Pharmacovigilance is the science and activities related to the detection, assessment, understanding, and prevention of adverse effects or any drug-related problems.

Pharmacovigilance involves monitoring and evaluating the safety of marketed drugs, collecting and analyzing data on adverse drug reactions, and taking appropriate regulatory actions whenever necessary. The history of Pharmacovigilance started in 1848 with the incidence of chloroform death.
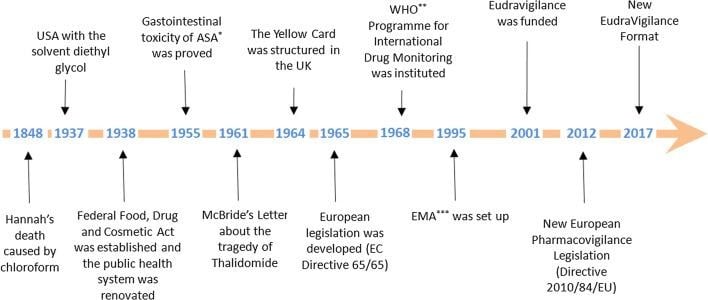
Source: https://link.springer.com/article/10.1007/s11096-018-0657-1
The objectives of pharmacovigilance are:
- Detection of Adverse Drug Reactions (ADRs) of drugs that are already on the market or under development.
- Assessment of drug safety by analyzing data from various sources, including clinical trials, spontaneous reports, literature, and regulatory submissions.
- Understanding risk factors that contribute to the occurrence of adverse drug reactions, including patient characteristics, drug interactions, dosage forms, and underlying medical conditions.
- Prevention of harm.
- Promotion of rational use of drugs.
- Pharmacovigilance involves communicating safety information to healthcare professionals, patients, and the public to raise awareness about potential risks and encourage reporting of adverse effects.
- It provides regulatory authorities with the necessary information to make informed decisions about the approval, labeling, and post-marketing surveillance of pharmaceutical products.
- Continuous monitoring.
The Importance of Drug Safety Monitoring
Pharmacovigilance and drug safety monitoring are crucial components for the evaluation of the safety and efficacy of drug products even after they are marketed. The importance of Drug Safety Monitoring is:
- Detection of Rare Adverse Drug Reactions: Medication errors and ADRs are well documented. Hospital-based surveillance reduces the risk and severity of ADRs. It also helps in the detection of rare ADRs through documented evidence.
- Identification of Long-term and Delayed Effects: Safety monitoring helps to prevent prolonged use of the drug.
- Monitoring Drug Use in Real-world Conditions: Rational drug use and how drugs behave under real-world conditions are monitored by drug profiles.
- Detection of Drug Interactions: Patients with chronic disease conditions or polypharmacy habits cause reduced drug effectiveness and unexpected adverse effects.
- Ensuring Safe and Rational Use of Medicines: Regulatory guidance and compliance help in ensuring safe and rational use of medicines.
- Protection of Public Health: Protection of patient confidentiality by applying careful standard operating procedures. Product recalls, safety alerts, and harmful drug withdrawal protect public health.
- Improvement of Risk-Benefit Assessment: Promoting understanding, educating, and clinical training in pharmacovigilance and its effective communication to the public about drug continuation and drug withholding.
- Building Confidence in Healthcare Systems: Training, transparency, and accountability in drug use provide confidence in the healthcare team.
- Regulatory Decision-Making: Transparency within the affairs body to clear misunderstandings. Open communication regarding the benefits, harm, effectiveness, and risk of medicines for the risk management strategy and safety warnings.
- Early Detection of Safety Signals: Identification of early warning signs of potential risk, early interventions, prevention of widespread harm, and timely communication to healthcare professionals helps in the prevention of the seriousness of drug therapy.
Classification of Adverse Drug Reactions
ADRs are a response to a drug that is noxious and unintended and occurs at doses normally used in humans for the prophylaxis, diagnosis, or therapy of disease, or for modification of physiological functions. ADRs are caused either by direct toxicity or by hypersensitivity reactions.
