The yellow fever virus is a mosquito-borne virus that belongs to the genus Flavivirus within the family Flaviviridae. This virus is primarily transmitted to humans through the bite of infected mosquitoes, especially Aedes aegypti.

This virus is endemic in tropical regions of Africa and South America. The virus is named “yellow fever” due to the yellowing of the skin and eyes, which may appear severe. Symptoms may range from mild fever with headache to severe life-threatening jaundice, hemorrhage, and multiple organ failure. Prevention of the yellow fever virus includes control of the vector and vaccination.
Taxonomy and Classification of Yellow Fever Virus
Kingdom: Orthornavirae
Phylum: Kitrinoviricota
Class: Flaviviricetes
Order: Amarillovirales
Family: Flaviviridae
Genus: Flavivirus
Species: Yellow Fever Virus
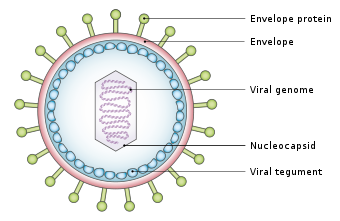
Structure and Morphology of Yellow Fever Virus
- Small, spherical virus with a diameter of approximately 50-60 nm.
- Enveloped virus.
- Shows icosahedral symmetry.
- Consists of a single-stranded, positive-sense RNA genome.
Source: https://study.com/academy/lesson/yellow-fever-virus-structure-and-function.html
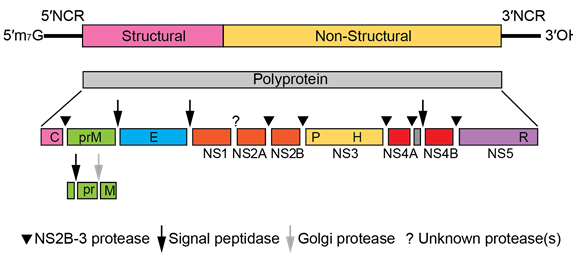
Genome Organization and Proteins of Yellow Fever Virus
Source: https://www.creative-diagnostics.com/tag-yellow-fever-virus-antigens-86.html
-The genome consists of single-stranded, positive-sense RNA, which is approximately 11 kb in length.
-The genome consists of the following regions:
- A 5’ untranslated region (5’ UTR)
- A 3’ untranslated region (3’ UTR)
- A single long open reading frame (ORF)
-5’ and 3’ Untranslated Region (UTR)
5’ UTR
- It possesses a methylated cap.
- It is essential for the initiation of translation and the stability of RNA.
3’ UTR
- Lacks poly A tail.
- Consists of secondary structures that are important for the replication of viral RNA and interaction with host proteins.
-Single Open Reading Frame (ORF)
- It is produced as a polyprotein, which is then processed by proteolytic cleavage.
Protein Organization
- The virus codes for structural proteins (C, M, and E) in the first quarter of its genome. These proteins form the virus particle. The rest of the genome codes for non-structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, NS5) that help the virus make more copies of itself.
Proteins
Structural Proteins
-Capsid (C) Protein
- Forms the nucleocapsid, which surrounds the viral RNA.
- Aids in the protection and packaging of the viral genome.
-Membrane (M) Protein
- Essential for the maturation and stability of the virus.
-Envelope (E)
- It is the major surface glycoprotein responsible for the attachment to host cell receptors and fusion of membranes during viral entry.
Non-Structural Proteins
-NS1
- Involved in the replication of viral RNA.
- Its secreted form assists in immune evasion.
-NS2A
- It is essential for RNA replication and virus assembly.
- Aids in the regulation of the production of viral protein.
-NS2B
- Functions as a cofactor for NS3 protease.
- Important for the processing of polyprotein.
-NS3
- Essential for the replication of the virus.
-NS4A
- Takes part in the rearrangement of the membrane and assists in the formation of the replication complex.
-NS4B
- Plays a crucial role in immune evasion and helps in viral replication.
-NS5
- Necessary for genome replication and transcription.
- It has RNA-dependent RNA polymerase activity and methyltransferase activity.
