Borrelia burgdorferi is a pathogenic spirochete that is the primary causative agent of Lyme disease, a common vector-borne illness. It is transmitted to humans through the bite of infected Ixodes ticks, particularly during their nymphal stage.
Symptoms include fever, headache, fatigue, and a characteristic skin rash known as erythema migrans. If left untreated, the disease may progress to cause arthritis, carditis, and neurological complications such as encephalopathy. This disease is particularly common in the Mid-Atlantic, Northeast, and Upper Midwest regions of the United States.
Taxonomy and Classification of Borrelia burgdorferi
Domain: Bacteria
Kingdom: Pseudomonadati
Phylum: Spirochaetota
Class: Spirochaetia
Order: Spirochaetales
Family: Borreliaceae
Genus: Borrelia
Species: B. burgdorferi
Morphology and Microscopy of Borrelia burgdorferi
- A Gram-negative, corkscrew-shaped spirochete.
- The bacteria are mostly pleomorphic and can change their morphology according to environmental conditions.
- Highly motile by peritrichous flagella.
- Approximately 10-30µm in length and 0.2-0.5µm in width.
- Non-spore-forming
- It possesses a linear chromosome approximately 950kb in size, having 28% G+C content.
- The outer membrane lacks lipopolysaccharide.
They appear as bright, motile spirochetes against a dark background in Dark-field microscopy and poorly visualized as pink-colored, corkscrew-shaped bacteria after Gram staining under the microscope.

Source: https://www.ncbi.nlm.nih.gov/books/NBK532894/figure/article-18460.image.f1/
Cultural and Growth Characteristics of Borrelia burgdorferi
- Microaerophilic
- Optimum temperature: 33˚C
- Optimum pH: 7.4-7.6
- It grows best on liquid media rather than on solid media and requires complex nutritional compounds such as serum, albumin, glucose, amino acids, and vitamins.
- On Barbour-Stornner-Kelly II (BSK II): 0.4-0.5mm, small, white disk, round, compact, restricted to surface.
Biochemical and Identification Tests of Borrelia burgdorferi
B. burgdorferi is biochemically inactive, making traditional biochemical identification unreliable. The biochemical characteristics of B. burgdorferi have not yet been studied due to its fastidious nature.
Pathogenesis and Virulence Factors of Borrelia burgdorferi
Source: https://www.mdpi.com/2076-0817/12/4/553
The pathogenesis of B. burgdorferi within Ixodes ticks involves survival, multiplication, and transmission between the tick and vertebrate hosts.
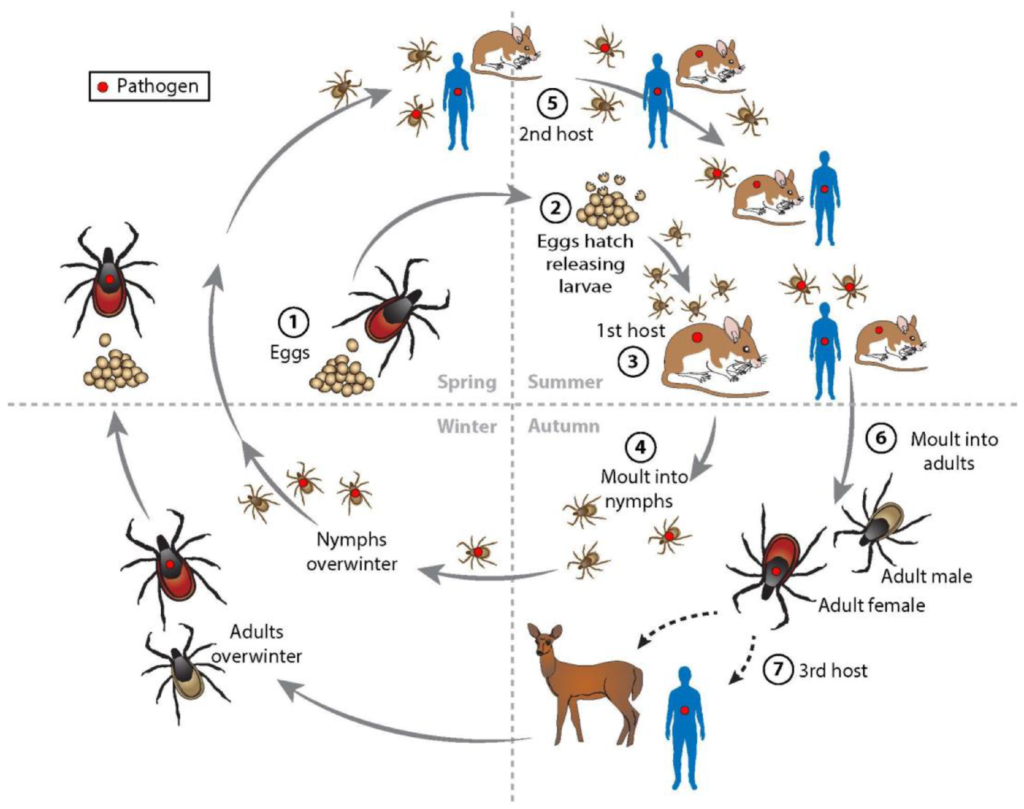
Acquisition by Tick
- Larvae or nymphs of ticks become infected when they feed on the infected reservoir hosts, such as rodents and deer.