Rawlins- Thompson classification of ADRs is:
Type- A
“A” stands for Augmented reaction. Eighty percent of reactions are Type- A adverse effects of a drug. These effects are pharmacological effects of a drug and are often predictable. They are dose-dependent, and an increase in dose increases the severity. This type of adverse effect is commonly seen in drugs with low therapeutic index and impaired elimination in patients. The prevention of this adverse effect is dose reduction. For example, bleeding with warfarin and other anti-coagulants, hypoglycemia from insulin, and bradycardia associated with beta-blockers.
Type- B
“B” stands for Bizarre reaction. These types of reactions are unpredictable, infrequent, and are characterized by qualitative variations in individual responses to drugs. Type B reactions often involve allergic or immunological mechanisms.
Type- C
“C” stands for Chronic drug use reaction. These reactions have been found to relate to the cumulative toxic effects of a drug over time, in which the adverse effects increase gradually. It is mostly uncommon and is time-related and dose-related. The prevention of this reaction is reducing the dose. For example: Hypothalamic- pituitary- adrenal- axis- suppression effect caused by corticosteroids.
Type- D
“D” stands for Delayed reaction. This type of reaction occurs after many years of drug use or treatment. It is due to extended exposure (accumulation) or limited exposure at a crucial time. For example: Phenytoin during pregnancy, Contact dermatitis due to metal exposure, etc.
Type- E
“E” stands for End of Use Reaction. This reaction is due to the abrupt withdrawal of a medicine. For example: Insomnia, anxiety, and perceptual disturbance due to the sudden withdrawal of benzodiazepines. This reaction can be prevented by slow withdrawal of the drug.
Type- F
“F” stands for Failure of therapy reaction. This reaction is characterized by unexpected failure of therapy, where a drug undesirably increases or decreases the efficacy of another drug. It is a dose-dependent reaction. The drug interactions, counterfeit medications, and underdosing are the causes of this type of reaction, and can be managed by overcoming these parameters. For example: Inadequate dosage of an oral contraceptive in co-administration of an enzyme inducer, Antimicrobial resistance, etc.
Serious vs. Non-Serious Adverse Events
As per the WHO, adverse effects are defined as “any untoward medical occurrence that may present during treatment with a medicine but which does not necessarily have a causal relationship with this treatment.” Most of the adverse events are non-serious, but some of them are fatal.
| Excessive pharmacological effects, dose-dependent toxic effectsRebound response after discontinuation/ Drug withdrawal reaction | Serious Adverse Events | Non- Serious Adverse Events |
| Reaction | Unpredictable or Immunologic Reaction | Predictable or Non-immunologic Reaction |
| Dependence | Patient dependent | Dose dependent |
| Severity | High | Low to moderate |
| Outcome | Hospitalization, disability, or death | Temporary discomfort |
| Impact on therapy | Often requires discontinuation | Usually manageable |
| Causes | -Idiosyncrasy or Pharmacogenetics -Genetically determined toxicity -Hypersensitive reaction -Intolerance/supersensitivity. | -Excessive pharmacological effects -Dose-dependent toxic effects -Rebound response after discontinuation/ Drug withdrawal reaction |
| Reporting need | Urgent | Routine reporting |
| Examples | -Drug- induced hearing loss, severe bleeding due to anticoagulants -Teratogenicity caused by phenytoin in pregnancy | -Headache -Dizziness -Non-severe skin rash, etc. |
Spontaneous Reporting Systems (FAERS, Yellow Card, DAEN, Canada Vigilance)
Spontaneous reporting systems are a passive surveillance platform for healthcare professionals and patients to report suspected ADRs to regulatory agencies. It is defined as the voluntary communication by a patient, healthcare professional, or pharmaceutical company to the National Pharmacovigilance Program about ADR which are not identified in the preclinical or clinical trials. This helps in identifying patterns and emerging safety concerns. There are different platforms for ADRs reporting in different countries, and they are listed below.
FAERS
The FDA Adverse Event Reporting Systems (FAERS) is a database that is designed to support the FDA’s post-marketing safety surveillance program for drug and therapeutic biologic products. It is also known as “Med watch” for primary reporting programs.
