Xanthoproteic Test Definition
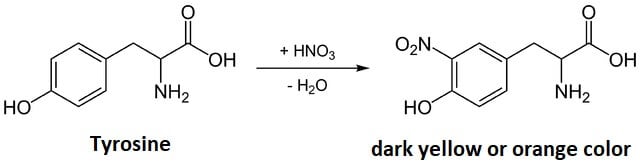
Xanthoproteic test is a biochemical test for the detection of amino acids containing phenolic or indolic groups like phenylalanine, tyrosine, and tryptophan (aromatic amino acids). The test is named Xanthoproteic test due to the formation of a yellow precipitate of xanthoproteic acid. The term ‘Xantho’ refers to ‘yellow’, so the test is often termed as the Yellow Protein Test. The test gives a positive result for amino acids containing benzene rings or other aromatic groups. The test is a qualitative test that provides information only on the presence or absence of the amino acids.
Objectives of Xanthoproteic Test
- To detect the presence of aromatic groups-containing amino acids like tyrosine and tryptophan.
- To differentiate tyrosine and tryptophan from other amino acids.
Principle of Xanthoproteic Test
The Xanthoproteic test is based on the fact that aromatic groups in the amino acids are nitrated by heating with concentrated HNO3 to yield intensely yellow-colored nitro derivative. On the addition of alkali, however, the residue turns orange due to the formation of a salt of the tautomeric form of the nitro compound. Benzene ring-containing amino acids like phenylalanine don’t give a positive test to this test because the phenyl group in phenylalanine is very stable, which doesn’t react with nitric acid in the conditions of this test. However, phenylalanine might give a positive result after an extended period of heating.
Requirements
Reagent
- Concentrated Nitric acid
- 40% NaOH
- Test solution
Material Required
- Test tubes
- Test tube stand
- Pipettes
Procedure of Xanthoproteic Test
- About 1 ml of the sample solution is taken in a test tube. To this, the same amount of concentrated nitric acid is added.
- The test tube is allowed to cool down to room temperature. If the sample is a protein solution, a white precipitate might develop due to the denaturation of proteins.
- Then, 1 ml of 40% NaOH solution is added to the test tube and observed for color change.
Result and Interpretation of Xanthoproteic Test
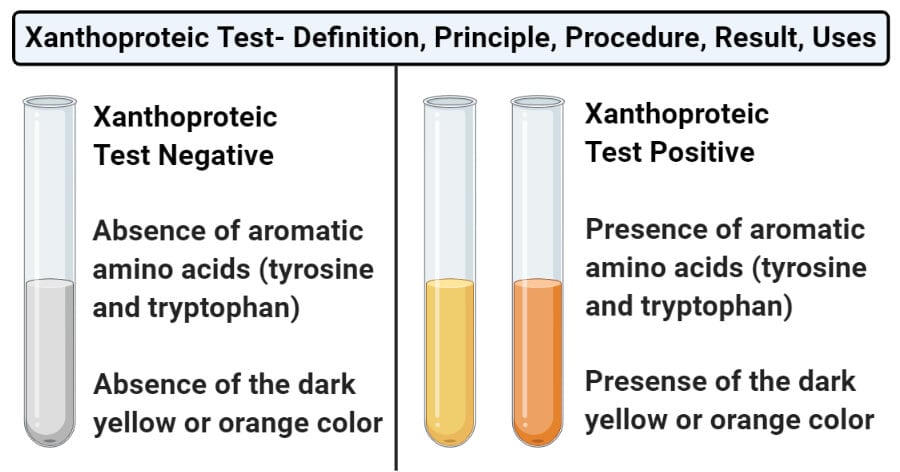
- Positive result: The appearance of a dark yellow or orange-colored solution represents a positive test. This indicates the presence of aromatic groups in the proteins and amino acids.
- Negative result: The absence of a dark yellow or orange-colored solution represents a negative test. This indicates the absence of aromatic groups in proteins and amino acids.
Uses
- This is a biochemical test for the detection of proteins and amino acids.
- The test allows the differentiation of aromatic amino acids from non-aromatic amino acids.
Limitations
- Even though phenylalanine is an aromatic group-containing amino acid, it doesn’t give a positive Xanthoproteic test due to the highly stable phenyl group.
References
- Nigam S. C. and Omkar (2003). Experimental Animal Physiology and Biochemistry. New Age International Pvt. Limited. New Delhi.
- D (2012). Biochemistry. Fourteenth Edition. Academic Publishers. Kolkata.
- Tiwari, A. (2015). Practical Biochemistry. LAP Lambert Academic Publishing.

Thank you 🥹
This site has been of useful help to me recently.
Thank you
This site had been a great impact to me.
Thank you
Highly and well simplified. Thank you
I this article gives it all,it has helped me in getting all the tests ????
Wow this site has being of great help to me,so easy to understand and assimilate. Thank you .