The lectin pathway (LP) proceeds like the classical pathway by activating the C3 convertase. The binding of mannose-binding proteins initiates the cascade of reactions in this pathway in blood plasma called lectins to mannose-containing proteoglycans on the surface of pathogens. Pattern recognition molecules of LP other than lectins are collectins, ficolins H, L, and M.
Definition and Introduction
The lectin pathway of the complement system in the innate immune system includes a cascade of enzymatic reactions in which carbohydrate-binding proteins called lectins initiate the reaction after binding to the surface of a wide range of pathogens. The pathway is based on the activation of mannose-binding lectin-associated serine proteases (MASPs), which are soluble serine proteases of serum.
Steps, Mechanism, Process of Lectin Pathway
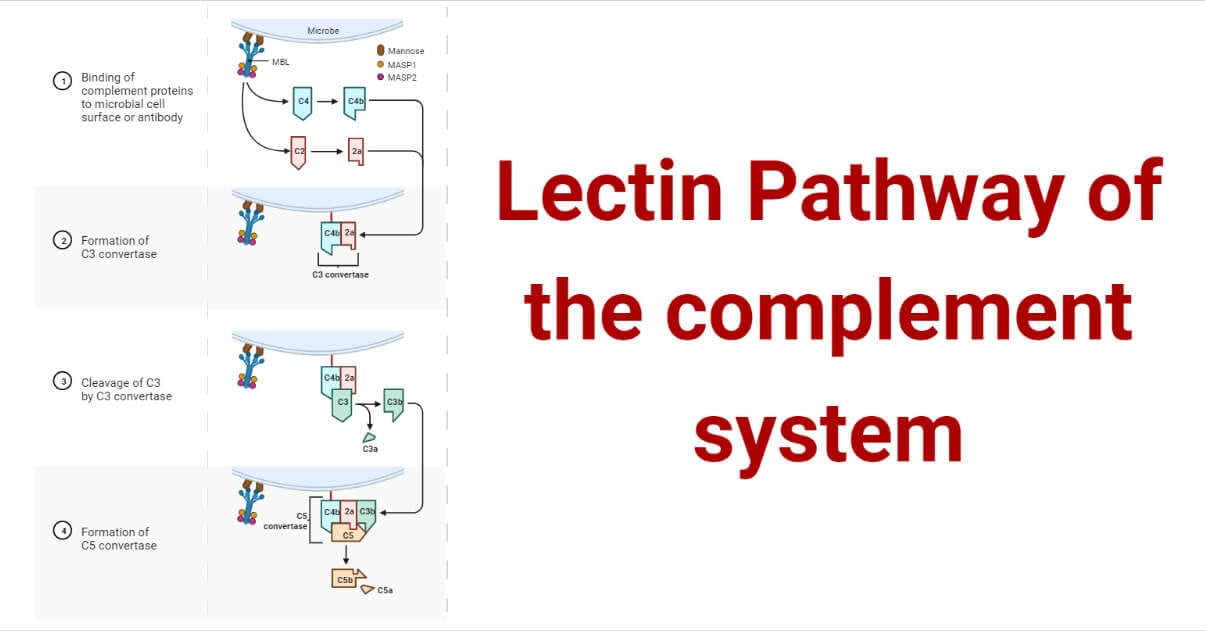
- Mannose Binding Lectin (MBL) is structurally similar to C1q (has a collagen-like region and globular heads) in the classical pathway and allows binding on many pathogens. The recognition of mannose-containing sugars on pathogens by MBL initiates a reaction. Like C1q in the classical pathway, MBL is a six-headed molecule that forms a complex with 2 protease zymogens MASP-1 and MASP-2, homologous to C1r and C1s.
Except for MBL; ficolin 1, 2, and 3 are also used as pattern recognition receptors.
- When the MBL complex binds to the pathogen’s surface, MBL-associated serine proteases MASP-1 and MASP-2 get activated, which cleave C4 and C2. MBL/ MASP-1/ MASP-2 form a trimolecular complex that activates MASPs and results in subsequent cleavage of C4.
In physiological conditions, MASP-1 is required for the cleavage of MASP-2, and both activated proteases can cleave C2, whereas MASP-2 can also cleave C4. MASP-1,2 and 3 are synthesized as proenzymes.