- The spirochetes enter the midgut of the tick during the blood meal.
Survival in Tick Midgut
- In the midgut, the bacterium multiplies slowly and expresses outer surface protein A (OspA), which aids in attaching to the gut epithelium and surviving inside the tick.
- The bacterium persists in this location throughout the inter-feeding period, i.e., between blood meals.
Maintenance in Tick
- The bacterium is maintained within the tick through transstadial transmission, meaning it is passed from the larva to the nymph to the adult stage.
- There is no transovarial transmission, so larvae hatch uninfected and must acquire infection from hosts.
Activation During Blood Feeding
- When the tick feeds again, environmental changes such as temperature, pH, and blood intake activate the spirochetes.
- Then the bacteria upregulate OspC and downregulate OspA, preparing for transmission.
Migration to Salivary Glands
- The spirochetes then detach from the midgut and multiply rapidly, and then migrate through the hemocoel to the salivary glands.
Transmission to Host
- Upon prolonged feeding, i.e., more than 24-48 hours, spirochetes are released into the host through the tick saliva.
- Also, the tick saliva possesses immunomodulatory substances that help the bacteria evade host defenses.
- The bacteria express OspC, which facilitates transmission and early infection.
Local Infection of the Skin
- After entry, the bacteria multiply at the site of bite, leading to the characteristic erythema migrans (bull’s eye rash).
- Spirochetes interact with host extracellular matrix using adhesins that attach to fibronectin, collagen, and integrins.
Dissemination
- After that, the organism spreads through the bloodstream and connective tissues to various organs such as the skin, joints, heart, and nervous system.
- Motility permits penetration via tissues that lead to the involvement of the skin, causing multiple EM lesions, the nervous system, causing meningitis, cranial neuropathy, and the heart, causing Lyme carditis.
Immune Evasion Mechanisms
- Bacteria survive in the host cell via various strategies:
- Antigenic variation continuously changes surface antigens, avoiding recognition by antibodies.
- Complement evasion binds host complement regulatory proteins, hindering destruction.
- The outer surface protein switches from OspC to other OspA during later stages.
- Low immunogenicity slows down the immune response, permitting early spread.
Inflammatory Response and Tissue Damage
- The organism does not produce toxins; disease is mainly due to the host’s immune response.
- Macrophages and cytokines activation causes inflammation, which leads to skin lesions, joint inflammation, and neurological damage.
Virulence Factors
Outer Surface Proteins (OSPs)
- Proteins like OspA, OspC, and VIsE aid in transmission, adherence, and immune evasion.
Antigenic Variation (VIsE System)
- It allows the bacterium to alter its surface antigens, enabling it to evade the host immune response and persist in the body.
Flagella
- Periplasmic flagella permit corkscrew movement, helping in tissue penetration and dissemination.
Adhesins
- The surface proteins adhere to the host components like fibronectin, collagen, and integrins, promoting colonization and spread.
Immune Evasion Mechanisms
- Prevents complement activation and thereby avoids injury by immune cells, allowing B. burgdorferi to evade host immune responses and persist in the host.
- It exhibits low immunogenicity, obstructing immune recognition.
Lack of Classical Toxins
- It does not produce exotoxins and endotoxins; disease manifestations are primarily due to the host immune response and inflammation.
