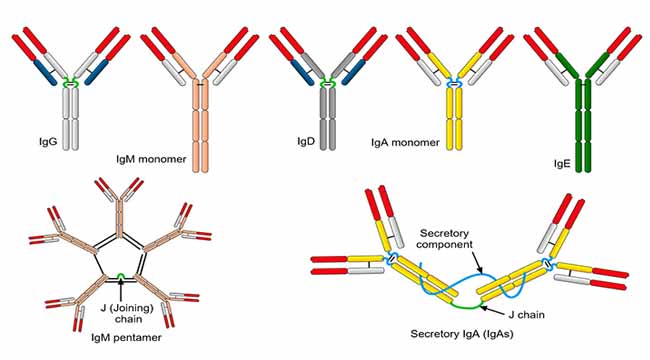
Antibodies are glycoproteins produced in membrane-bound or secreted form by B lymphocytes in response to exposure to foreign structures known as antigens. The human immunoglobulins are a family of proteins that confer humoral immunity and perform vital roles in promoting cellular immunity. There are five classes of antibodies or immunoglobulins termed immunoglobulin G (IgG), IgM, IgA, IgD and IgE. All these classes have the basic four – chain antibody structure but they differ in their heavy chains termed γ, μ, α, δ and ε respectively.
The antibodies are the gamma globulins. Antibodies are often referred to as “first line of defense” against infection. The most important function of antibodies is to confer protection against microbial pathogens. Antibodies confer protection in the following ways:
- They prevent attachment of microbes to mucosal surfaces of the host.
- They reduce virulence of microbes by neutralizing toxins and viruses.
- They facilitate phagocytosis by opsonization of microbes.
- They activate complement, leading to complement-mediated activities against microbes.
Immunoglobulin A (IgA)
Predominant immunoglobulin in colostrum, saliva, tears, bronchial secretions, nasal mucosa, prostatic fluid, vaginal secretions, and mucous secretions of the small intestine.
- Blocks uptake of antigen, bacterial or viral attachment.
- Limits inflammation induced by classical pathway complement activation.
- Promote microbial destruction through ADCC by binding to leukocyte receptors.
- Secretory IgA can play an important first line of defense in antigen clearance by binding to antigens that leak across an epithelium and transporting them back across to prevent their entry.
- Activates alternative pathway of complement.
Immunoglobulin G (IgG)
Most abundant class of immunoglobulins in the serum, comprising about 80% of the total serum immunoglobulin. There are four IgG subclasses IgG1, IgG2, IgG3, and IgG4. IgG1, IgG3, and IgG4 are special because these are the only immunoglobulins with the ability to cross the placental barrier.
- Protects the developing fetus against infections.
- Activates complement (IgG3, IgG1, and IgG2, in order of their efficiency).
- Mediate opsonization due to affinity towards Fc receptors on phagocytic cells (IgG1 and IgG3 bind with high affinity, IgG4 with intermediate affinity and IgG2 with extremely low affinity).
- Binds microorganisms and facilitates the process of phagocytosis of microorganisms.
- Takes part in precipitation, complement fixation, and neutralization of toxins and viruses.
- Confers protection against the microorganisms that are present in the blood and tissues.
Immunoglobulin M (IgM)
IgM is basically a pentamer, composed of five immunoglobulin subunits (monomeric subunits, IgMs) and one molecule of J chain. Two subclasses of IgM (IgM1 and IgM2) are found, which differ in their µ chains. IgM1 consists of µ1 and IgM2 consists of µ2 chains
- Highly efficient in activating the classical complement pathway.
- Major immunoglobulin that is expressed on the surface ofB cells where it serves as an antigen receptor.
- Gives primary immune esponse.
- Act as agglutinin, opsonin, lysins and complement fixing antibody.
Immunoglobulin E (IgE)
Also known as reaginic antibody that mediates the type I immediate hypersensitivity (atopic) reaction. Mostly found extravascularly in lining of the respiratory and intestinal tracts.
- Important role in protection against parasitic infection.
- Activates type- I hypersensitive reaction (anaphylactic reaction).
Immunoglobulin D (IgD)
Comprises less than 1% (about 0.2%) serum immunoglobulins. Precise function of IgD remains unclear.
- Present on the surface of B lymphocytes along with IgM. IgD and IgM serve as recognition receptors for antigens.
- May play role in antigen triggered lymphocyte differentiation.

This website has helped me a lot during my preparation for exams. I am so grateful that I can’t even express in words. The notes are written in simple and lucid language and are informative as well. Nice job. Thanks a lot and all the best for future.
Woow, its very motivating to get such a reply from our readers, Thanks a lot for liking our website. 🙂
Great sir g
Nice job