Marburg virus, also known as the Green Monkey Virus, is named after the West German city of Marburg. It was the first virus discovered by humans that can cause Marburg hemorrhagic fever.

It is a deadly virus responsible for causing Marburg virus disease, which is a rare but severe hemorrhagic fever. This disease can cause serious illness and even death. The virus spreads through contact with fruit bat excreta, body fluids of infected persons, and exposure in healthcare or laboratory settings.
Taxonomy and Classification of Marburg Virus
Kingdom: Orthornavirae
Phylum: Negarnaviricota
Class: Monjiviricetes
Order: Mononegavirales
Family: Filoviridae
Genus: Marburgvirus
Species: Marburg marburgvirus
Common Name: Marburg Virus
Structure and Morphology of Marburg Virus
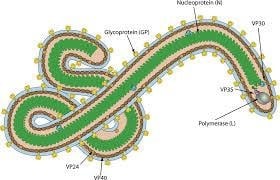
Source: https://www.antibodysystem.com/archive/72.html
- The virus body is polymorphic, exhibiting a filamentous (thread-like) appearance.
- It can be straight rods, curved, or coiled into “U” or “6” shape, or circular.
- Approximately 80 nm in diameter, and its length varies from 800 nm to 1,000 nm.
- It contains spike-like glycoproteins on the outer surface.
- Enveloped with helical nucleocapsid.
- Contains non-segmented, negative-sense single-stranded RNA genome that is about 19kb in length.
Genome Organization and Proteins of Marburg Virus
- Viral genome is a non-segmented, single-stranded negative-sense RNA molecule which encodes 7 structural proteins that are arranged linearly as:
- 3’-NP-VP35-VP40-GP-VP30-VP-L-5’
- It consists of regulatory regions that possess the following parts:
- Leader Sequence (3’ end): controls transcription and replication initiation.
- Trailer Sequence (5’ end): take part in the replication of the genome and packaging.
- Intergenic Regions: They are short sequences between genes that control transcription termination and re-initiation.
Proteins
Nucleoprotein (NP)
- It forms the helical nucleocapsid and encloses the viral RNA genome.
- It plays a crucial role in the synthesis of the genome of the virus, transcription, genome stability, and replication.
Viral Protein 35 (VP35)
- Acts as a polymerase cofactor and a potent interferon antagonist, which plays a crucial role in immune evasion by inhibiting host immune response.
- It helps in the formation of the nucleocapsid.
Viral Protein 40 (VP40)
- Acts as a major matrix protein that is responsible for virus assembly and budding from host cells.
- It links the nucleocapsid with the viral envelope.
Glycoprotein (GP)
- It is the surface protein of the virus that forms spikes on the envelope of the virus.
- It helps in attachment and entry of the virus into host cells.
- Is the primary antigen that activates neutralizing antibody responses.
Viral Protein 30 (VP30)
- It acts as a transcription activator.
- It is necessary for the synthesis of viral mRNA.
Viral Protein 24 (VP24)
- Acts as a minor matrix protein that is vital for virus assembly.
- It also contributes to immune evasion.
L (RNA-dependent RNA polymerase)
- It is the largest protein that catalyzes the transcription and replication of the RNA of the virus.
- It functions together with VP35 and VP30.
Replication Cycle of Marburg Virus
Attachment and Entry
- The viral glycoprotein (GP) binds to the host receptors such as C-type lectins, folate, TIM-1, and receptor α.
- Virus is internalized via macropinocytosis, a form of endocytosis.
Fusion and Uncoating
- After the entry into the endosome, the viral glycoprotein undergoes proteolytic cleavage by the host cathepsins.
- Acidic pH in the endosome triggers the fusion of viral and endosomal membranes, releasing the nucleocapsid into the cytoplasm.
Transcription
- The viral L protein, together with VP35 and VP30, transcribes the negative-sense RNA genome into positive-sense mRNAs.
- The newly formed mRNAs are then capped and polyadenylated by the viral polymerase.
- Then each gene is individually transcribed from the 3’ end to the 5’ end, creating seven monocistronic mRNAs.