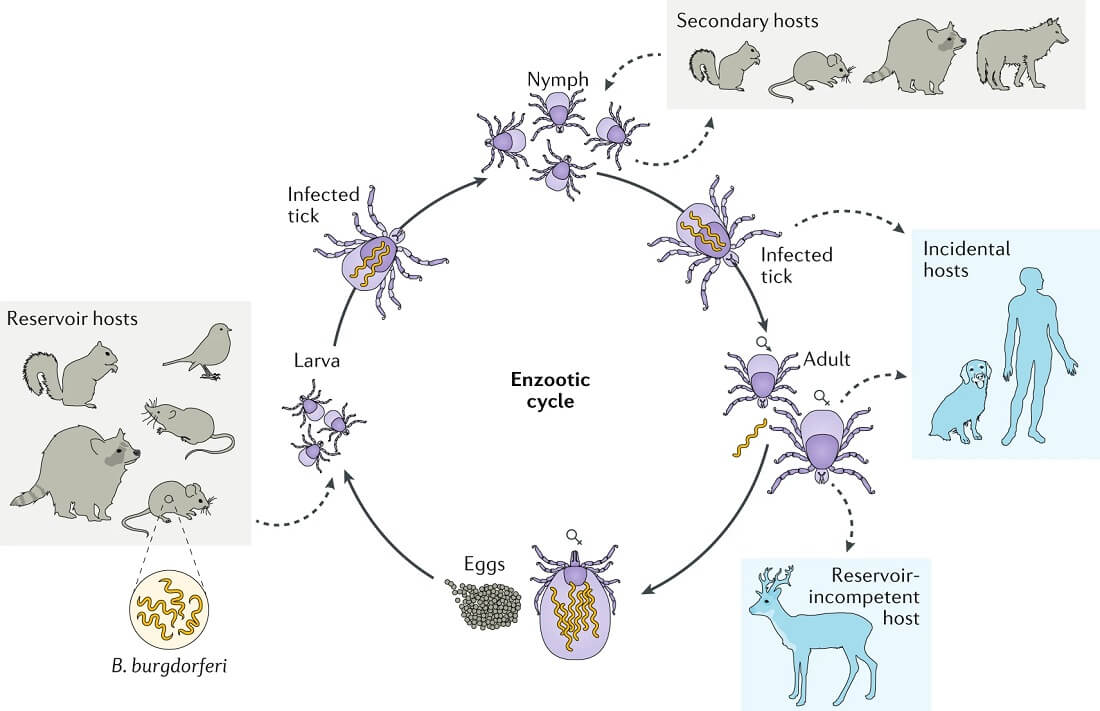
Figure: Life cycles of Ixodes scapularis and Borrelia burgdorferi. Image Source: Cheyne Kurokawa et al. 2020.
Epidemiology and Transmission of Borrelia burgdorferi
B. burgdorferi infection is ubiquitous, as it is the only species of spirochete that is the most prevalent tick-borne disease in Europe and North America. This bacterium was first identified in 1977 among children. According to the CDC, B. burgdorferi infects approximately 65,000 people every year in Europe, and around 300,000 to 400,000 cases of Lyme disease are reported in the USA every year.
In the USA, most infection associated with B. burgdorferi primarily occurs in the states across the Northeastern Seaboard and in the Midwest. Similarly, in the USA, the age distribution of infection caused by B. burgdorferi is commonly bimodal, with high prevalence in children aged 5-15 years and adults aged 45- 55 years. Also, in the USA, Lyme disease is higher in men than women in people aged below 60 years. In Canada, the disease primarily occurs in temperate regions of Central and Eastern Canada, but has been increasing in Western Canada.
Likewise, in 2019, adults aged between 55 and 79 years accounted for 46% of reported cases, while 11% of cases occurred in children aged 5-14 years. The disease onset is commonly reported in the summer and spring, linked to the peak activity of the nymphal stage of tick vectors. Also, the presence of deer is related to a rise in the number of ticks that increases the risk of infections in humans.
Transmission of B. burgdorferi occurs through various routes, such as:
-Tick Vector
- The bacterium is transmitted mainly by Ixodes ticks.
- Ticks acquire the infection when they feed on infected animal hosts, generally small mammals like mice or birds.
-Animal Reservoirs
- Wild animals, especially white-footed mice, deer, and some bird species, serve as reservoirs, maintaining the bacterium in nature.
-Human Infection
- Humans are accidental hosts.
- Infection takes place when an infected tick bites and remains adhered for at least 24-48 hours, permitting spirochetes to move from the tick’s gut to its salivary glands and into the host.