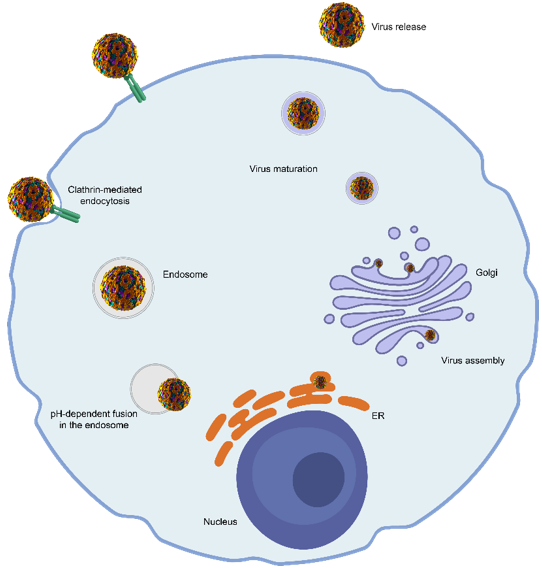
Replication Cycle of Yellow Fever Virus
Source: https://antiviral.creative-diagnostics.com/yellow-fever-virus-antiviral-services.html
Attachment and Entry
- The E protein adheres to the specific host cell receptors on the target cells.
- Then the virus enters the host cell by clathrin-mediated endocytosis, forming an endocytic vesicle.
Fusion and Uncoating
- The acidic environment of the endosome triggers the fusion of the viral envelope with the endosomal membrane.
- After the fusion, the nucleocapsid is liberated into the cytoplasm, releasing the viral RNA genome.
Translation
- After that, the positive-sense single-stranded RNA genome functions as mRNA, which is then translated into a single polyprotein by host ribosomes.
- Then, the polyprotein is co- and post-translationally cleaved by host and viral proteases into structural and non-structural proteins.
Formation of Replication Complex
- Assembly of non-structural proteins takes place in the endoplasmic reticulum, leading to the formation of the replication complex.
- This newly formed replication complex then synthesizes the negative-sense RNA intermediate and new positive-sense RNA genome.
Assembly
- The newly synthesized positive-sense RNA is then packaged with the C protein, forming nucleocapsids.
- The nucleocapsids then bud into the lumen of the endoplasmic reticulum, obtaining prM and E proteins, forming an immature virus.
Maturation
- The newly formed immature virus is then transported to the Golgi apparatus, where prM is converted to M protein via furin, making the virus infectious.
Release
- After that, the mature virus is transferred to secretory vesicles and freed from the cell through exocytosis.
Pathogenesis and Host Immune Response of Yellow Fever Virus
Entry and Early Infection
- The Yellow Fever Virus is transmitted through a mosquito bite.
- Initially, the virus infects macrophages and dendritic cells in the skin and lymph nodes.
- The virus then replicates locally, which then enters regional lymph nodes, causing primary viremia.
Systemic Spread
- Via the bloodstream, the yellow fever virus spreads to the target organs:
- Liver: The primary site of yellow fever virus replication is the hepatocyte. Viral replication in these cells leads to cell swelling (ballooning degeneration), apoptosis, and necrosis, contributing to the characteristic liver damage and jaundice observed in severe cases.
- Kidneys: Yellow fever virus can cause acute kidney injury (AKI) through both direct and indirect mechanisms. Directly, the virus infects renal tubular epithelial cells, leading to cell damage and tubular necrosis. Indirectly, systemic factors such as hypotension, dehydration, and a robust inflammatory response contribute to reduced renal perfusion and further tubular injury. These processes result in tubular necrosis and interstitial inflammation, which are key features of yellow fever-associated renal involvement.
- Heart: myocardial involvement is due to direct infection of cardiac myocytes and systemic effects of cytokines and hypoperfusion.
- Spleen and Lymphoid Tissues: the yellow fever virus infects dendritic cells and macrophages present in lymphoid tissues, which leads to depletion of lymphoid cells and necrosis of the white pulp.
- Cardiovascular System: endothelial cells of blood vessels can be destroyed by direct viral effects or via cytokine-induced inflammation, which leads to increased vascular permeability and hemorrhage.
- Gastrointestinal Tract: gastric and intestinal hemorrhage is common in severe yellow fever, which is caused by endothelial damage, coagulopathy, and thrombocytopenia.
The severity is dependent on viral load, host immunity, and age.
Host Immune Response Due to Yellow Fever Virus
Innate Immunity
- Natural Killer cells, macrophages, and dendritic cells identify viral RNA through pattern recognition receptors such as TLR7 and RIG-I.
- This activates type I interferons (IFN-α/β) and pro-inflammatory cytokines (IL-6, TNF-α) production.
- The early innate response aids in the restriction of viral replication; however, it can cause systemic inflammation.
Adaptive Immunity
-Humoral Response (B cells)
- Neutralizing antibodies are important for infection control.
- Anti-E protein antibodies hinder the binding and fusion of the virus.
-Cell-Mediated Response (T cells)
- CD8+ Cytotoxic T lymphocytes (CTLs) destroy infected hepatocytes and other cells.
- CD4+helper T cells help in the production of antibodies and increase the responses of CTL.
