethal. Aspergillus flavus organisms in immunocompromised hosts cause a disseminated illness that results in abscesses in the viscera, including the liver, spleen, kidney, soft tissues, and bone, as well as endocarditis and endophthalmitis.
- Invasive Sino nasal aspergillosis
It frequently affects diabetic or immunocompromising people. Nasal congestion, swelling, proptosis are symptoms.
- Allergic bronchopulmonary aspergillosis
Wheezing, cough, mucus plugging, respiratory infections are main symptoms. The sensitization is specifically caused due to A. flavus which occurs in asthma or cystic fibrosis patients.
- Keratitis
It often occurs due to ocular trauma, eye surgery or contact lens use. The symptoms include eye pain, redness, blurred vision, photophobia, corneal ulceration, etc.
Human Aspect of Aspergillus flavus Infection
- The conidial spores of Aspergillus flavus bind to the lung cell basal lamina which leads to the development of invasive aspergillosis, enhanced by various proteins including fibronectin, laminin, type IV collagen, fibrinogen, complement, albumin, and surfactant proteins.
- Aspergillus flavus is majorly attracted to the fibrinogen proteins which allow adhesiveness to the basal lamina.
Aspergillosis
- Aspergillus flavus is the second leading cause of invasive and noninvasive aspergillosis and it is the most common cause of superficial infection.
- The clinical features of aspergillosis include allergic (extrinsic asthma, extrinsic allergic alveolitis, and allergic bronchopulmonary aspergillosis), pulmonary extrapulmonary colonization, and invasive infection (pulmonary and extrapulmonary).
- Other symptoms may include chronic granulomatous sinusitis, keratitis, cutaneous aspergillosis, wound infections, and osteomyelitis following trauma and fungal inoculation.
- Fungal sinusitis occurs due to deposition of the large spores in the upper respiratory tracts.
- Secondary transmission of fungal spores arise from infection via wounds and smoking contaminated plant material such as tobacco or marijuana.
- Nosocomial infections can occur during surgery, such as transplant patients.
Aflatoxicosis
- Toxicity is enhanced by the ingestion of the aflatoxins produced by the fungi.
- The sclerotia forms of the fungi are known to produce aflatoxins, commonly known as B1 and B2
- The S strain produces aflatoxin G1 and G2 which are not commonly produced by A.flavus.
- The L strain is more aggressive than S strain however it produces fewer aflatoxins, fewer sclerotia, with more acidity.
- The aflatoxins are carcinogenic and they cause aflatoxicosis
- Aflatoxicosis is associated with:
- vomiting, abdominal cramping, and pain, pulmonary edema, hemorrhaging, disruption of food digestion, poor abdominal absorption and metabolism, severe progressive effects may cause liver damage, liver cancer, mental impairment, coma, and death.
Carcinogenic effects of aflatoxins
- These are cancers arising from long-term exposure to aflatoxin B1. B1 is a potential hepatocarcinogen which induces tumors mainly in the liver, but also the kidney, lungs, and colon in humans and animals.
- Hepatocellular carcinoma (HCC), primary liver cancer, is associated with aflatoxin B1 consumption.
- Individuals with increased risks of developing aflatoxin associated Hepatocellular carcinoma (HCC) have chronic Hepatitis B and C, which synergically interacts with aflatoxin B1 increasing the development of HCC.
Plant pathologies
- Colonization of plants by Aspergillus flavus is enhanced by the mode of dispersion and damage by plant and insect eaters.
- The insects and plants provide a point of entry of the fungi into the plants while the insects and blowing wind, allows the spores to land on the damaged surfaces of these damaged plants and start to grow dormant until the plants are harvested and stored.
- During storage, the fungi start to germinate and spread within the crop and the surrounding crops.
- Colonization of plants by Aspergillus flavus forms powdery masses of yellowish-green spores on the upper surface and reddish-gold on the lower surface.
- In both grains and legumes, infection is minimized to small areas, and discoloration and dullness of affected areas are often seen. Growth is rapid and colonies appear downy or powdery in texture.
Aspergillus Ear Rot
- It is caused by aflatoxins produced by Aspergillus flavus.
- It is a powdery olive-green (yellow-green) mold that grows on the ears of corn and then turns brown as the masses age.
- Higher aflatoxin levels are associated with discolored, shriveled kernels that are often found near the tip of the ear.
- This infection is favored by hot dry conditions during pollination and during grain fill.
- Yellow-brown silks are most susceptible to infection.