Pregnant women with untreated Lyme disease can cause placental infection, which can be transmitted to the fetus from the mother, but this is extremely rare.
Although pets infected with Lyme disease do not transmit it to humans, they may bring infected ticks into the home. So make sure to protect your pets from ticks through the use of tick-borne disease prevention products for animals.
Haunting or using a dress made from wild animals can bring you into close contact with infected ticks.
Transstadial Transmission
- The spirochetes are maintained in ticks as they progress through developmental stages: from larva to nymph to adult.
- There is no evidence of transovarial transmission, so each new tick generation must acquire the bacterium from an infected host.
Clinical Manifestations of Borrelia burgdorferi
The symptoms vary according to the stage of the disease.
Stage I (Early Localized Infection)
Symptoms appear days to weeks after a tick bite.
- The hallmark symptom: a red, expanding “bull’s-eye” rash appears at the site of the bite.
- A small red macule or papule appears, which changes to a warm, painless, annular lesion.
- A violaceous infiltration of the skin from where nodules or plaques are generated.
- A lymphocytic infiltration of the skin on the ears, head, or neck.
- A localized scleroderma
- Eosinophilic fasciitis
- Flu-like symptoms: fever, chills, fatigue, headache, myalgia, and arthralgia
Stage II (Early Disseminated Infection)
Symptoms appear weeks to months after a tick bite.
- Develop multiple erythema migrans rashes which spread to other areas and are associated with fatigue, fever, chills, malaise, and headache.
- Neurological involvement causes meningitis, cranial neuritis, radicular neuritis, encephalitis, cranial nerve palsy (commonly Bell’s palsy), and radiculopathy.
- Cardiac involvement, including atrioventricular block, palpitations, syncope, myocarditis, dyspnoea, and T wave abnormalities.
- Musculoskeletal symptoms include migratory joint pain and myalgia.
Stage III (Late or Progressive Infection)
Symptoms appear months to years after a tick bite.
- Arthritis occurs mainly in the knees, shoulders, ankles, and elbows.
- Migratory musculoskeletal pain in joints, tendons, bursae, muscle, or bone with swelling.
- Neurological symptoms include peripheral neuropathy, encephalopathy, and cognitive disturbances.
- Acrodermatitis Chronica atrophicans
- Uveitis,
- Keratitis
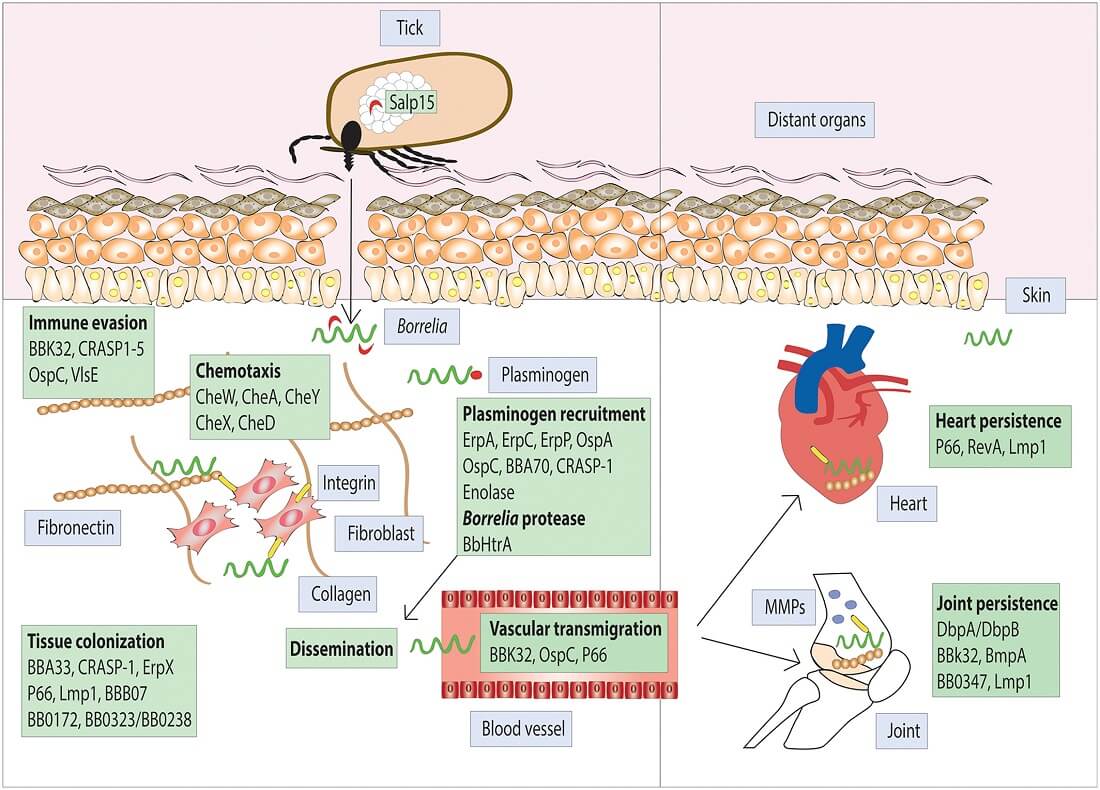
Figure: Protein interaction involved in Borrelia burgdorferi infection of mammalian hosts transmitted via Ixodes ticks. Image Source: Quentin Bernard at al. 2018.
