Nystatin belongs to the polyene class of antifungals. It is the first active antifungal agent.
Polyenes are broad-spectrum antifungals, biochemically characterized by a macrolide (a macrocyclic lactone ring with conjugated double bonds attached to one or more sugar moieties) and a hydroxyl group, making them amphiphilic.
- This amphiphilic nature helps them to target, bind, and disrupt the fungal cell membrane (made of ergosterol) by forming pores and cause depolarization.
- Nystatin is effective against the WHO list of critical priority fungal pathogens, including “C. albicans, Cryptococcus neoformans, Aspergillus fumigatus,” and the high-priority group, including “Histoplasma spp., C. tropicalis, C. glabrata”.
Introduction to Nystatin
Nystatin was discovered in 1950 from the fermentation product of Streptomyces noursei by chemist Rachel Fuller Brown and microbiologist Elizabeth Lee Hazen. They later identified, characterized, and purified it.
- Its generic name includes Mycostatin (oral suspension), Nystop (topical powder), and nilstat (oral drops).
- It is commonly used as a topical antifungal agent for the treatment of superficial candidiasis, e.g., vaginal candidiasis, diaper candidiasis, and oral thrush.
- Due to its low solubility, it is classified as a class IV drug of the Biopharmaceutical Classification System. This property makes its usage in the treatment of invasive fungal disease difficult.
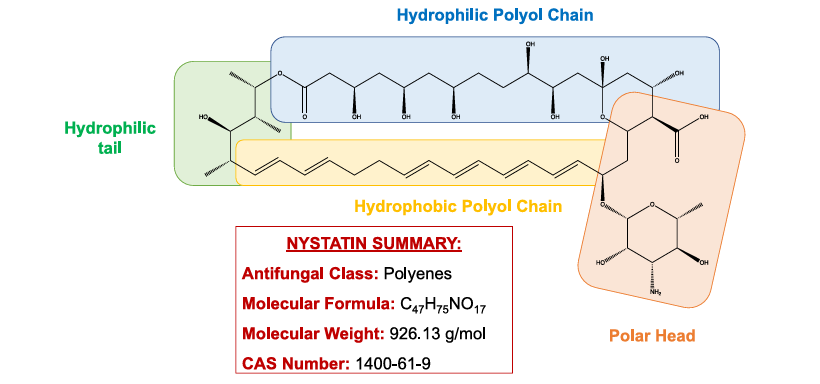
Chemical Properties of Nystatin
- Nystatin has 4 conjugated double bonds that interact with ergosterol, hence called tetraene.
- The compound is composed of a hydrophobic polyol chain and a hydrophilic polyol tail with 7 hydroxyl groups and a polar head D-deoxycosamine/mycosamine (a sugar amino responsible for antifungal properties), which is linked to a macrolactone ring via a β-glycosidic bond and a carboxylic group.
- The structure is almost similar to Amphotericin B, the differing factor being the number of conjugated double bonds (7 conjugated double bonds in Amphotericin B and 6 conjugated double bonds in Nystatin) and the position of hydroxyl groups on the hydrophilic polyol chain.
- The compound is amphoteric because of the charged carboxylic and amino groups at 7.2 to 7.4 pH.
Physical Properties of Nystatin
- The compound is light yellow in color and hygroscopic, which can be easily affected by heat and oxygen.
- The taste is bitter.