Epidemiology and Transmission of Yellow Fever Virus
Yellow Fever Virus is a mosquito-borne virus that causes life-threatening illness, which is currently endemic in various parts of Africa and South America, with prior dissemination in different parts of North America. In the 17th to 19th centuries, the yellow fever virus was transported to North America and Europe, leading to large outbreaks. In America, this virus re-emerged as a public health threat around 1970.
This disease is endemic in 13 countries in Central and South America. Similarly, the virus spread beyond the Amazon basin in 2014. Likewise, the Pan American Health Organization issued an epidemiological alert in 2025 about yellow fever in America because of a rise in confirmed cases in humans in the various regions of America. This rise lasts from the last months of 2024 to the first week of 2025. In 2025, the disease began to spread outside Amazon, while it was only concentrated in the Amazon region in 2024.
In the USA and Europe, 11 cases of yellow fever were recorded in people who travelled to West Africa and South America during 1970-2011. Likewise, the number of travel-associated yellow fever cases increased significantly in 2016, mainly due to the outbreaks in Angola and Brazil. More than 37 travel-associated cases were recorded in unvaccinated travelers who were residents of non-endemic countries. However, in 2019, travel-associated cases decreased due to a reduction in the occurrence of a large number of outbreaks caused by yellow fever and a decline in travel during the COVID-19 pandemic.
It is estimated that annually, more than 200,000 persons are affected by the yellow fever virus in the tropical regions of South America, Africa, and Central America, with at least 30,000 fatalities.
The yellow fever virus is transmitted primarily via the bite of infected mosquitoes. Transmission takes place through distinct ecological cycles:
Sylvatic (Jungle) Cycle
- It occurs mainly in tropical rainforests.
- The virus disseminates between non-human primates such as monkeys and mosquitoes dwelling in the forest.
- Humans get infected after being bitten by infected mosquitoes present in the forest areas.
Savannah (Intermediate) Cycle
- It is mainly found in Africa.
- It involves the transmission from mosquitoes to both monkeys and humans in semi-urban or rural areas.
- It causes localized outbreaks in villages.
Urban Cycle
- It occurs mainly in urban settings where the population density is high.
- Transmission occurs from human to mosquito to another human.
- It causes large epidemics, especially in cities with poor mosquito control.
Other Modes of Transmission
- Vertical Transmission: It is rare, but transmission can occur from an infected mother to the fetus.
- Blood transfusion and organ transplantation: very rare but possible.
- Laboratory Exposure: can occur in a laboratory by accident.
Clinical Manifestations of Yellow Fever Virus
The incubation period ranges from 3 to 6 days.
Mild Infection
- Sudden onset of high fever (39 to 40˚C)
- Chills
- Severe Headache
- Muscle Pain
- Nausea
- Vomiting
- Loss of Appetite
- Fatigue and Weakness
- Constipation
- Severe prostration
- Restlessness
- Irritability
Many patients recover completely; however, some show mild liver involvement with slight elevation of liver enzymes.
Severe Infection
- Jaundice
- Acute liver failure
- Bleeding from the gums, nose, and gastrointestinal tract
- Hematemesis
- Oliguria
- Acute kidney failure
- Hypotension and Shock
- Bradycardia
- Confusion and apathy
- Intractable hiccups
- Delirium
- Seizures
- Coma
- Multiple organ failure
Laboratory Diagnosis of Yellow Fever Virus
Sample Collection
Various samples are collected from patients with yellow fever, such as:
- Blood or Serum
- Whole Blood
- Liver Tissue
- Cerebrospinal Fluid
Virus Isolation
- Virus isolation is performed in cell cultures such as Vero cells or C6/36 cells under BSL2.
- Isolation is also done in laboratory animals such as mice by intracerebral inoculation, but in rare cases.
- This test confirms the active infection, but it is time-consuming and requires a high biosafety level lab.
Molecular Detection (RT-PCR)
- It is sensitive, rapid, and highly specific in detecting viral RNA.
- Viral RNA can be detected during the first 10 days after the onset of symptoms in serum samples.
- Widely used in a reference laboratory.
Serological Tests
-IgM Antibody Detection
- IgM antibody is detected by MAC-ELISA (IgM capture ELISA).
- It indicates a recent infection.
-IgG Antibody Detection
- This antibody is detected by ELISA.
- It indicates past infection and vaccination response.
-Immunohistochemistry
- This test is considered the “gold standard” for the diagnosis of yellow fever in severe cases.
- Histopathological test is performed on liver and other tissue sections, where liver biopsy shows midzonal necrosis and councilman bodies (apoptotic hepatocytes).
Treatment and Antiviral Therapy for Yellow Fever Virus
Currently, there is no specific antiviral therapy available for the yellow fever virus. Management is mainly supportive to lower symptoms and prevent complications.