Laboratory Diagnosis of Borrelia burgdorferi
Sample Collection and Transportation
-Samples for diagnosis of B. burgdorferi are:
- Blood (Serum)
- Skin Biopsy
- Cerebrospinal Fluid (CSF)
- Synovial Fluid
-Avoid preservatives while transporting body fluids.
-To avoid drying of the tissue biopsy, it should be placed on sterile saline.
Microscopy
- They appear as bright, motile spirochetes against a dark background in Dark-field microscopy.
- Poorly visualized as pink colored corkscrew shaped bacteria after Gram staining.
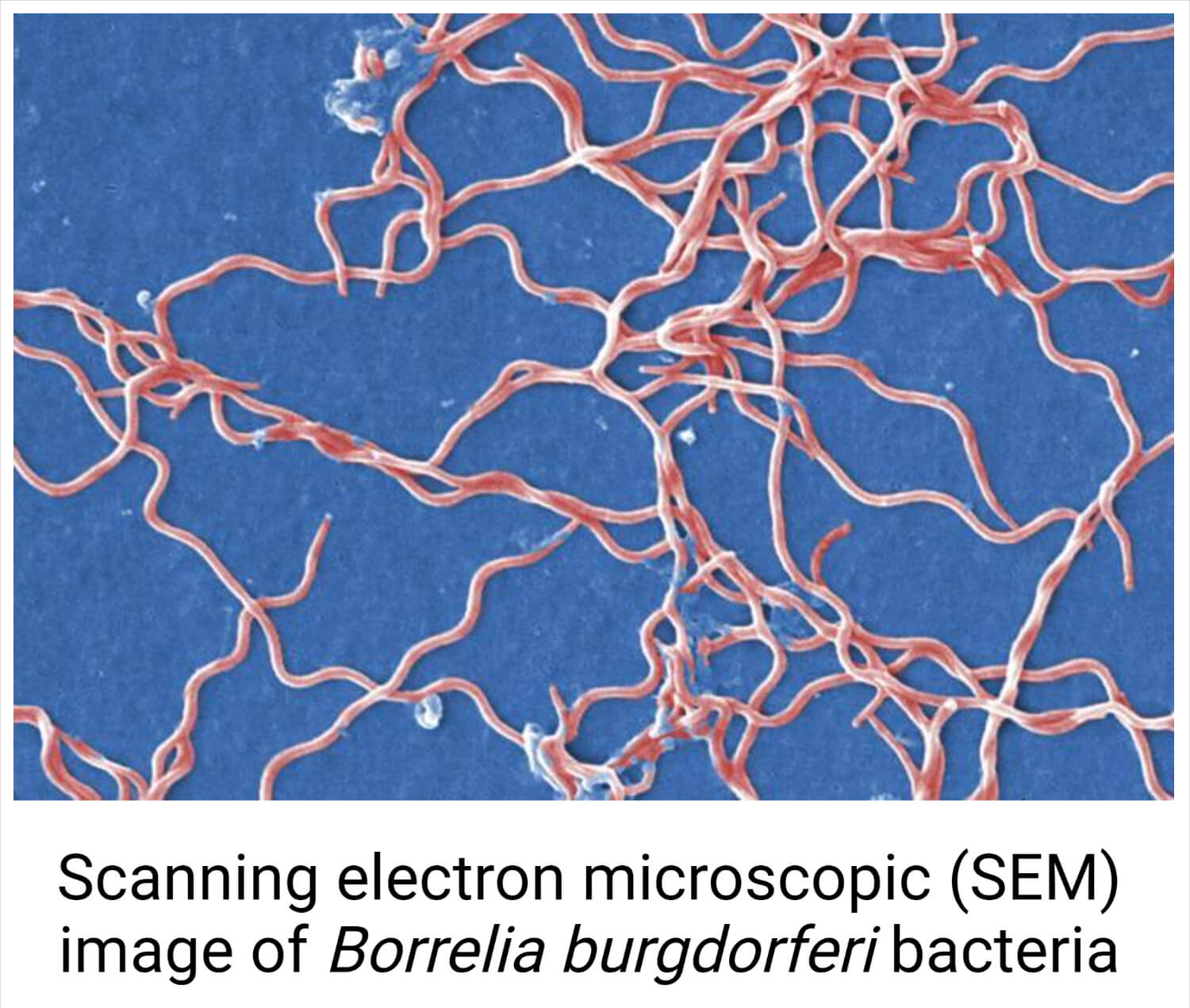
Figure: This digitally colorized scanning electron microscopic (SEM) image depicts a grouping of numerous, Gram-negative, anaerobic, Borrelia burgdorferi bacteria, which had been derived from a pure culture. Image Source: Janice Haney Carr.