Translation
- The viral mRNAs are then translated by the ribosomes of host cells.
- The glycoprotein is processed in the Golgi apparatus and endoplasmic reticulum before it is transferred into the plasma membrane.
Genome Replication
- After enough accumulation of viral proteins, the polymerase shifts from transcription to replication.
- A full-length antigenomic (+) RNA is synthesized, which serves as a template to produce a new negative-sense genomic RNA.
- Then the nucleoprotein binds nascent RNA to form helical nucleocapsids.
Assembly
- Nucleocapsids then move to regions of the plasma membrane that contain viral glycoprotein and matrix protein VP40.
- After that, the VP40 coordinates the packaging of nucleocapsid and recruits glycoprotein to budding sites.
Budding and Release
- After that, the virus particles bud from the host cell membrane, obtaining the lipid envelope of the host with GP spikes.
- Then the VP40 drives membrane curvature and scission, liberating infectious virus.
- The virus can cause cell death; however, release may occur before cell lysis.
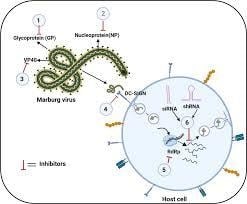
Source: https://link.springer.com/article/10.1007/s11908-023-00828-2
Pathogenesis and Host Immune Response for Marburg Virus
Transmission and Initial Infection
- Entry of the virus occurs via contact with body fluids of an infected person, such as blood, saliva, vomit, and semen.
- After the entry, initial replication takes place in monocytes, macrophages, and dendritic cells at the entry site.
- These infected cells then move to lymph nodes, expanding the virus systemically.
Systemic Spread
- Then these viruses infect endothelial cells, hepatocytes, and adrenal cortical cells.
- Endothelial cell infection can cause vascular dysfunction, increased permeability, and hemorrhage.
- Infection of hepatocytes contributes to coagulopathy by disturbing the production of clotting factors.
Cytokine Storm
- Then the infected monocytes and macrophages liberate pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β.
- This then leads to systemic inflammation, shock, and multiple organ failure.
Tissue Damage
- Widespread replication of the virus influences apoptosis in lymphocytes.
- Hepatic and renal damage cause coagulopathy and metabolic failure.
- In severe conditions, capillary leak and hemorrhage are dominant.
Host Immune Response
-Innate Immune Response
- This virus disturbs the function of dendritic cells, causing impaired antigen presentation and production of cytokines such as IL-12, IL-18, IL-10, 1L-6, IL-1β, and IFN-α.
- It obstructs the main pathways, like interferon signaling, leading to early depletion of lymphocytes.
Adaptive Immune Response
-Humoral Immunity
- Antibodies target viral glycoproteins, and production of neutralizing antibodies is usually delayed.
-Cellular Immunity
- CD8+T cells destroy infected cells; however, apoptosis of lymphocytes and immunosuppression lower the effectiveness.
Impact on NK and Invariant NKT cells
- Natural killer cells exhibit decreased cytotoxicity because of co-inhibitory signaling.
- Invariant NKT cells show restricted participation and dysfunction, further deteriorating immune control over the spread of the virus.
-Antibody Kinetics
- IgM generally appears after 4-7 days of the onset of symptoms, peaks in 1-2 weeks, and declines within a 6-month time period.
- IgG antibodies emerge simultaneously or after IgM and remain for a long time, contributing to long-term immunity.
-Proteases and Viral Pathogenesis
- Proteases like furins and cathepsins contribute to entry, exit, and pathogenesis of the virus, governing vascular leakage and modulation of innate immune response.
Epidemiology and Transmission of Marburg Virus
Marburg virus was first discovered in 1967 after infected monkeys were brought from Uganda to laboratories in Marburg (Germany) and Belgrade (former Yugoslavia). These monkeys caused the laboratory-based outbreak of Marburg Virus Disease (MVD) in both cities. There have been several outbreaks in African countries. These include Kenya (1987), the Democratic Republic of Congo (1998-2000), Angola (2004-2005), Uganda (2007, 2012, 2014, 2017), and Equatorial Guinea (2023). In 2008, the virus was detected in the Netherlands and the United States after travelers returned from Uganda and visited a popular fruit bat cave in a national park. In August 2021, the first case was reported in Guinea and West Africa. In 2022, the first confirmed case was reported from Ghana. Three cases from the same household were reported from 28 June to 5 August 2022. In 2023, the first outbreak of MVD was reported in the Kagera region, in northwestern Tanzania. This outbreak lasted for about 72 days and was declared over by Tanzania on June 1, 2023.
