Parvovirus B19, also called the B19 virus or Human parvovirus B19, is a human virus in the Parvoviridae family. It often infects children but can also infect adults.

In healthy people, infection is usually mild, but people with blood problems or weak immune systems can have complications. Symptoms are a runny nose, fever, muscle pain, and joint pain.
Taxonomy and Classification of Parvovirus B19
Kingdom: Shotokuvirae
Phylum: Cossaviricota
Class: Quintoviricetes
Order: Piccovirales
Family: Parvovirus
Genus: Erythroparvovirus
Species: Erythroparvovirus
Synonyms: B19 virus, Erythrovirus B19, Human Parvovirus B19
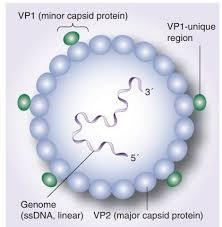
Structure and Morphology of Parvovirus B19
- It is a very small virus, measuring approximately 18-26 nm in diameter.
- It has icosahedral symmetry.
- It is non-enveloped.
- It consists of a linear, single-stranded DNA genome.
Source:https://www.creative-diagnostics.com/human-parvovirus-b19-and-parvovirus-infection.html
Genome Organization and Proteins of Parvovirus B19
- The genome is linear, single-stranded DNA of negative sense, approximately 5,600 nucleotides in length.
- Terminal structures show hairpin-shaped palindromic sequences at both ends, and they play a vital role in DNA replication.
- The genome consists of two major regions (Open Reading Frames):
Non-Structural (NS) Region
- It encodes the NS1 protein, which is necessary for viral DNA replication and cytotoxic effects on host cells.
- It helps in the replication of DNA, transcriptional regulation, and induction of apoptosis.
Structural (VP) Region
- It encodes for capsid proteins VP1 and VP2.
- VP1 possesses a unique N-terminal region that is necessary for infectivity and eliciting neutralizing antibodies.
- VP2 is the major component that comprises approximately 95% of the capsid.
Proteins
NS1 Protein
- It plays a crucial role in DNA replication, the regulation of transcription, and the induction of apoptosis in infected cells.
VP1
- It forms part of the capsid.
- The unique region of VP1 mediates receptor binding and is immunogenic.
VP2
- It is the main capsid protein, which forms about 95% of the viral shell.
VP3
- It stabilizes the capsid, which is formed post-translationally from VP2.
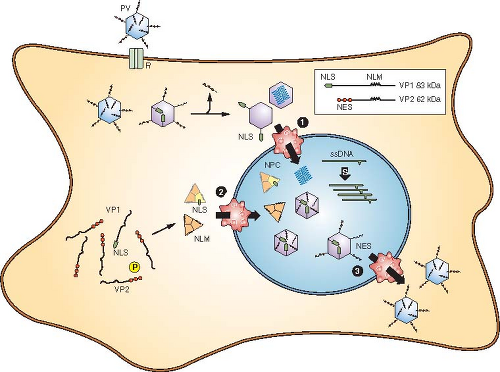
Replication Cycle of Parvovirus B19
Source: https://basicmedicalkey.com/parvoviridae
Attachment (Adsorption)
- The virus attaches to the host cells through the P antigen (globoside receptor) present on erythroid precursor cells.
- Entry of the virus is facilitated by co-receptors such as α5β1 integrin.
Entry and Uncoating
- The virus enters the cell through receptor-mediated endocytosis.
- Inside the cell, the capsid is transported to the nucleus, where uncoating occurs.
- Then the viral genome is released into the host cell.
Conversion to Double-Stranded DNA
- After that, the host cell DNA polymerase converts viral ssDNA to dsDNA.
- This step requires actively dividing cells (S phase), which is why B19 targets erythroid precursors.
Transcription
- Host RNA polymerase then transcribes viral dsDNA into mRNA.
- Multiple mRNAs are produced through alternative splicing.
Translation
- Viral mRNAs are then translated into proteins.
- In this step, NS1 is crucial for viral replication and cytotoxicity, and VP1 and VP2 form the viral coat.
DNA Replication
- Viral DNA then replicates via a rolling hairpin mechanism:
- Hairpin loops found at the end of the genome act as primers.
- The NS1 protein plays a crucial role in replication.
Assembly
- Newly synthesized viral DNA is then packaged into capsid proteins inside the nucleus.
- Assembly of capsid forms complete virions.
Release
- Mature virions are then released by cell lysis.
- This leads to the destruction of erythroid progenitor cells, causing a temporary cessation of production of RBC and clinical conditions such as aplastic crisis.
Pathogenesis and Host Immune Response of Parvovirus B19
Entry and Initial Infection
- The virus spreads through respiratory droplets, blood products, and vertical transmission.
- Then the virus enters the bloodstream, causing primary viremia.
Target Cell Tropism
- It specifically infects erythroid progenitor cells in bone marrow.
- It then binds to P antigen (globoside receptor).