Culture
- On Barbour-Stornner-Kelly II (BSK II): 0.4-0.5mm, small, white disk, round, compact, restricted to surface.
- Observed using dark-field or phase-contrast microscopy.
PCR (Polymerase Chain Reaction)
- It detects the B. burgdorferi DNA in skin biopsy, synovial fluid, and CSF.
- It is highly sensitive for early or localized infection, especially in joints or CSF.
Two-tiered Antibody Testing
ELISA
- It detects IgM and IgG antibodies against B. burgdorferi.
- It is highly sensitive but can produce false positives, so it requires confirmation.
Western Blot
- It confirms the ELISA results by detecting specific antibodies to bacterial proteins.
- IgM indicates recent infection, while IgG indicates past or ongoing infection.
Treatments of Borrelia burgdorferi
Early Localized Infection
-For general symptoms
- Amoxicillin 50 mg/kg/day is given in 3 doses.
- Doxycycline 4 mg/kg/day is given in 2 doses for 14 days.
-For erythema migrans
- Amoxicillin 50 mg/kg/day is given in 3 doses.
- Cefuroxime axetil 20-30 mg/kg/day is administered in 2 doses.
- Doxycycline 4 mg/kg/day is given in 2 doses for 14 days.
-For Lymphocytoma
- Doxycycline 4 mg/kg/day is given in 2 doses for 28 days.
-For Lymphocytic Meningitis
- Ceftriaxone 50 mg/kg/day is given intravenously in one dose.
-For Cranial neuritis
- Cefotaxime 200 mg/kg/day is given intravenously in 3 doses.
-For Cardiac Disease
- Penicillin G 0.5 million U/kg/day is given intravenously in 4 to 6 doses for 14 days.
- Doxycycline 4 mg/kg/day is given in 2 doses for 14 to 28 days.
-For Conjunctivitis
- Amoxicillin 50 mg/kg/day is given in 3 doses.
-For Arthralgia
- Doxycycline 4 mg/kg/day is given in 2 doses for 14 days.
Late Stage Infection
-For Acrodermatitis Chronica Atrophicans
- Cefotaxime 50 mg/kg/day is given intravenously in one dose.
-For Meningoradiculoneuritis
- Cefotaxime 200 mg/kg/day is given intravenously in 3 doses.
-For Encephalomyelitis
- Penicillin G 0.5 million U/kg/day is given intravenously in 4 to 6 doses.
-For Cardiomyopathy
- Penicillin G 0.5 million U/kg/day is given intravenously in 4 to 6 doses for 14 days.
-For Uvetitis, Keratitis
- Doxycycline 4 mg/kg/day is given in 2 doses for 28 days.
-For Joint and Muscle pain
- Non-steroidal anti-inflammatory drugs are given.
Prevention and Control of Borrelia burgdorferi
Personal Protective Measures
- Avoid tick-infested areas, especially wooded, grassy, and bushy environments.
- Use protective clothing like long-sleeved t-shirts, long pants, and closed-toe shoes, and tuck pants into socks to prevent ticks from reaching the skin.
- Always apply insect repellents to your skin when going out.
- If available, use permethrin-treated clothing for added protection.