Transmission of the Marburg virus occurs from animals to humans (zoonotic transmission) and then between humans.
Animal-to- Human Transmission
Fruit bats of the species Rousettus aegyptiacus are the primary natural hosts. Humans can get infected through the following ways:
- Prolonged exposure to the bat-inhabited caves or mines.
- Contact with infected monkeys, especially African Green Monkeys or their tissues during laboratory processing or hunting activities.
Human-to –Human Transmission
- Occurs via direct contact (broken skin or mucus membranes) with:
- Blood, vomit, saliva, feces, or urine of an infected person.
- Contaminated materials like bedding, clothing, or medical equipment, such as needles or syringes.
-Healthcare settings are the major risk zones if control of infection is poor.
-Burial ceremonies involving direct contact with the deceased can also transmit the virus.
-Can be transmitted through sexual contact, as it can survive a long time period in the testicles.
Clinical Manifestations of Marburg Virus
The incubation period ranges from 2 to 21 days.
Early (Initial) Symptoms
- Sudden onset of high fever
- Severe headache
- Malaise (general weakness)
- Muscleaches (Myalgia)
- Chills
Gastrointestinal Phase (Day 3-5)
- Severe Watery Diarrhea
- Abdominal Pain and Cramps
- Nausea and Vomiting
- Loss of Appetite
- Patients may appear extremely weak, often described as “ghost-like”.
Hemorrhagic Phase (Severe Stage)
- Internal bleeding mainly from the gastrointestinal tract.
- Bleeding from the nose, gums, and injection sites.
- Non-itchy, maculopapular rash mainly on the trunk.
- Conjunctivitis
Systemic and Organ Involvement
- Liver Damage (elevated liver enzymes)
- Pancreatitis
- Delirium, Confusion, and Irritability
- Shock
- Multiple Organ Failure
Late Stage
- Severe hemorrhage
- Hypotension
- Disseminated intravascular coagulation (DIC)
- Death usually occurs 8-9 days after the onset of symptoms in fatal cases.
Laboratory Diagnosis
-Sample Collection
- Blood (serum or plasma): most common
- Tissue samples: in fatal cases from the liver, spleen, and lymph nodes
- Throat swabs or urine: less common
-Molecular Detection
- It is useful in the early stage of infection, which detects viral RNA.
- It is sensitive and specific, which confirms the active infection rapidly.
-Antigen Detection Tests
ELISA
- It is useful during the acute phase, which can provide quicker results in outbreak settings.
- It detects viral antigens.
-Serological Tests
Antibody Detection (ELISA)
- It detects IgM antibodies in recent infection and IgG antibodies in past exposure.
- It is more useful in later stages or in the recovery phase.
-Virus Isolation
- It provides a definitive diagnosis and is not routinely performed because of its high risk.
- The virus is isolated in cell culture.
- It requires Bio Safety Level (BSL) 4.
-Immunohistochemistry (IHC)
- It is used mainly in post-mortem diagnosis.
- It detects antigens of the virus in tissue samples.
Treatment and Antiviral Therapy of Marburg Virus
Currently, no vaccines or antiviral treatments are available for infection with the Marburg virus. However, supportive treatments are given to reduce the worsening of diseases:
Fluid and Electrolyte Management
- Oral or intravenous fluids are given.
- Electrolyte imbalance is maintained.
Hemodynamic Support
- It helps to maintain blood pressure.
- It is useful in the treatment of shock.
Oxygen and Organ Support
- Ventilation or oxygen therapy is used if necessary.
- It provides support for liver and kidney function.
Symptomatic Care
- For fever, antipyretics are used.
- For vomiting, antiemetics are used.
- Management of pain.