Destruction of Erythroid Cells
- Viral replication causes cell lysis that results in temporary suppression of erythropoiesis and decreased reticulocyte count.
Hematological Effects
- In healthy individuals, it causes mild or asymptomatic anemia due to short-lived suppression.
- In high-risk groups, such as people with hemolytic disorders, it causes aplastic crisis, in immunocompromised individuals, it causes chronic anemia, and in people with fetuses, it causes severe anemia.
Immune-Mediated Disease
- Symptoms are mainly immune-mediated.
- Formation of immune complexes leads to erythema infectiosum and arthritis, especially in adults.
Host Immune Response of Parvovirus B19
Innate Immune Response
- Early response consists of interferon production and activation of natural killer (NK) cells.
- It helps to control initial viral replication.
Humoral Immunity
- IgM antibodies appear 7-10 days after infection.
- It indicates acute infection and aids in the clearance of viremia.
- IgG antibodies appear later and provide long-term immunity.
Cellular Immune Response
- CD8+ T cells destroy infected erythroid cells.
- It also supports viral clearance but plays a secondary role compared to antibodies.
Immune Complex Formation
- Antigen, along with antibody complexes, deposits in tissues, which cause skin rash and joint symptoms.
Immunocompromised State
- Reduced antibody production causes persistent infection.
- Continuous destruction of RBC precursors causes chronic anemia.
Epidemiology and Transmission of Parvovirus B19
Parvovirus B19 infection is widespread and can affect people of all ethnicities and races. The Parvovirus infection is commonly found in school-aged children, with peak incidence in children aged between 5 and 15 years. The prevalence of parvovirus B19 antibodies ranges from 2-10% in children younger than 5 years, 50-60% in adults older than 20 years, and 85% or more in people 70 years or older. The infection with Parvovirus B19 shows clear seasonal patterns, with more cases in late winter, spring, and early summer. Mini-outbreaks and epidemics of Parvovirus B19 infection occur cyclically approximately every 3-4 years in temperate climates. Studies from Central Europe reported a substantial increase in circulation of Parvovirus B19 after the SARS-CoV-2 pandemic, based on up to 9 months of surveillance data collected between 2012 and 2024. The United States demonstrated similar trends, although incidence remained approximately 6-fold lower than that observed in central Europe during the same time period.
Parvovirus B19 can be transmitted through several routes, detailed below:
Respiratory Transmission
- Spread via respiratory droplets formed during coughing, sneezing, and close contact.
- It occurs mainly during the viremic phase, before the rash appears.
- It is common in schools, households, and daycare centers.
Vertical Transmission
- It is transferred from the mother to the fetus.
- Virus crosses the placenta during maternal viremia.
- It can infect fetal erythroid cells, leading to severe anemia, hydrops fetalis, and fetal loss in severe cases.
Blood-borne Transmission
- It spread mainly through blood transfusion and organ transplantation.
- Risk is higher because the virus can persist in blood products.
Nosocomial Transmission
- Occurs in hospital settings through close contact with infected patients and exposure to respiratory secretions or blood.
Occupational Exposure
- Higher risk in teachers, healthcare workers, and childcare providers.
Clinical Manifestations of Parvovirus B19
In children
Erythema Infectiosum (Fifth Disease)
- “Slapped cheek” rash on the face.
- Lacy, reticular rash on trunk and limbs.
- Mild fever, malaise, and cold-like symptoms may precede rash.
Mild Systemic Symptoms
- Low-grade fever
- Fatique
- Headache
- Sore throat
In Adults
Arthropathy/ Arthritis
- Painful, symmetric joint swelling
- Can mimic rheumatoid arthritis
- Usually self-limiting within 1-3 weeks.
Mild rash and systemic symptoms similar to those of children may also occur.
In individuals with Hemolytic Disorders
Aplastic Crisis
- Sudden fall in hemoglobin level.
- Fatigue, pallor
- Shortness of breath
In Immunocompromised Individuals
Chronic anemia
- Reticulocytopenia
- Persistent viremia
In Pregnant Women
Fetal Complications
- Severe fetal anemia leads to hydrops fetalis
- Risk of miscarriage in early pregnancy.
Laboratory Diagnosis of Parvovirus B19
Sample Collection
- Blood: for antibody detection
- Bone Marrow: for the detection of severe aplastic anemia and immunocompromised patients
- Amniotic Fluid: for prenatal diagnosis
- Tissue biopsies: for immunohistochemistry
- Peripheral blood mononuclear cells: for research or specialized PCR assays
Serological Tests
- Used for the detection of antibodies like IgM and IgG in blood.
Molecular Tests
- Detects viral DNA in blood, bone marrow, or amniotic fluid.
- It is useful in immunocompromised patients and fetal infections.
- It is highly sensitive and specific.
Bone Marrow Examination
- Giant pronormoblasts with viral inclusion bodies are present.