Tick Management
- Check skin, scalp, and hidden areas like armpits and groin after outdoor activities.
- If ticks bite the skin, remove them promptly using fine-tipped tweezers.
Environmental Control
- Always keep grass short, remove leaf litter, and clear bushes around homes.
- Decrease exposure to animals such as rodents and deer near residential areas.
As a post-exposure prophylaxis, a single dose of Doxycycline is given after a tick bite in a Lyme disease endemic area.
Public Awareness and Education
- Educate people about the habitats of ticks and prevention strategies.
- Regular monitoring of tick populations and incidence of disease in the endemic area.
Conclusion
B. burgdorferi is a motile, spiral-shaped spirochete that is the primary cause of Lyme disease, which is transmitted to humans through Ixodes tick bites. Its virulence factors, including outer surface proteins, antigenic variation, adhesins, immune evasion mechanisms, and motility through periplasmic flagella, permit it to evade the immune system, circulate through the tissues, and survive in the host.
Infection exhibits in 3 stages, from localized bull’s eye rash to disseminated cardiac, neurological, and musculoskeletal involvement. Since culture is challenging, diagnosis mainly relies on PCR and serology. Effective antibiotic therapy leads to recovery, while preventive measures focus mainly on avoiding tick exposure and environmental control.
References
- Public Health Agency of Canada. (n.d.). Pathogen safety data sheet: Borrelia burgdorferi. Government of Canada. https://www.canada.ca/en/public-health/services/laboratory-biosafety-biosecurity/pathogen-safety-data-sheets-risk-assessment/borrelia-burgdorferi-material-safety-data-sheets-msds.html
- Meriläinen, L., Herranen, A., Schwarzbach, A., & Gilbert, L. (2015). Morphological and biochemical features of Borrelia burgdorferi pleomorphic forms. Microbiology, 161(3), 516–527. https://doi.org/10.1099/mic.0.000027
- Tatum, R., & Pearson-Shaver, A. L. (2023, July 17). Borrelia burgdorferi. In J. T. Lastname (Ed.), StatPearls [Internet]. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK532894/
- Petzke, M., & Schwartz, I. (2015). Borrelia burgdorferi pathogenesis and the immune response. Clinics in Laboratory Medicine, 35(4), 745–764. https://doi.org/10.1016/j.cll.2015.07.004
- Norris, S. J. (2012). How do Lyme Borrelia organisms cause disease? The quest for virulence determinants. The Open Neurology Journal, 6, 119–123. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3474939/
- Akoolo, L., Djokic, V., Rocha, S. C., & Parveen, N. (2021). Pathogenesis of Borrelia burgdorferi and Babesia microti in TLR4-competent and TLR4-dysfunctional C3H mice. Cellular Microbiology, 23(9), e13350. https://doi.org/10.1111/cmi.13350
- Centers for Disease Control and Prevention. (2024, September 24). How Lyme disease spreads. U.S. Department of Health & Human Services. https://www.cdc.gov/lyme/causes/index.html
- Cleveland Clinic. (n.d.). Lyme disease. Cleveland Clinic. https://my.clevelandclinic.org/health/diseases/11586-lyme-disease
- Branda, J. A., & Steere, A. C. (2021). Laboratory diagnosis of Lyme borreliosis. Clinical Microbiology Reviews, 34(2), e00018-19. https://doi.org/10.1128/cmr.00018-19
- Royal College of Pathologists of Australasia. (n.d.). Diagnostic laboratory testing for Borreliosis (Lyme disease) – Position statement. https://www.rcpa.edu.au/Library/College-Policies/Position-Statements/Diagnostic-Laboratory-testing-for-Borreliosis-Lyme
- Wormser, G. P., Dattwyler, R. J., Shapiro, E. D., Halperin, J. J., Steere, A. C., Klempner, M. S., … Nadelman, R. B. (2006). Treatment of Lyme borreliosis. Expert Review of Anti-infective Therapy, 4(1), 1–12. https://pmc.ncbi.nlm.nih.gov/articles/PMC3003502/
- Ogden, N. H. (2018). Lyme disease prevention and control: The way forward. Canada Communicable Disease Report, 44(2), 37–41. https://pmc.ncbi.nlm.nih.gov/articles/PMC5864472/
- Centers for Disease Control and Prevention. (2024, August 16). Treatment and intervention for Lyme disease. https://www.cdc.gov/lyme/treatment/index.html