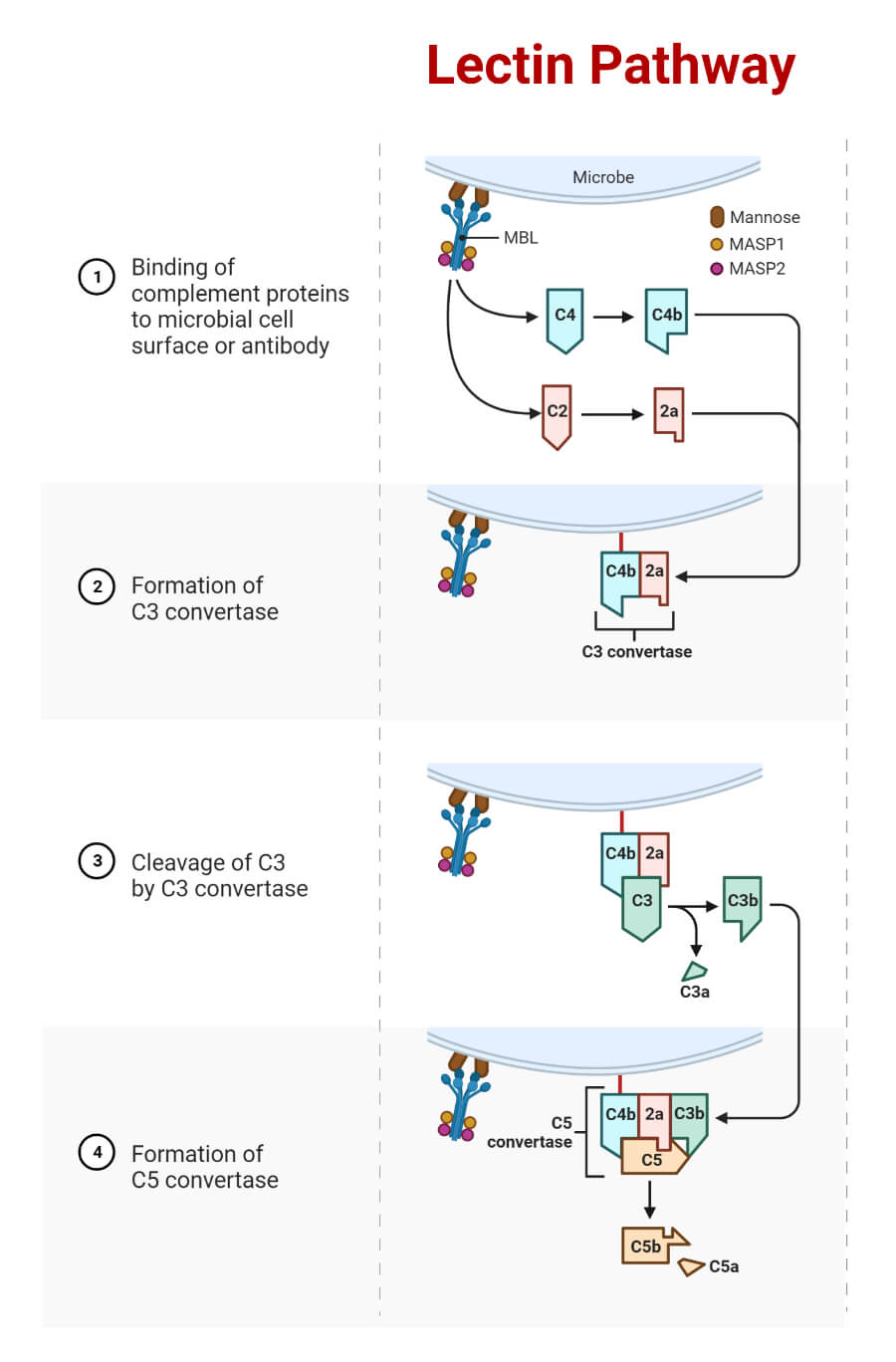
- C4 is cleaved into C4a and C4b. C4b attaches to the surface of the pathogens inducing C2 to bind, which is cleaved by MASP2 to form C2b and C2a. C2a binds to C4b, forming complex C3 convertase.
- The convertase complex, in turn, cleaves C3 to C3a and C3b. The 3a fragment is released into the fluid phase.
- This complex C4b2a3b, the C5 convertase, synthesis proceeds on the surface membrane of the pathogen.
- The membrane attack complex (MAC) is formed following the same pathway as the classical pathway of the complement system. C5 convertase cleaves C5 into subunits C5a and C5b. C5b recruits C6, C7, C8, and C9 forming MAC that induces cell lysis.
Inhibitors of Lectin Pathway
Like any other pathway of complement activation, uncontrolled activation of the lectin pathway can lead to serious damage.
Some inhibitors include C1-inhibitor, antithrombin (AT), and α(2)-macroglobulin that inhibits MASP-1 and MASP-2. Another important component Map44 (Mannose-binding lectin-associated protein of 44 Kilodalton), has been suggested to act as a competitive inhibitor of lectin pathway activation by displacing MASP-2 from MBL, hence preventing cleavage of C4 and C2.