Yellow Card Scheme
These are ADRs reporting platforms in the United Kingdom for suspected or observed ADRs to medicinal products or medical devices that were started since 1964. It uses a black triangle symbol that identifies drugs that need intensive monitoring. Information included on the yellow card scheme is suspected drugs, suspected reaction, patient details, and reporter details.
DAEN
In Australia, the Database of Adverse Events Notifications serves to provide public access to report suspected side effects. The Therapeutic Goods Administration (TGA) manages DAEN. It is also known as “Blue card” ADRs reporting.
Canada Vigilance
In Canada, a spontaneous reporting program is operated by Health Canada to collect reports of suspected adverse reactions to health products (both prescribed and non-prescribed drugs).
Signal Detection and Management
A signal is the information on a new or previously unknown or a known adverse reaction of a drug that requires further investigation and assessment. Signal detection helps to provide early warning of a drug causing a new adverse effect or a known effect becoming more frequent and severe in a new population. Signal is a matter of looking for patterns or clusters of reports that stand out from the background. There are two types of signal detections:
-Qualitative Methods: It includes spontaneously reported data and case series, which help to understand why a potential risk might exist. It includes:
- Clinical review of individual case safety reports.
- Case series analysis of similar cases by comparing dose, timing, and outcomes across patients.
- Causality assessment to determine the likelihood that a specific medicine or vaccine caused an observed adverse event.
- Medical literature review.
-Quantitative methods: Data mining to identify early safety risks from epidemiological or ongoing clinical trial data. This method uses numbers and models to highlight unusual drug-event patterns.
- Disproportionality analysis: It compares how often a drug-event pair appears versus how often expert drug-events occur by chance.
- Proportional Reporting Ratio: It measures whether an event is reported more often with one drug than with all others.
- Reporting odds ratio: It compares the odds of an event with a specific drug versus all other drugs.
Signal Management is the process of handling safety signals from detection to the final regulatory action. Signal management ensures that drug safety is evaluated continuously. The steps in Signal Management are:
- Signal Detection: Collection of the data from the appropriate source and identification of signals. The safety signals are generated by various sources such as spontaneous reporting, case control and cohort studies, preclinical as well as clinical studies.
- Signal Assessment: The detected signals are qualitatively reviewed, and patterns are checked to determine if they are real or not. Various software and techniques are used for the generation of good-quality data. In signal assessment, the seriousness of adverse events is assessed.
- Signal Evaluation: The signal is reviewed for its seriousness, frequency, and vulnerability and placed with an appropriate label, warnings, extra monitoring, or studies.
- Decision making and action: Implementation, communication, and monitoring product information and tracking follow-up dates to see if risks change.
Causality Assessment Methods (Naranjo Scale, WHO-UMC)
Causality Assessment Methods help in determining the relationship between a drug, a drug reaction, and the likelihood of adverse events. It is essential for evaluating drug safety, detecting potential safety signals, and ensuring patient welfare.
Naranjo Scale
It was developed in 1991 by Naranjo and coworkers from the University of Toronto. This scale was also designed for use in controlled trials and registration studies of new medications at their therapeutic dose. This method includes questions related to the timing of adverse events, improvement after drug withdrawal, reappearance after re-administration, and alternative causes. These questions consist of 10 questions that are answered as either Yes, No, or Do not know. The ADR probability scale has point values of -1, 0, +1, and +2 for each answer. After adding all points, the ADR is classified as Definite ADR (≥9), Probable ADR (5-8), Possible ADR (1-4), and Doubtful ADR (0 or less).