Supportive Treatment
- Proper rest
- Use of oral or intravenous fluids to control dehydration.
- Use of pain relievers to decrease fever and reduce pain.
- Use of acetaminophen (paracetamol) for pain or fever while avoiding aspirin or other nonsteroidal anti-inflammatory drugs like ibuprofen, which can increase bleeding risk.
- Patients with severe disease should be admitted to the hospital for continuous monitoring of vital signs and liver function.
- After returning from the yellow fever endemic area, individuals should be monitored for the symptoms of yellow fever.
- If the person has symptoms of yellow fever, they should be isolated for up to 5 days after the symptoms begin, as this will reduce the spread of the virus to other uninfected mosquitoes, which can transmit the virus to other healthy individuals.
Prevention and Control of Yellow Fever Virus
Vaccination
- The live attenuated yellow fever vaccine is used as a primary control.
- This vaccine is highly effective, which provides lifelong immunity in most people.
- Vaccination is recommended for those people living or travelling to endemic areas or travelers before travelling to yellow fever risk countries.
Vector Control
- Elimination of breeding sites of mosquitoes, such as buckets, tires, and containers.
- Use of insecticides and larvicides in high-risk areas.
Proper personal protection, such as the use of insect repellent, long-sleeved clothing, and bed nets.
Surveillance and Outbreak Control
- Early detection of the yellow fever virus through laboratory diagnosis can reduce further transmission to others.
- Rapid response to outbreaks, including mass vaccination campaigns.
- Public awareness about symptoms, prevention, and vaccination against the yellow fever virus.
Travel Measures
- Travelers should be vaccinated before travelling to high-risk areas.
- Travelers should follow WHO guidelines for endemic regions.
- After returning from travel, individuals should be monitored and isolated if they show symptoms of yellow fever so as to prevent spread to other people.
Conclusion
The yellow fever virus belongs to the genus Flavivirus of the family Flaviviridae, which is a mosquito-borne pathogen that causes a wide range of diseases from mild illness to severe infection, such as hemorrhage, delirium, seizures, multiple organ failure, and even coma. Diagnosis is mainly done by methods such as virus isolation, RT-PCR, and serology.
Treatment is mainly supportive since no specific antiviral therapy is available. Prevention through vaccination, surveillance, and outbreak control remains the most effective strategy for the control of the yellow fever virus.
References
- Carmona, S. (2025). Yellow fever. In MSD Manual Professional Edition. MSD Manuals. Retrieved from https://www.msdmanuals.com/professional/infectious-diseases/arboviruses-arenaviridae-and-filoviridae/yellow-feve
- Creative Diagnostics. (n.d.). Yellow fever virus.
https://www.creative-diagnostics.com/Yellow-Fever-Virus.htm - Gardner, C. L., & Ryman, K. D. (2010). Yellow fever: A reemerging threat. Clinical Laboratory Medicine, 30(1), 237–260.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4349381/ - Monath, T. P., & Barrett, A. D. T. (2003). Pathogenesis and pathophysiology of yellow fever. In A. M. Kielian, C. R. M. Fauquet, & M. L. Van Regenmortel (Eds.), Advances in virus research (Vol. 60, pp. 343–395). Academic Press. https://www.researchgate.net/publication/8946680_Pathogenesis_and_Pathophysiology_of_Yellow_Fever
- Srivastava, S., Dhoundiyal, S., Kumar, S., Kaur, A., Khatib, M. N., Gaidhane, S., Zahiruddin, Q. S., Mohanty, A., Henao-Martinez, A. F., Krsak, M., Rodriguez-Morales, A. J., Montenegro-Idrogo, J. J., Bonilla-Aldana, D. K., & Sah, R. (2024). Yellow fever: Global impact, epidemiology, pathogenesis, and integrated prevention approaches. Infez Med, 32(4), 434–450. https://doi.org/10.53854/liim-3204-3
- Pan American Health Organization. (n.d.). Yellow fever. https://www.paho.org/en/topics/yellow-fever
- Pan American Health Organization. (2018, July 1). Laboratory diagnosis of yellow fever virus infection [Technical document]. World Health Organization. https://www.who.int/publications/i/item/laboratory-diagnosis-of-yellow-fever-virus-infection
- Centers for Disease Control and Prevention. (2024, May 15). Transmission of yellow fever virus. U.S. Department of Health and Human Services. https://www.cdc.gov/yellow-fever/php/transmission/index.html
- Centers for Disease Control and Prevention. (2025, November 19). Symptoms, diagnosis, and treatment of yellow fever. U.S. Department of Health and Human Services. https://www.cdc.gov/yellow-fever/ symptoms-diagnosis-treatment/index.html