Applications and Significance of Lectin Pathway
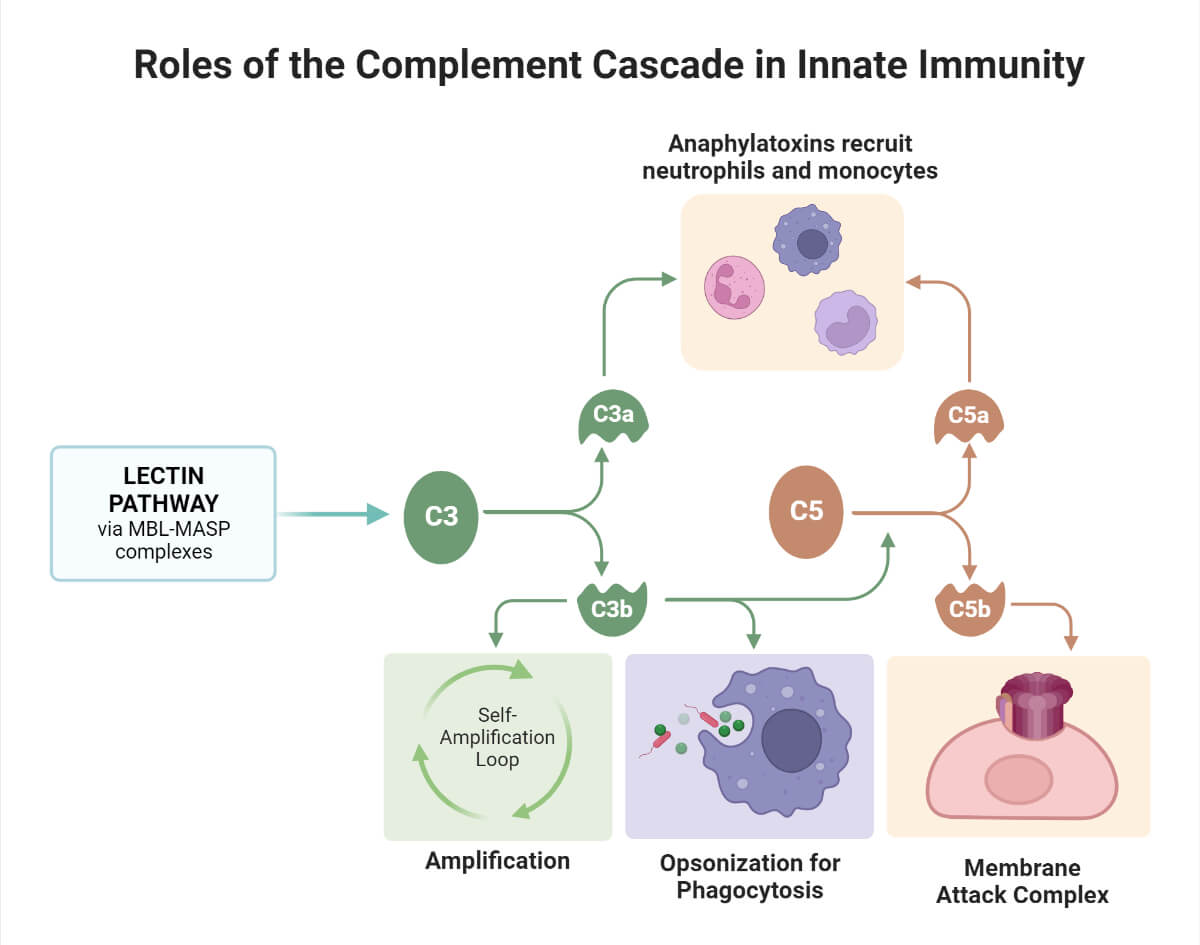
1. This pathway effectively protects against invading pathogens and apoptotic cells. Example: In dengue, this pathway has been found to prevent virus attachment to the target cells.
2. Activation of the pathway induces:
- inflammatory reactions (C5a, C4a, and C3a are important mediators of inflammation),
- opsonization (C3b, C4b, C1q)
- phagocytosis and lysis of target pathogens (via Membrane attack complex)
References
- Fujita, T., Matsushita, M., & Endo, Y. (2004). The lectin-complement pathway–its role in innate immunity and evolution. Immunological reviews, 198, 185–202. https://doi.org/10.1111/j.0105-2896.2004.0123.x
- Janeway CA Jr, Travers P, Walport M, et al. Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001. The complement system and innate immunity. Available from: https://www.ncbi.nlm.nih.gov/books/NBK27100/
- INHIBITORS OF THE LECTIN COMPLEMENT PATHWAY (LCP) AND THEIR USE – Abstract – Europe PMC
- Beltrame, M. H., Catarino, S. J., Goeldner, I., Boldt, A. B., & de Messias-Reason, I. J. (2015). The lectin pathway of complement and rheumatic heart disease. Frontiers in pediatrics, 2, 148. https://doi.org/10.3389/fped.2014.00148.

Thankyou so much. This was super helpful