- There is a depletion of erythroid precursors.
- It confirms the cytopathic effect of parvovirus B19.
Treatment and Antiviral Therapy of Parvovirus B19
General Treatment (Supportive Care)
- There is no specific antiviral drug routinely recommended for parvovirus B19.
- Most infections are self-limiting, especially in healthy individuals.
- Supportive measures are used, such as:
- Rest and hydration
- Antipyretics (such as Paracetamol) for fever
- NSAIDs (e.g., Ibuprofen) for joint pain
Treatment in Specific Conditions
-Aplastic Crisis
- Blood transfusion (packed RBCs)
- Close monitoring of hemoglobin and reticulocyte count
-Chronic Infection
- Intravenous immunoglobulin (IVIG) is used, which provides neutralizing antibodies and helps in the clearance of persistent infection.
- It may require repeated doses.
-Arthropathy (Adults)
- Usually self-limiting.
- NSAIDSs such as Ibuprofen, and rarely corticosteroids in severe cases, are used for the management of infection caused by Parvovirus B19.
-Infection During Pregnancy
- No specific antiviral therapy is available.
- Management is done by regular fetal monitoring (ultrasound) and, in severe fetal anemia, intrauterine blood transfusion.
No specific licensed antiviral drugs are available for Parvovirus B19. However, recently, cidofovir has been used to suppress Parvovirus B19 replication.
Prevention and Control of Parvovirus B19
General Preventive Measures
-Good Respiratory Hygiene
- Cover your mouth or nose while coughing or sneezing.
- Use masks in high-risk settings.
-Frequent hand washing with soap or the use of sanitizers.
-Avoid close contact with infected individuals, especially during the viremic phase.
Infection Control in Community Settings
-Isolate the infected person during early infection (before the rash stage).
-Encourage hygiene practices among children.
-Once rash appears, infectivity is reduced.
Protection of High-Risk Groups
-Pregnant Women
- Avoid exposure to infected individuals.
- If exposed, monitor for infection (serology/PCR), and in case of complication, a fetal ultrasound is done.
-Immunocompromised Individuals
- Reduce exposure to infected persons.
- In some cases, it may require IVIG prophylaxis.
Patients with Hemolytic Disorders
- Early detection and monitoring are done.
- Reduce exposure to infected patients during outbreaks.
Blood and Transfusion Safety
-Screening of blood products in high-risk areas.
-Use of virus-inactivated blood products where possible.
Nosocomial Control
-Use of gloves, masks, and proper washing of hands.
-Isolation in healthcare settings when possible.
Conclusion
Parvovirus B19 is a small, non-enveloped DNA virus which shows a marked tropism for erythroid progenitor cells, causing temporary suppression of production of red blood cell. While infection is usually mild and self-limiting in healthy individuals, it may cause significant complications such as aplastic crisis, chronic anemia in immunocompromised patients, and hydrops fetalis in pregnant women. The clinical manifestations are mainly immune-mediated, and diagnosis relies on serological and molecular methods. Since no specific antiviral therapy or vaccine is currently available, management is mainly supportive, with preventive measures focusing on hygiene and protection of high-risk groups.
References
- Pattison, J. R., & Patou, G. (1996). Parvoviruses. In S. Baron (Ed.), Medical Microbiology (4th ed.). University of Texas Medical Branch at Galveston. National Center for Biotechnology Information (NCBI). https://www.ncbi.nlm.nih.gov/books/NBK7715/
- Heegaard, E. D., & Brown, K. E. (2002). Human parvovirus B19. Clinical Microbiology Reviews, 15(3), 485–505. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC118081/
- News-Medical. (n.d.). Parvovirus replication. News-Medical. https://www.news-medical.net/health/Parvovirus-Replication.aspx
- Ganaie, S. S., & Qiu, J. (2018). Recent advances in replication and infection of human parvovirus B19. Frontiers in Cellular and Infection Microbiology, 8, 166. https://doi.org/10.3389/fcimb.2018.00166
- van Beers-Tas, M. H., & Heidema, J. (2013). Review: Pathogenesis of parvovirus infections in children. Virology & Mycology, 2(1), 110. https://doi.org/10.4172/2161-0517.1000110
- Vadivel, K., Nandagopal, B., Ramamurthy, M., Saravanan, N., & Rajendiran, P. (2022). An overview of the epidemiology, pathogenesis, diagnosis, and treatment of human parvovirus B19. Asian Journal of Research in Infectious Diseases, 11(4), 32–43. https://doi.org/10.9734/ajrid/2022/v11i4226
- Centers for Disease Control and Prevention. (2023, February 16). About parvovirus B19. U.S. Department of Health & Human Services. https://www.cdc.gov/parvovirus-b19/about/index.html
- Centers for Disease Control and Prevention. (2025, December 23). Parvovirus B19. https://www.cdc.gov/infection-control/hcp/healthcare-personnel-epidemiology-control/parvovirus.html