Source: doi: 10.1016/j/addr.2023.114969
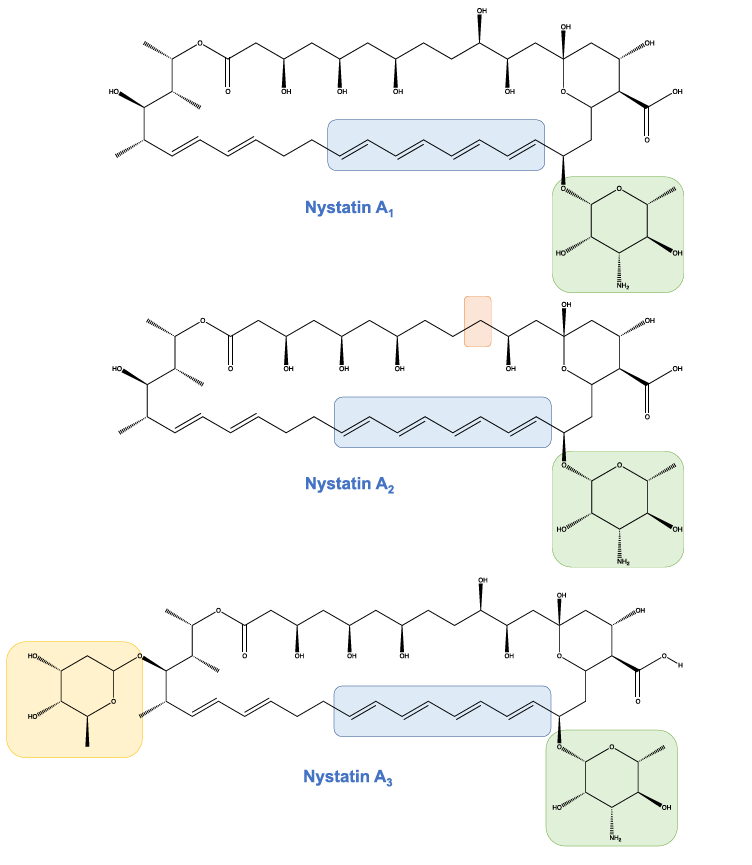
Isomer of Nystatin
Natural isomers of nystatin are a mixture of closely related products. The tetraene and N-acetyl-glucosamine are common structural similarities. These products vary in 3 forms:
- Nystatin A1 is very similar to amphotericin B.
- Nystatin A2 contains a deoxy group in C10
- Nystatin A3 contains the L-digitoxose group in a glycosidic linkage with C35.
Source: doi: 10.1016/j/addr.2023.114969
Biosynthesis of Nystatin
Nystatin is produced from S. noursei. Besides this compound, S. noursei has also yielded cycloheximide and antibiotics phalamycin and nourseothricin.
Nystatin is assembled via condensation of 16 malonyl-CoA units with 3 methylmalonyl-CoA units into a 38-carbon macrolactone ring.
The malonyl-CoA is formed via the carboxylation of Acetyl-CoA, catalyzed by the enzyme Acetyl-CoA carboxylase, and methylmalonyl-CoA is formed via the carboxylation of propionyl-CoA by the enzyme methylmalonyl-CoA carboxyltransferase.
Polyenes, including nystatin, are synthesized by Polyketide Synthase type I (module) enzyme. Polyketide synthase (PKS) type I modules contain the following domains:
- Acyl Transferase domain (AT)
- Acyl carrier protein domains (ACP)
- Keto synthase domain (KS)
Acyl Transferase domain selects chain building units (i.e, malonyl CoA and methylmalonyl CoA) and transports them to the Acyl carrier protein domain, which carries the chain building unit and transfers it to the Keto synthase domain via phosphopantetheine arm (a prosthetic group that provides a flexible arm to ACP). The keto synthase domain condenses the chain-building unit with the previous polyketide chain.
Additional catalytic domains in PKS modules include ketoreductase (KR), enoyl reductase (ER), and dehydratase (DH). These catalytic domains help determine the degree of reduction of the ketide unit. The absence of these domains leaves only the keto group on nystatin.
When the KR domain is present, it produces a hydroxyl group. The presence of the KR & DH domains yields hydroxyl groups and double bonds; the presence of all three KR, ER & DH domains produces hydroxyl groups, double bonds, and saturated bonds.
High levels of organic phosphate, glucose, and ammonia hinder the synthesis of nystatin, as these conditions prompt the cell to remain in trophophase, extinguish secondary metabolism production by blocking the polyketide synthase pathway.
After PKS modification, 3 crucial steps occur, known as post-PKS modifications, to convert the macrolactone ring into a completely active nystatin:
-Attachment of mycosamine/deoxyamino sugar at C19
-C16 methyl group oxidizes into a carboxyl group
-Hydroxylation of C10
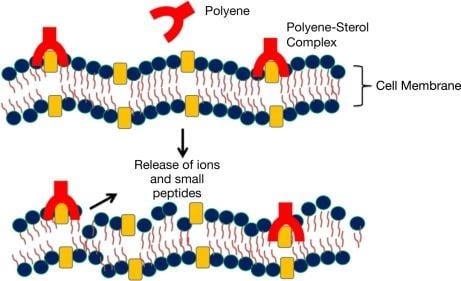
Mechanism of Action of Nystatin
- The mechanism of action is similar to that of other polyenes. The hydrophobic segment interacts selectively with fungal sterol (ergosterol) and forms a transmembrane channel/pore (size: 0.36nm). The hydrophilic polyol chain interacts with the aqueous part.