Management of Bleeding
- Blood Transfusions.
- Replacement of platelets.
- Treatment of coagulopathy.
Till now, no antiviral therapy is available; supportive care remains the mainstay of treatment for the disease caused by the Marburg Virus.
Prevention and Control of Marburg Virus
- Protective equipment, such as a mask, goggles, apron, and gloves, must be used when taking care of a person with MVD.
- Keep away from touching the body fluids of an infected person, and if accidentally touched, wash hands after contact, even when gloves are used.
- Avoid sexual contact with the person having MVD or use condoms while having sex.
- Do not touch any surfaces that may have been contaminated with body fluids of infected persons.
- Do not touch the bodies of patients who died from MVD, or maintain proper protection when handling a dead body.
- Avoid contact with animals such as non-human primates and fruit bats.
- Avoid going to places where fruit bats live, such as caves and mines.
- Do not consume the meat of wild animals, and if you have to eat, cook meat thoroughly before consumption.
- Travelers must be monitored quickly after returning from an area with an MVD outbreak for 21 days.
Conclusion
Marburg virus Disease is a rare but severe hemorrhagic fever caused by the Marburg virus, a member of the Filoviridae family. It presents initially with non-specific flu-like symptoms, rapidly progressing in severe cases to gastrointestinal distress, hemorrhage, multiple organ failure, and shock, with a high fatality rate. This disease spreads through contact with animals like fruit bats or a person infected with the Marburg virus. Laboratory diagnosis relies primarily on antigen detection and PCR. Till now, there is no treatment or vaccine for the disease caused by the Marburg virus; however, supportive treatment plays a crucial role in management and can significantly improve survival.
References
- Creative Diagnostics. (n.d.). Marburg virus. Retrieved March 24, 2026, from https://www.creative-diagnostics.com/marburg-virus.htm
- Abir, M. H., Rahman, T., Das, A., Etu, S. N., Nafiz, I. H., Rakib, A., Mitra, S., Emran, T. B., Dhama, K., Islam, A., Siyadatpanah, A., Mahmud, S., Kim, B., & Hassan, M. M. (2022). Pathogenicity and virulence of Marburg virus. Virulence, 13(1), 609–633. https://doi.org/10.1080/21505594.2022.2054760
- Mühlberger, E. (2017). Marburg virus reverse genetics systems. Viruses, 9(7), 178. https://doi.org/10.3390/v9070178
- AntibodySystem. (n.d.). Marburg virus. Retrieved March 24, 2026, from https://www.antibodysystem.com/archive/72.html
- Siddig, E. E., Ndembi, N., Ahmed, A., & Muvunyi, C. M. (2025). Immunogenicity, pathogenesis, and host’s immuno-responses to Marburg virus infection. Pathogens, 14(4), 323. https://doi.org/10.3390/pathogens14040323
- Africa Centres for Disease Control and Prevention. (n.d.). Marburg virus disease (MVD). Africa CDC Knowledge Hub. Retrieved March 24, 2026, from https://khub.africacdc.org/records/resource?id=168
- Cleveland Clinic. (n.d.). Marburg virus disease. Cleveland Clinic. https://my.clevelandclinic.org/health/diseases/25097-marburg-virus-disease
- Communicable Diseases Agency. (2025, October 31). Marburg virus disease. https://www.cda.gov.sg/professionals/diseases/marburg-virus-disease/
- World Health Organization. (2025, January 20). Marburg virus disease. https://www.who.int/news-room/fact-sheets/detail/marburg-virus-disease
- Institut Pasteur. (2026, January). Marburg virus disease. https://www.pasteur.fr/en/medical-center/disease-sheets/marburg-virus-disease
- National Emerging Special Pathogens Training and Education Center (NETEC). (2023, April 11). Marburg virus: Diagnostic testing, packaging, and shipping. https://netec.org/2023/04/11/marburg-virus-diagnostic-testing-packaging-and-shipping/
- World Health Organization. (2024). Diagnostic testing for Ebola and Marburg virus diseases: Interim guidance, 20 December 2024. World Health Organization. https://iris.who.int/handle/10665/380073
