The rubella virus, an RNA virus of the genus Rubivirus in the Matonaviridae family, causes rubella (German measles). It typically causes a mild disease transmitted via respiratory droplets, with symptoms such as low-grade fever, lymphadenopathy, and a distinct maculopapular rash.
Maternal transmission during pregnancy can result in vision and hearing loss, heart defects, and other serious conditions. Vaccination effectively prevents rubella.
Taxonomy and Classifications of Rubella Virus
Kingdom: Orthornavirae
Phylum: Kitrinoviricota
Class: Alsuviricetes
Order: Hepelivirales
Family: Matonaviridae
Genus: Rubivirus
Species: Rubivirus rubella
Common name: Rubella virus
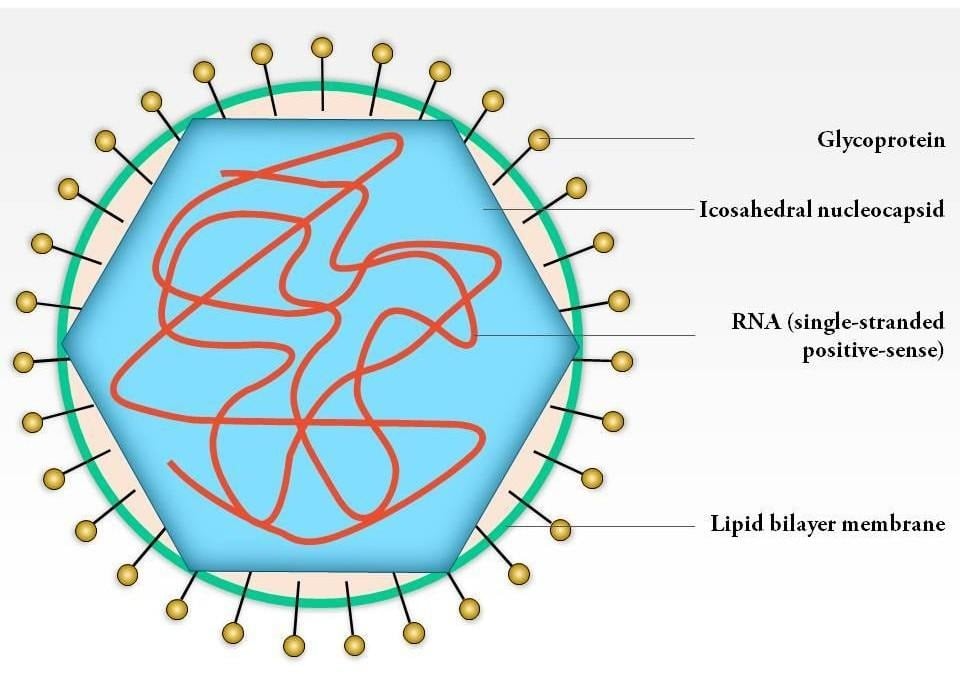
Structure and Morphology of Rubella Virus
- It is a small, spherical virus with a diameter of about 50-70 nm.
- Rubella virus is a single-stranded, positive-sense RNA virus, meaning its genome can function directly as mRNA for protein synthesis upon infection.
- It is enveloped.
- It has icosahedral symmetry.
- It consists of a single-stranded, positive-sense RNA genome approximately 9.7kb.
Source: https://www.slideteam.net/0614-rubella-virus-medical-images-for-powerpoint.html
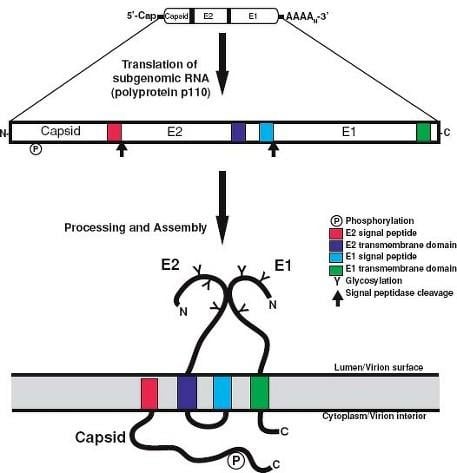
Genome Organization and Proteins of Rubella Virus
-The genome is single-stranded, positive-sense RNA, which is around 9.7 kb in size.
-It is linear RNA with a 5’ cap and 3’poly-A tail, allowing it to function directly as mRNA for the synthesis of protein.
-Open Reading Frames (ORFs)
- Structural ORF (S-ORF): It is located at the 3’ end and codes for the protein that forms the viral particle.
- Non-Structural ORF (NS-ORF): It is located at the 5’ end and codes for the protein that is involved in the replication of the virus.