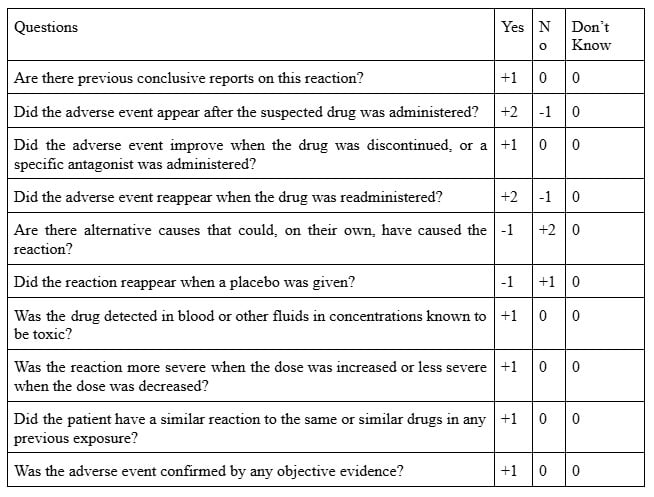
WHO-UMC
WHO-UMC stands for World Health Organization Uppsala Monitoring Centre. This method is used widely in International Pharmacovigilance Programs. It includes the time relation between drug use and the adverse event, presence or absence of competing cause, and response to drug withdrawal, dose reduction or drug readministration. It categorizes the adverse events into six categories:
Table: WHO-UMC Casualty Categories
| Causality term | Assessment criteria* |
| Certain | -Event or laboratory test abnormality, with a plausible time relationship to drug intake -Cannot be explained by disease or other drugs -Response to withdrawal plausible (pharmacologically, pathologically) -Event definitive pharmacologically or phenomenologically (i.e., an objective and specific medical disorder or a recognised pharmacological phenomenon) -Rechallenge satisfactory, if necessary |
| Probable/ Likely | Event or laboratory test abnormality, with a reasonable time relationship to drug intake -Unlikely to be attributed to disease or other drugs -Response to withdrawal clinically reasonable -Rechallenge not required |
| Possible | -Event or laboratory test abnormality, with a reasonable time relationship to drug intake -Could also be explained by disease or other drugs -Information on drug withdrawal may be lacking or unclear |
| Unlikely | -Event or laboratory test abnormality, with a time to drug intake that makes a relationship improbable (but not impossible) -Disease or other drugs provide plausible explanations |
| Conditional/ Unclassified | -Event or laboratory test abnormality -More data for proper assessment is needed, or -Additional data under examination |
| Unassessable/ Unclassifiable | -Report suggesting an adverse reaction -Cannot be judged because the information is insufficient or contradictory -Data cannot be supplemented or verified |
* All points should be reasonably complied with
The Role of the Qualified Person for Pharmacovigilance (QPPV)
The qualified person for pharmacovigilance is responsible for maintaining the pharmacovigilance system within the pharmaceutical industry, clinical, and hospital settings. The person holding a degree in medicine, pharmacy, or any relevant discipline with several years of experience in practical drug safety roles with in-depth knowledge of pharmacovigilance legislation, procedures, and guidelines is said to be a qualified person for pharmacovigilance activity. The qualified person serves as a bridge between the regulatory agencies and pharmaceutical settings regarding drug safety issues. They play a pivotal role in patient safety by managing risks associated with medicinal products.
The Roles:
- Responsible for maintaining and overseeing the company’s pharmacovigilance system and adequately documenting the file.
- Ensuring timely and accurate adverse event reporting to regulatory bodies and overseeing the collection, assessment, and submission of individual case safety reports.
- Development, maintenance, implementation, and evaluation of risk management plans and their effectiveness.
- Preparing and submitting the periodic safety update reports to specify the individual product safety profile by complying with the regulatory guidelines.
- Regulating and preparing audits and inspections, and implementing CAPAs.
- Role to keep up-to-date with changes in pharmacovigilance legislation and practice.
- Providing training to relevant personnel about pharmacovigilance requirements, procedures, and documenting the training.
Periodic Safety Update Reports/ Periodic Benefit-Risk Evaluation Report (PSUR/PBRER)
Periodic Safety Reports are prepared by regulatory authorities with the help of routine pharmacovigilance of all the medicinal products and implementation of appropriate action as a part of a pharmacovigilance plan. Monitoring of the safety profile of approved products, including signal detection, issue evaluation, updating of labeling, and liaison with regulatory authorities.
