Pauly’s Test Definition
Pauly’s test is a biochemical test for the detection of tyrosine and histidine where the reagent couples with amines, phenols, and imidazole groups. The test was discovered by and is named after the German Chemist Hermann Pauly. Pauly’s test is a specific test for proteins containing tyrosine and histidine as the reagent undergoes diazotization with aromatic groups. The reaction is performed under cold conditions as the diazonium compound can only form at cold temperatures. The test is a specific test for proteins, which is performed for further differentiation between amino acids and proteins.
Objectives of Pauly’s Test
- To detect the presence of tyrosine and histidine-containing proteins.
- To differentiate between histidine and tyrosine from other amino acids.
Principle of Pauly’s Test
Pauly’s test is based on the principle of coupling between the amino acids and the diazonium ion formed in the reagent. The pauly’s reagent consists of sulphanilic acid dissolved in concentrated hydrochloric acid. The sulfanilic acid undergoes diazotization in the presence of sodium nitrate and hydrochloric acid. The result of the diazotization reaction is the diazonium salt (p-phenyldiazosulphonate). The diazonium salt thus formed couples with histidine molecule in an alkaline condition to form a dark red or cherry colored compound. The color might decrease in intensity and changes into an orange color when the solution is made acidic. In the case of tyrosine, a similar cherry red-colored complex is formed which then changes into yellow color on dilution and bronze-yellow color on acidification. Each molecule of histidine and tyrosine reacts with two moles of diazonium compound reacts to form a mole of bis(azobenzenesulphonic acid)histidine or bis(azobenzenesulphonic acid)tyrosine.
Reaction
Sulphanilic acid + HNO2 + HCl → Diazonium salt + 2H2O
Diazonium salt + Tyrosine → Azo dye (red color)
Requirements
Reagent
- 1% Sulphanilic acid (chilled)
- 10% HCl
- 5% Sodium nitrite (chilled)
- 10% Sodium carbonate
- Chilled sample (1% tyrosine, 1% histidine)
Materials required
- Ice bath
- Vortex
- Test tubes
- Test tube stand
- Pipettes
Procedure of Pauly’s Test
- To a test tube, 1 ml of chilled sulfanilic acid is added, and a few drops of pre-chilled sodium nitrite are mixed in a vortex.
- One ml of amino acid sample is immediately added to the test tube and mixed in a vortex.
- Few drops of sodium carbonate are added to the test tube drop by drop until color begins to appear.
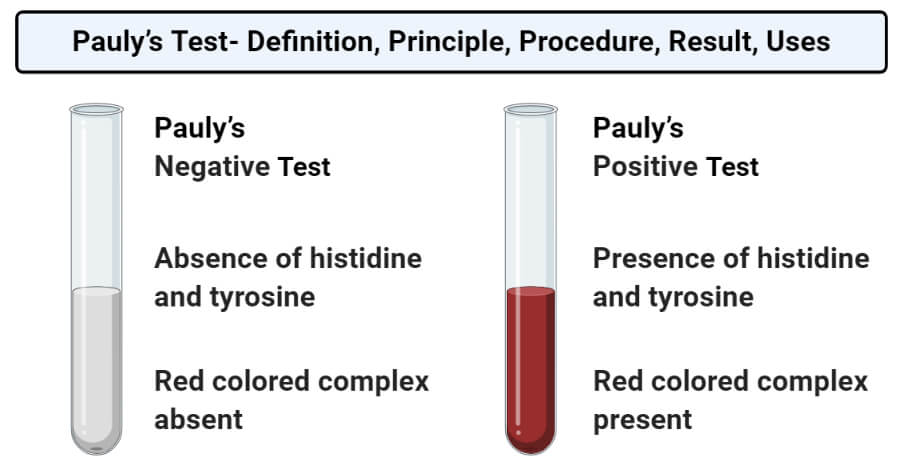
Result and Interpretation of Pauly’s Test
- Positive result: A positive result is demonstrated by the appearance of a red-colored complex, indicating the presence of histidine and tyrosine in the solution.
- Negative result: A negative result is demonstrated by the absence of a red-colored solution, indicating the absence of histidine and tyrosine in the sample.
Uses of Pauly’s Test
- Pauly’s test is used to detect the presence of tyrosine and histidine-containing proteins.
- The test also allows the differentiation of histidine and tyrosine from other amino acids.
Limitations of Pauly’s Test
- The formation of diazonium salt occurs at cold temperatures; thus, the test should be performed in the presence of ice.
- The test doesn’t allow the differentiation between histidine and tyrosine. Millon’s Test can be performed as histidine gives a negative result in Millon’s test.
References
- D (2012). Biochemistry. Fourteenth Edition. Academic Publishers. Kolkata.
- Tiwari, A. (2015). Practical Biochemistry. LAP Lambert Academic Publishing.
- https://www.onlinebiologynotes.com/paulys-test-objective-principle-reagents-procedure-and-result/