-Non-Structural Proteins (NSP)
- Non-structural proteins are essential for viral RNA replication, processing of viral proteins, and regulation of viral gene expression, ensuring efficient virus multiplication and infection.
-Structural Proteins
- Capsid protein (C): It encloses the viral RNA and forms the nucleocapsid.
- Envelope glycoproteins (E1 and E2): E1 contributes to attachment to host cell receptors and fusion of the membrane. Similarly, E2 aids in the assembly of the virus and interacts with E1.
Source: https://basicmedicalkey.com/rubella-virus/
Protein
Capsid Protein
- It forms the nucleocapsid via binding and protecting the RNA of the virus.
- It gives structural stability to the virus particle.
Envelope Glycoproteins
-E1 Glycoprotein
- It is the main protein that is responsible for the attachment of the virus to the host cell receptors.
- It helps in the fusion of the membrane during the entry of the virus.
-E2 Glycoprotein
- It functions along with E1 for the assembly of the virus.
- It plays a crucial role in the maturation and budding of the virus.
Non-Structural Proteins
- It is encoded by the 5’ region of the genome.
- It is involved in the replication of RNA, processing, and regulation of viral gene expression.
- Examples of non-structural proteins are viral proteases and RNA polymerase components.
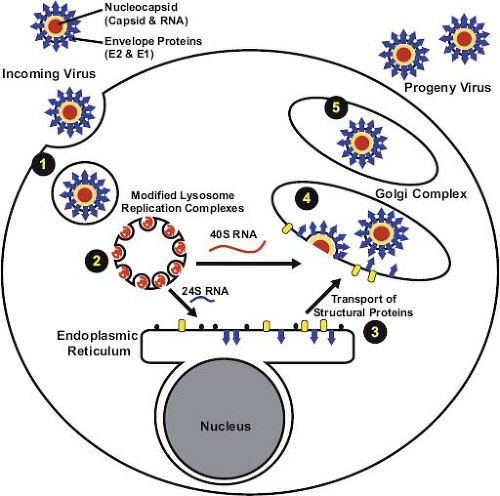
Replication Cycle of Rubella Virus
Attachment
- The virus adheres to the host cell receptors through the E1 glycoprotein.
Entry
- The virus then enters the host cell via receptor-mediated endocytosis.
Uncoating
- After that, the viral envelope merges with the endosomal membrane, causing the release of the RNA genome into the cytoplasm.
Translation
- The positive-sense RNA acts as mRNA, which is then translated to produce non-structural proteins that are essential for replication.
RNA Replication
- The viral RNA-dependent RNA polymerase synthesizes a negative-strand RNA as a template.
- This template is then utilized to generate new genomic and sub-genomic RNA.
Translation
- Then the newly formed sub-genomic RNA is translated into structural proteins such as capsid, E1, and E2 glycoproteins.
Assembly
- After that, the capsid proteins enclose the newly synthesized genomic RNA to form nucleocapsids.
Maturation
- Envelope glycoproteins are then processed in the Golgi apparatus and endoplasmic reticulum.
Budding and Release
- Then the virions acquire their envelope through budding via intracellular membranes.
- Finally, the mature virions are transported to the surface of the host cell and liberated via exocytosis.
Source: https://basicmedicalkey.com/rubella-virus/
Pathogenesis and Host Immune Response to Rubella Virus
- Entry into Host Cell
- The virus enters the body via the respiratory tract through the inhalation of droplets from an infected person.
- Primary Replication
- The initial replication takes place in the nasopharyngeal epithelium and regional lymph nodes.
- Viremia
- The virus then enters the bloodstream, causing primary viremia, which then spreads to other organs and tissues of the host cell.
- Secondary Replication
- After that, further multiplication takes place in reticuloendothelial tissues, causing secondary viremia.
- Immune Response
- The host immune system then produces IgG and IgM antibodies, which activate cellular immunity that leads to the clearance of the virus.
- Formation of an immune complex contributes to rash and joint symptoms.
Transplacental Transmission
- The virus can be transferred from the mother to the fetus by crossing the placenta during pregnancy.
Fetal Infection
- The virus replicates and damages the vital organs of the fetus, causing Congenital Rubella Syndrome.
Host Immune Response
Innate Immunity
- Early response consists of the production of interferon and stimulation of natural killer cells to restrict the replication of the virus.
Humoral Immunity
- Initially, IgM antibodies appear, indicating recent infection, and are unnoticeable after 2 months.
- While IgG antibodies persist for a lifetime and protect against reinfection.
Cell-mediated Immunity
- It starts 1 week after the appearance of humoral immunity and kills infected cells, causing clearance of the virus.
Immune Complex Formation
- Formation of antigen-antibody complexes results in rash and joint symptoms.