- This phenomenon disintegrates the homeostasis of the cell membrane and its permeability and leads to ion and cellular content leakage, and metabolic disruption, which induce cell death.
Source: doi: 10.1016/B978-0-12-384947-2.00363-6
- Other hypothesized models besides the pore-forming models include the sterol sponge model, the half-pore model, and the surface adsorption model.
- It is theorized that reactive oxidation stress prompts degeneration of DNA, causes lipid peroxidation and protein carbonylation, and leads to fungal cell death.
Toxicity of Nystatin
- Nystatin is less toxic than amphotericin B.
- Both oral and topical forms possess side effects like itching, rash, burning, and acute generalized exanthematous pustulosis.
- Oral form is also known to cause vomiting, diarrhea, and stomachache.
- Parenteral administration had led to infusion-related reactions and dose-limiting toxicity. The infusion-related reaction is attributed to the immunomodulatory action of nystatin through a toll-like receptor (TLR) dependent mechanism, prompting secretion of proinflammatory cytokines like IL-1β, IL-8, and TNF-α in the macrophages and immune cells.
- Nystatin, undergoing self-aggregation, has been another factor in exerting systemic toxicity. The aggregate renders the relative specificity of nystatin to bind to ergosterol. Rather, this state haphazardly prompts nystatin to target both the fungal and mammalian sterol, leading to cytotoxicity.
- The affinity of nystatin to Low-Density Lipoproteins (LDL) exerts nephrotoxicity due to the presence of LDL receptors on glomerulus endothelial cells.
Conclusion
Nystatin is a potent antifungal of the polyene class, and although it was discovered back in 1950, it is still a very relevant treatment for superficial candidiasis with a less resistance profile. Although not used now, the parenteral form causes less systemic toxicity compared to amphotericin B but is still capable of causing side effects. It has shown better prophylaxis against oral candidiasis than fluconazole for immunosuppressed people (e.g., HIV patients). Nystatin is cost-effective and more accessible than fluconazole. Further modification of this antifungal to exert low side effects paves the path for an optimal treatment option.
Reference
- Denyer, S.P., Hodges, N.A. & Gorman, S.P. (2004). Hugo and Russell’s Pharmaceutical Microbiology (7th edition, pp.). Blackwell Science
- Fjærvik, E. & Zotchev, S.B. (2005). Biosynthesis of polyene macrolide antibiotic nystatin in Streptomyces noursei. Appl. Microbiol Biotechnol, 67, 436-443. doi: 10.1007/s00253-004-1802-4
- Larson, J.L., Wallace, T.L., Tyl, R.W., Marr, M.C., Myers, C.B. & Cossum, P.A. (2000) The reproductive and developmental toxicity of the antifungal drug Nyotran (liposomal nystatin) in rats and rabbits. Toxicol. Sci., 53, 421-429.
- Lule, V.K., Garg, S., Gosewade, S.C. & Khedkar, C.D. (2016). Natamycin. In Caballero, B., Finglas, P.M. & Toldra, F., Encyclopedia of Food and Health (Vol. 4, pp. 56-62). doi: 10.1016/B978-0-12-384947-2.00363-6
- Ngece, K., Ntondini, T. L., Khwaza, V., Paca, A.M. & Aderibigbe, B.A. (2024). Polyene-Based Derivatives with Antifungal Activities. Pharmaceutics, 16, 1065. doi: 10.3390/pharmaceutics16081065
- Sousa, F., Nascimento, C., Ferreira, D., Reis, S. & Costa, P. (2023). Reviving the interest in the versatile drug nystatin: A multitude of strategies to increase its potential as an effective and safe antifungal. Advanced Drug Delivery Reviews, 199. doi: 10.1016/j/addr.2023.114969
