Sullivan and McCarthy’s Test Definition
Sullivan and McCarthy’s test is a biochemical test for the detection of the amino acid methionine or methionine-containing proteins. The test is a specific test for methionine which has a high degree of specificity towards methionine as it gives negative results with all other amino acids. The test, however, gives a positive result with glycylmethionine. It is a desirable method for methionine detection as it is a rapid and simple method for the assay of methionine even if fermented extract. Sullivan and McCarthy’s test is a color reaction of proteins which indicates the presence of a certain molecule by the development of a particular color with a particular reagent.
Objectives of Sullivan and McCarthy’s Test
- To detect the presence of the amino acid methionine or methionine-containing proteins.
- To differentiate methionine from other sulfur-containing amino acids like cysteine and cystine.
Principle of Sullivan and McCarthy’s Test
The Sullivan and McCarthy’s test is based on the reaction between nitroprusside and alkaline solution of methionine under acidification. The test was developed when it was discovered that tryptophan and histidine both produce a red color with nitroprusside under similar conditions. Thus, acid is added to the sample solution in order to destroy any tryptophan present by the process of acid hydrolysis. Similarly, it has been stated that the addition of glycine eliminates the detection of histidine. The red color is obtained after the addition of sodium nitroprusside to an alkaline solution of methionine followed by the acidification of the reaction.
Requirements
Reagent
- 10% sodium nitroprusside
- 5N Sodium hydroxide
- 6N HCl
- 2% glycine
- Sample solution
Materials required
- Water bath
- Ice bath
- Vortex
- Test tubes
- Test tube stand
- Pipettes
Procedure of Sullivan and McCarthy’s Test
- A few drops of sodium hydroxide are added to 1 ml of test solution in a test tube.
- To this, a few drops of 2% glycine and 10% sodium nitroprusside solution is added.
- The solution is then mixed together by placing it in a vortex for 2 minutes.
- The test tube is then placed in a water bath at 40°C for 15 minutes.
- The tube is then cooled in ice-cold water for 5 minutes followed by the addition of 0.5ml of 6N HCl.
- The contents are then mixed together in a vortex, and the test tube is allowed to stand at room temperature for 15 minutes. The observation is then made.
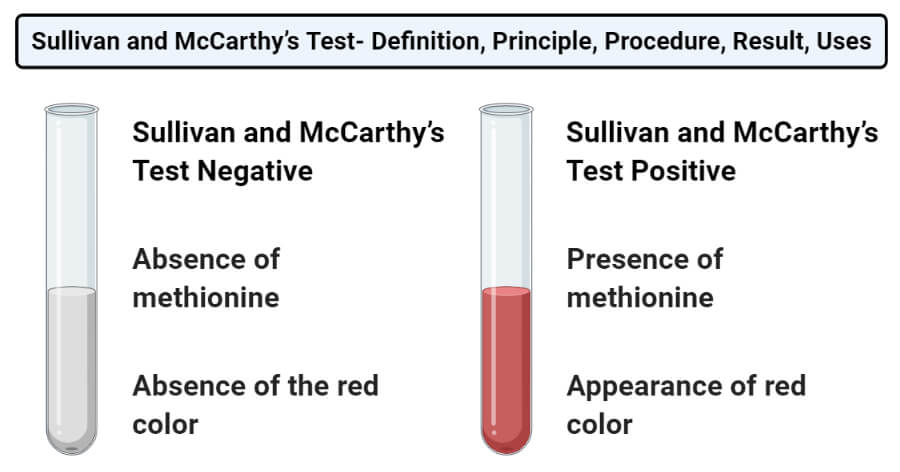
Result and Interpretation of Sullivan and McCarthy’s Test
- Positive result: The positive result is represented by the appearance of the red color. This indicates the presence of methionine.
- Negative result: The negative result is represented by the absence of the red color. This indicates the absence of methionine.
Uses of Sullivan and McCarthy’s Test
- Sullivan and McCarthy’s test is used for the detection of methionine either in a free form or in proteins.
- The test can be modified for quantification where the test sample can be matched against the standard methionine solution.
Limitations
- The test might give a positive result with commercial leucine as it might contain some traces of methionine.
- The test might not give reproducible results with standard solutions of methionine.
References
- D (2012). Biochemistry. Fourteenth Edition. Academic Publishers. Kolkata.
- Tiwari, A. (2015). Practical Biochemistry. LAP Lambert Academic Publishing.
- McCarthy T.E and Sullivan M.X (1941). A New and Highly Specific Colorimetric Test For Methionine. Journal of Biological Chemistry. 151. 635.
- Csonka F and Denton C. (1946). Methionine Determination in Proteins and Foods. J. Biol. Chem. 1946, 163:329-338.

Hallo
How can we determine the percentage of methionine by using the Kjeldahl apparatus?