Epidemiology and Transmission of Rubella Virus
The rubella virus is widespread and infects only humans. Rubella remains a public health concern worldwide, especially in developing countries, due to frequent misdiagnosis from symptom overlap with other diseases. Rubella can cause Congenital Rubella Syndrome (CRS) in fetuses. In 2010, around 105,000 CRS cases were reported globally. Before the rubella vaccine’s introduction in 1969, epidemics occurred every 6-9 years, affecting large adult populations. Incidence peaks in children aged 5-9, with maximum transmission during early spring and late winter. From 1966-1968 in the USA, about 60% of cases occurred in children under 10, and 23% in people 15 and older. Between 1963 and 1964, the USA reported roughly 12.5 million rubella cases, leading to 2084 encephalitis cases, 11250 spontaneous abortions, 2100 neonatal deaths, and over 20000 infants with CRS. CDC data show 39 annual CRS cases from 1969-1979, dropping to seven per year between 1979 and 1988. In 1987, 48% of rubella infections occurred in individuals over 15. From 1989 to 1991, rubella and CRS reemerged slightly. A single rubella-containing vaccine (RCV) dose is 95% effective, significantly reducing rubella and CRS globally. By the end of 2018, 168 countries had introduced RCV, covering around 87% of the global population. By December 2019, 81 of 194 WHO member countries had eliminated rubella, with an additional 6 controlling rubella and CRS. As of 2023, 84% of WHO member countries had incorporated rubella vaccines into national immunization schedules.
Rubella virus is transmitted via various routes, described as:
- Virus is primarily transmitted through the coughing and sneezing of an infected person.
- It is also transmitted via close contact with the nasal or throat secretions of infected persons.
- In crowd settings, the virus can also spread via droplets over short distances.
- The virus is transmitted from infected pregnant women to the fetus through the placenta, causing Congenital Rubella Syndrome.
- The asymptomatic individuals can also spread the virus.
- The infants born with congenital infection discharge the virus in their urine and respiratory secretions, acting as a source of infection.
Clinical Manifestations of Rubella Virus
The incubation period typically ranges from 14 to 21 days. The infectious period is typically 7 days prior to and 7 days after the onset of rash. Infants with CRS can shed the virus for up to 1 year after birth. Most of the cases are subclinical and self-limiting. While some may develop symptoms described as:
Mild symptoms
- Headache
- Low-grade fever
- Malaise
- Anorexia
- Fatigue
- Coryza
- Conjunctivitis
Lymphadenopathy
- Is characterized by swollen postauricular, occipital, and cervical lymph nodes.
- These symptoms disappear as soon as the rash appears.
Rash
- Maculopapular rash appears on the face, which then spreads to the trunk and limbs.
- At the end of the first day, rashes are present all over the body.
- This rash usually lasts for about 3 days and disappears without any stain.
Forchheimer Spots
- Usually, small, red spots appear on the soft palate, which is not always present but is characteristic of rubella.
Mild Disease in Children
- Children are often asymptomatic or show very mild symptoms.
Congenital Manifestations
If a pregnant woman is infected, the virus may cross the placenta and cause Congenital Rubella Syndrome (CRS) in the fetus, which has the following features:
- Cataracts and other eye defects
- Deafness
- Heart Disease
- Intellectual disability and developmental delay
Complications
Complications are rare and occur most commonly in adults. Most common complications are:
- Arthralgia/Arthritis: occurs in 70% of adult women and often affects fingers, wrists, or knees.
- Hemorrhagic Manifestations: often occur in children, and vascular damage and thrombocytopenia are common. Intrarenal, cerebral, and gastrointestinal hemorrhage occur.
- Encephalitis: It is more common in women.
- Orchitis
- Neuritis
Laboratory Diagnosis of Rubella Virus
Sample Collection
Various samples are collected from rubella and CRS patients, such as:
- Throat or Nasopharyngeal swabs
- Blood or Serum
- Urine
- Cerebrospinal Fluids
Serology
- The presence of IgM antibodies demonstrates recent infection, which is detectable 5 days after the onset of rash.
- The presence of IgG antibodies demonstrates previous infection, and rising titers between acute- and convalescent- phase serum samples confirm the recent infection.
- False positive tests have been demonstrated in people with parvovirus infections, positive rheumatoid factor, and positive heterophile test for infectious mononucleosis.
Virus Isolation
- The virus is isolated in cell cultures such as Vero or RK13 cells.
- However, the culture of viruses is done rarely due to labor intensity and complexity.
Enzyme-linked Immunosorbent Assay (ELISA)
- This test is sensitive, easy to perform, and is widely available.
- It is used for the detection of IgM antibodies.
