Immunofluorescence is a type of assay performed on biological samples to detect specific antigens in any biological specimen or sample and vice-versa. The specificity of antibodies to their antigen is the base for immunofluorescence.
It was described in 1942 and refined by Coons in 1950, which used a fluorescence microscope able to read the specific immunological reaction and cellular slide preparations.
It is an effective method for visualizing intracellular processes, structures, and conditions as well.
- In Vitro type of Ag-Ab Interaction.
- Detects surface antigens or antibodies.
- Fluorescent dyes are used for the visualization of Ag-Ab reactions.
The property of certain dyes absorbing light rays at one particular wavelength (ultraviolet light) and emitting them at a different wavelength (visible light) is known as fluorescence. In the immunofluorescence test, a fluorescent dye that illuminates in UV light is used to detect/show the specific combination of an antigen and antibody. The dye usually used is fluorescein isothiocyanate, which gives yellow-green fluorescence. Immunofluorescence tests are also termed fluorescent antibody tests (FAT).
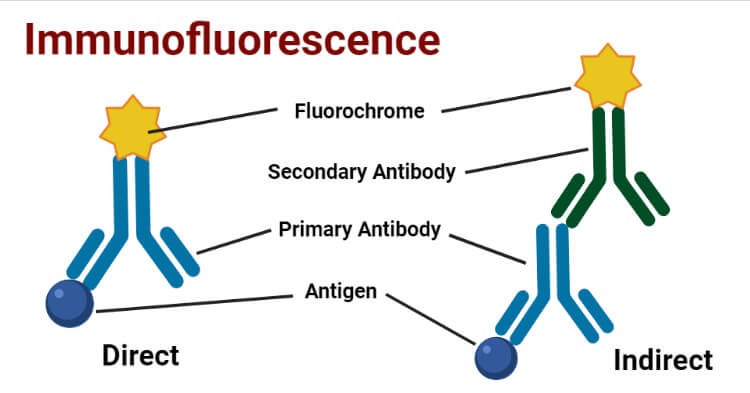
Requirements of Immunofluorescence
The primary requirement is specific antibodies that can bind to the antigen of interest to form the Ag-Ab complex. They can be :
a. Primary Antibody: The specific antibody which directly binds with antigen.
b. Secondary Antibody: The antibody which binds to the Fc region of a primary antibody that is already bound with the specific antigen. It can be effectively used for different types of assays.
A secondary requirement is Fluorescent dye or Fluorchromes or Fluorophores which are conjugated to the antibody. Commonly used Fluorochromes are:
- Fluorescein
- Rhodamine
- Phycoerythrin
– Immunofluorescence microscope for visualization
– Wash buffers such as PBS ( Phosphate Buffered Saline ): Helps to was away unbound antibodies.
Principle of Immunofluorescence
- Specific antibodies bind to the protein or antigen of interest.
- Antibodies could be labeled with molecules that have the property of fluorescence (fluorochromes)
- When light of one wavelength falls on fluorochrome, it absorbs that light to emit light of another wavelength.
- The emitted light can be viewed with a fluorescence microscope.
Types of Immunofluorescence
- Direct Immunofluorescence Test
- Indirect Immunofluorescence Test
Direct Immunofluorescence Test
Single antibody i.e. primary antibody is used that is chemically linked to a fluorochrome. If the antigen is present, the primary antibody directly reacts with it and fluorescence can be observed under the fluorescent microscope.
Procedure of Direct Immunofluorescence Test
- Fixing of Specimen (Antigen) into the slide.
- Fluorochrome labeled antibodies are then added to the slide.
- Incubation and careful washing with wash buffers like PBS to remove other components except for the complex of antigen and fluorochrome-labeled antibody.
- Observed under a fluorescence microscope.
Uses of Direct Immunofluorescence Test
- For the detection of rabies virus antigen in the skin smear collected from the nape of the neck in humans and the saliva of dogs.
- For the detection of N. gonorrhoeae, C. diphtheriae, T. pallidum, etc. directly in appropriate clinical specimens.
Advantages of Direct Immunofluorescence Test
- Protocols for direct IF are usually shorter as they only require one labeling step.
- Species cross-reactivity is minimized indirect methods as the fluorophore is already conjugated to the primary antibody.
Disadvantages of Direct Immunofluorescence Test
- Separately labeled antibodies need to prepared for each pathogen.
- Requires the use of a much more primary antibody, which is extremely expensive.
- Less sensitive than indirect immunofluorescence.