- Spores landing on the silks germinate, rapidly grow down the silk and colonize the surface of the developing kernels.
- When the plant is maturing when the moisture levels drop, the fungi start to colonize the internal tissues and continue to grow until the moisture level decrease to <15%.
Laboratory diagnosis of Aspergillus flavus
To properly identify Aspergillus flavus, a combination of microscopy, culture, and molecular or biochemical techniques are used in the laboratory.
Specimen collection
Specimen is collected according to the site of infection. For instance- Sputum, Bronchoalveolar lavage (BAL), skin tissue, corneal scrapings, biopsy specimen, wound scrapings, etc.
Direct Microscopy
Direct KOH (10-20%) preparation of specimen shows non-pigmented, septate hyphae with characteristic dichotomous branching at an angle of 45 degree where biopsy specimens show septate, branching hyphae invading tissues. The vesicles are about 800-1200um in diameter, producing phialides. the phialides have uniseriate or biseriate or combined. The conidia are 250-450um with thin rough walls.
Culture and Colony observation
- Sabouraud Dextrose Agar– In the early days of growth (24-48 hours), the colonies are white with a soft velvety surface. After 4 days of growth, the colonies become raised and floccose at the center. On sporulation, the colonies appear yellowish-green because of the conidia color. Sclerotia are produced appearing white initially then they turn brown after 6 days of growth. Colonies are 55-70mmn in diameter
- Potato Dextrose Agar– they produce green conidia, with a dominated colony appearance. they are plain and flat at the edges and raised at the center and wrinkled cerebriform pattern. They also produce exudates that are colorless or brown. The sclerotia which are the compact mass of hardened fungal mycelia and deep brown in color. The colonies are encircled by a white border and a pale inner side.
- Malt extract agar- white Colonies vary in shape and size. The initial formation of smooth white mycelia which grows and produces olive and dark green conidia. Sclerotia are white and deep brown with colorless exudates at the center of the colonies.
- Czapek yeast agar– The mycelia are white, flat with large raised tufted wool of white mycelia. The colonies appear dry and exudated but no sclerotia are produced in CZA. No pigmentation observed do the colonies are uncolored. Some isolates produce velutinous, grey-blue-green, and uniseriate conidial heads.
Lactophenol cotton blue (LPCB) mount method
LPCB staining is the primary technique for microscopic identification following colony growth. It exhibits the typical characteristics of A. flavus, such as septate hyphae, rough pattern of conidiophores, smooth wall of conidiophores, uniseriate and biseriate phialides.
Toxin detection tests
Chromatographic techniques like Thin Layer Chromatography (TLC) or HPLC, which help identify toxic strains, can be used to assess the production of aflatoxin in some isolates.
Molecular tests
For accurate species identification and separation from closely related species, molecular methods like PCR amplification and sequencing of ITS regions or β-tubulin genes are used.
Treatments
The most common use of amphotericin B. Nevertheless, the treatment’s results are unsatisfactory. An alternative medication administered to patients is Capsofungin.
The best way to treat aspergilloma is to surgically remove the fungus ball from the cavity.
Antimicrobial Resistance
Aspergillus flavus is resistant to Amphotericin B, Itraconazole, Voriconazole. However, the frequency of resistance is rare.
Prevention and Control of Aspergillus flavus
- To prevent Aspergillus flavus from growing and reduce the possibility of aflatoxin generation, grains, nuts, and cereals should be stored in cold, dry, and well-ventilated settings.
- Aspergillus flavus contamination of crops can be reduced by using excellent agricultural techniques, such as crop rotation, timely harvesting, and adequate field cleanliness.
- To reduce fungal spore inhalation, workers handling stored grains and agricultural commodities should wear protective masks and make sure there is enough ventilation.
- Maintaining efficient air filtration systems and stringent hygiene protocols in hospital settings helps lower exposure to airborne Aspergillus spores, especially in immunocompromised patients.
Conclusion
Aspergillus flavus is a widespread environmental fungus that can be found in soil, decomposing plants, and agricultural items that have been kept, like grains and nuts. It can infect humans by infected wounds or spore inhalation, it primarily affects immunocompromised people and causes diseases like invasive aspergillosis, keratitis, and sinusitis. A. flavus is known to produce aflatoxins, which are dangerous mycotoxins that contaminate food and pose major health hazards, in addition to causing illnesses. To lower morbidity, early discovery and appropriate treatment are crucial, particularly in areas with warm climates where the fungus is common. Controlling the spread and effects of Aspergillus flavus requires preventive measures such appropriate food storage, routine crop monitoring for aflatoxin contamination, and upholding hygienic ambient conditions.