These reports are prepared and submitted by the marketing authorization holder of an approved product. After drug approval for marketing, pharmaceutical companies are required to submit regular safety reports. It includes adverse drug reactions, risk-benefit evaluation, new safety information, and regulatory actions. These reports help regulatory authorities determine whether the drug should be sustained in the market or not. Both of these follow ICH-defined structures and are submitted according to regulatory- defined schedules and fixed deadlines.
Global Harmonization (ICH E2 Guidelines)
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) E2 series helps to set international standards for reporting and managing drug safety information throughout a product’s lifecycle. It plays an important role in ensuring global harmonization for drug safety monitoring and regulatory compliance. ICH E2 guideline is subdivided into:
- ICH E2A
It defines standards for managing and reporting adverse drug reactions (ADRs). It also defines timelines for expedited reporting during clinical development.
- ICH E2B (R2) and E2B (R3)
It covers electronic transmission of safety reports (ICSRs). They establish data fields, message formats, and the transmission of reports to regulators by a company.
- ICH E2C
It defines periodic safety update reports (PSUR)/ (PBRER) and ensures periodic benefit-risk evaluations of marketed drugs. These documents are used to assess emerging risks after approval.
- ICH E2D
It sets post- approval reporting standards for accelerated safety communications. It aligns global timelines for serious and unexpected adverse reactions.
- ICH E2E
It focuses on pharmacovigilance planning to manage identified and potential risks.
- ICH E2F
It provides a framework for annual safety updates on investigational drugs. The main focus of the guideline is on integrating data from interventional clinical trials of drugs and biological products that are under investigation.
Conclusion
Pharmacovigilance helps in the early detection of adverse effects, which ultimately improves patient safety, confidence in medications, facilitation of drug development, and optimization of drug therapy by reducing medication errors.
By continuously collecting and analyzing safety data, the detection of harm, miscommunication, poor decision-making, and disruption of patient confidentiality is prevented. Through monitoring of Adverse Drug Reaction, rare, serious, and long-term adverse effects can be detected.
The proper signal detection and its assessment are the most important aspects in pharmacovigilance. Various methods are used for the detection of signals and identification and management of drug risks using tools like the Naranjo Scale, global guidelines from ICH E2.
Overall, Pharmacovigilance guidelines support proactive safety monitoring and reinforce the culture of continuous patient protection throughout the product lifecycle. It maintains a favorable risk-benefit balance of medicines.
References
- Fornasier, G., Francescon, S., Leone, R., & Baldo, P. (2018). An historical overview over Pharmacovigilance. International Journal of Clinical Pharmacy, 40(4), 744–747. https://doi.org/10.1007/s11096-018-0657-1
- Yartsev, A. (2023, December 18). Classification of adverse drug reactions. Deranged Physiology. https://derangedphysiology.com/main/cicm-primary-exam/variability-drug-response/Chapter-320/classification-adverse-drug-reactions
- ICH. (2004). INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE PHARMACOVIGILANCE PLANNING E2E. https://database.ich.org/sites/default/files/E2E_Guideline.pdf
- Oludaiye Oluwaseyi, Oludaiye Micheal, Williams, B., & Asante, G. (2018, April 18). Pharmacovigilance and Safety Monitoring. https://www.researchgate.net/publication/391400260_Pharmacovigilance_and_Safety_Monitoring
- Health, W. (2002). The IMPORTANCE of PHARMACOVIGILANCE Safety Monitoring of medicinal products. https://iris.who.int/server/api/core/bitstreams/002b78d5-4dfc-433c-a518-f92ebdee8706/content
- Martin, J. H., & Lucas, C. (2021). Editorial: Reporting adverse drug events to the Therapeutic Goods Administration. Australian Prescriber, 44(1), 2–3. https://doi.org/10.18773/austprescr.2020.077
- Health Canada. (2008, January 24). Canada Vigilance Program. Aem. https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/canada-vigilance-program.html
- (2014). Fda.gov. https://open.fda.gov/data/faers/
- Kumar, A., & Khan, H. (2015). Signal Detection and their Assessment in Pharmacovigilance. Open Pharmaceutical Sciences Journal, 2(1). https://doi.org/10.2174/1874844901502010066