Immunofluorescent Antibody Assay (IFA)
- This test is sensitive and rapid to perform.
- Proper precautions must be taken while performing the test to prevent false-positive results.
Hemagglutination Inhibition (HI):
- This test is sensitive and simple to perform, which allows the screening of the rubella virus.
- A 4-fold increase in HI-derived antibody titer in serum is indicative of recent infection.
- This test can also be modified for the detection of IgM antibody, which indicates the primary infection.
Treatment and Antiviral Therapy for Rubella Virus
Till now, there is no antiviral drug used for the rubella virus. However, supportive care can be used as a treatment for infection caused by the rubella virus.
Supportive Care
- Rest and adequate hydration.
- Analgesics are used for joint pain and arthritis.
- Antipyretics such as acetaminophen are used for fever.
Symptoms Management
- Mild cases usually resolve within 1-3 days.
- For joint pain, non-steroidal anti-inflammatory drugs can be used.
- Infection during pregnancy requires careful monitoring of the development of the fetus.
Prevention and Control of Rubella Virus
Vaccination
- The MMR vaccine is the most effective method of prevention.
- Two doses are given: typically at 12-15 months and 4-6 years of age, and the interval between two doses is 4 weeks.
- Vaccination provides long-term immunity and prevents outbreaks.
Screening and Immunization of Adult Women
- Women of childbearing age should be regularly screened for rubella immunity before pregnancy.
- Non-immune women should be vaccinated before pregnancy to stop Congenital Rubella Syndrome (CRS).
Isolation of an Infected Person
- Individuals having an infection with the rubella virus should be isolated for at least 7 days after the onset of rash to lower transmission of rubella.
Public Education and Surveillance
- Providing knowledge to communities about vaccination, transmission routes, and the risks of rubella during pregnancy.
- Timely surveillance and monitoring of cases of rubella aid in outbreak identification and help in the implementation of control measures.
Control in Healthcare Settings
- Always use standard precautions in the hospital while handling suspected cases of rubella.
- Pregnant healthcare workers should avoid direct contact with the person infected with the rubella virus.
Conclusion
Rubella is a small, enveloped RNA virus that causes Rubella, that is characterized by fever, lymphadenopathy, and a characteristic maculopapular rash that spreads from the face to the trunk and limbs. Generally, the infection is mild and self-limiting in children and adults, whereas it may impose risk in pregnant women as transplacental transmission can result in Congenital Rubella Syndrome, which causes serious illness in the child. The diagnosis relies mainly on serological and molecular methods. Since no specific antiviral drug is available, vaccination and management are mainly supportive, with preventive measures focusing mainly on protection of high-risk groups.
References
- World Health Organization. (n.d.). The structure and biology of rubella virus. TechNet-21. https://www.technet-21.org/en/manual-introduction/chapter-1-measles-and-rubella-an-overview/1-5-the-structure-and-biology-of-rubella-virus
- Leonor, M. C., Afzal, M., & Mendez, M. D. (2025). Rubella. In StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK559040/
- Das, P. K., & Kielian, M. (2021). Molecular and structural insights into the life cycle of rubella virus. Journal of Virology, 95(10), e02349-20. https://doi.org/10.1128/JVI.02349-20
- National Center for Biotechnology Information. (n.d.). Togaviruses: Rubella virus. In Medical Microbiology. National Library of Medicine. Retrieved March 31, 2026, from https://www.ncbi.nlm.nih.gov/books/NBK8200/
- World Health Organization. (n.d.). Infection, immune response and laboratory diagnosis of rubella and congenital rubella syndrome (CRS). In Manual for the laboratory-based surveillance of measles, rubella, and congenital rubella syndrome. https://www.technet-21.org/en/manual-introduction/chapter-1-measles-and-rubella-an-overview/1-7-infection-immune-response-and-laboratory-diagnosis-of-rubella-and-crs
- Centers for Disease Control and Prevention. (2024, April 25). Chapter 20: Rubella. In Epidemiology and Prevention of Vaccine-Preventable Diseases (The Pink Book). https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-20-rubella.htm
- National Center for Biotechnology Information. (2020). The epidemiology of rubella, 2007–18: An ecological analysis of surveillance data. The Lancet Global Health, 8(11), e1399–e1407. https://doi.org/10.1016/S2214-109X(20)30320-X
- Communicable Diseases Agency. (2025, March 26). Rubella. https://www.cda.gov.sg/professionals/diseases/rubella/
- Centers for Disease Control and Prevention. (2015). Rubella. In Epidemiology and Prevention of Vaccine-Preventable Diseases (13th ed.). https://commed.vcu.edu/IntroPH/Communicable_Disease/pinkbook/rubella9e.pdf