References
- Aspergillus flavus. (2026). In Wikipedia. https://en.wikipedia.org/w/index.php?title=Aspergillus_flavus&oldid=1340057830
- Barton, R. C. (2013). Laboratory Diagnosis of Invasive Aspergillosis: From Diagnosis to Prediction of Outcome. Scientifica, 2013, 459405. https://doi.org/10.1155/2013/459405
- Bertin-Biasutto, L., Paccoud, O., Garcia-Hermoso, D., Denis, B., Boukris-Sitbon, K., Lortholary, O., Bretagne, S., Gits-Muselli, M., Herbrecht, R., Letscher-Bru, V., Danion, F., Cassaing, S., Morio, F., Nourrisson, C., Pihet, M., Sasso, M., Desoubeaux, G., Durieux, M.-F., Bonhomme, J., … Lanternier, F. (n.d.). Features of Invasive Aspergillosis Caused by Aspergillus flavus, France, 2012–2018—Volume 31, Number 5—May 2025—Emerging Infectious Diseases journal—CDC. https://doi.org/10.3201/eid3105.241392
- Cotty, P. J. (1989). Virulence and cultural characteristics of two Aspergillus flavus strains pathogenic on cotton. Phytopathology, 79(7), 808–814.
- Leema, G., Kaliamurthy, J., Geraldine, P., & Thomas, P. A. (2010). Keratitis due to Aspergillus flavus: Clinical profile, molecular identification of fungal strains and detection of aflatoxin production. Molecular Vision, 16, 843–854.
- Morphology of Aspergillus flavus. (n.d.). Retrieved March 2, 2026, from https://encyclopedia.pub/entry/10330
- Parija, S. C. (2014). Textbook of Microbiology & Immunology (2nd ed). Elsevier Health Sciences APAC.
- (PDF) Ecology, development and gene regulation in Aspergillus flavus. (n.d.). ResearchGate. Retrieved March 2, 2026, from https://www.researchgate.net/publication/43289878_Ecology_development_and_gene_regulation_in_Aspergillus_flavus
- Suarez, O. J., Ruiz-Ayala, D. C., Contreras, L. R., Forero, M. G., Medrano-Hermosillo, J. A., & Rodriguez-Mata, A. E. (2025). Morphological Characterization of Aspergillus flavus in Culture Media Using Digital Image Processing and Radiomic Analysis Under UV Radiation. Agriculture, 15(17). https://doi.org/10.3390/agriculture15171888
- Walsh, T. J., Hayden, R. T., & Larone, D. H. (2018). Larone’s Medically Important Fungi: A Guide to Identification. ASM Press. https://doi.org/10.1128/9781555819880
- Alessandro C. P, 2009: Differences in pathogenicity and clinical syndromes due to Aspergillus fumigatus and Aspergillus flavus: Medical Mycology Review. Pages S261–S270
- Klich MA, 2007: Aspergillus flavus: the major producer of aflatoxin. Molecular Plant Pathology: 10.1111/j.1364-3703.2007.00436.x.
- Raymond J., Steven E, Screen, Bijan Shams-Pirzadeh. 2000: Lack of Host specialization in Aspergillus flavus: Applied and Environmental Microbiology. 10.1128/aem.66.1.320-324.2000
- Fausto A., Marcio L. R., Carolina C., 2019: The Still Underestimated Problem of Fungal Disease worldwide: Frontier of Microbiology: 2019/oo214
- McClenny N., Laboratory Detection and Identification of Aspergillus species by Microscopic observation and culture: The Traditional Approach. Medical Mycology. Volume 43, Pages S125–S128
- https://www.hindawi.com/journals/ijmicro/2017/5273893/
- https://www.microbiologyresearch.org/content/journal/micro/10.1099/mic.0.2007/007641-0?crawler=true
- https://crops.extension.iastate.edu/cropnews/2012/08/aspergillus-ear-rot-and-aflatoxin-production
- https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/aspergillus-flavus
- https://wiki.bugwood.org/Aspergillus_flavus
- http://www.biologydiscussion.com/fungi/aspergillus-habitat-reproduction-and-importance-ascomycotina/24000
- https://www.bustmold.com/resources/mold-library/aspergillus-flavus/
- http://agris.fao.org/agris-search/search.do?recordID=QY870002588