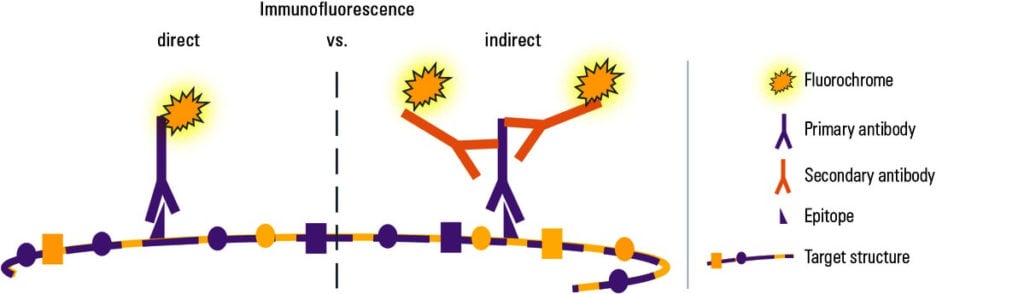
Indirect Immunofluorescence Test
Double antibodies are used i.e. primary and secondary antibodies. The primary antibody is not labeled and a fluorochrome-labeled secondary antibody is used for detection. The antigen used is known and it binds to the specific primary antibodies of interest in the sample. The secondary antibody then binds to the Fc region of the primary antibody.
Procedure of Indirect Immunofluorescence Test
- Fixing of a known antigen on a slide.
- The specimen to be tested is applied to the slide.
- Incubation and careful washing with PBS.
- A secondary antibody (e.g., fluorescently labeled anti-IgG) is added.
- Incubation and careful washing again with PBS.
- Observed under the fluorescence microscope.
Uses of Indirect Immunofluorescence Test
- In detection of specific antibodies for diagnosis of syphilis, amoebiasis, leptospirosis, toxoplasmosis, and other diseases.
- Also used in the detection of autoantibodies that cause auto immune disorders.
Advantages of Indirect Immunofluorescence Test
- In case of secondary antibodies, a single fluorochrome-labeled antibody is used for detecting many Ag-Ab interactions.
- More sensitive than direct immunofluorescence test.
- Multiple secondary antibodies can bind to the Fc region of primary antibody which amplifies the fluorescence signal.
Disadvantages of Indirect Immunofluorescence Test
- It is more complex and time-consuming than the direct IF.
- Cross-reactivity of secondary antibody to other agents can be problematic.
Result interpretation of Immunofluorescence
If there is the presence of a specific antigen or antibody of interest they would form an Ag-Ab complex. So the fluorochrome-conjugated antibody will remain bound in the preparation even after washing and fluorescence of yellow-green or green or red ( depending on the types of fluorochromes used ) can be observed while visualizing through a fluorescent microscope. And the test can be considered positive.
If there is no presence of antigen or antibody of interest then Ag-Ab complex won’t be formed and all the unbound antibodies would be washed away hence we cannot observe fluorescence if the test is negative.
Applications of Immunofluorescence
- Immunofluorescence can be used on tissues or cell sections to determine presence of different biological molecules which also includes proteins, carbohydrates, etc.
- Also used in molecular biology for visualization of cytoskeletons such as intermediate filaments.
- It also plays a key role in the detection of autoimmune disorders.
- It can be used with some non-antibody methods of fluorescent staining, like the use of DAPI (4′,6-diamidino-2-phenylindole ) to label DNA.
Limitations of Immunofluorescence
- The main problem can be photobleaching i.e. degradation of fluorochromes. It can be prevented by using higher concentration of flurochromes and decreasing exposure time to the light.
- Extraneous unnecessary fluorescence can occur due to impurity of targeted antigen.
- Autofluorescence can occur due to some agents which bear the property of fluorescence in the given specimen.
- It is mostly used for only fixed cells or dead cells.
- Expensive and require higher expertise.
What is Flow Cytometry?
Flow cytometry is the advancement of the immunofluorescence technique as it is used for qualitative as well as quantitative analysis of antigen and antibodies present in the sample. In flow cytometry, laser beam and light detector are used to count single intact cells in suspension. It is a computer-based advanced technology. International Society for Advancement of Cytometry is an organization for flow cytometry. (https://isac-net.org/)
References
- Betterle C, Zanchetta R. The immunofluorescence techniques in the diagnosis of endocrine autoimmune diseases. Auto Immun Highlights. 2012;3(2):67-78. Published 2012 Jun 6. doi:10.1007/s13317-012-0034-3.
- Goldsby R.A., Kindt T.J., Osborne B.A., (1999) Kuby Immunology, 4th edition, W.H.Freeman & Co Ltd., pg. 152-155.
- Parija S.C., (2009), Textbook of Microbiology and Immunology, 2nd edition, Elsevier, a division of Reed Elsevier India Private Limited, pg. 111-112.

Makes for an easy read. Thanks
awesome article!! this really cleared up a lot of questions after going through lecture about this topic.
Hi, what are the parameters of immunoassay?
This is incredible. I sometimes wonder why we make things so difficult yet they can be summarised
Thank you. It’s written by me. Felt happy to see this comment.
Awsome explainance.. I completely understood it…… It’s awesome nd incredibly easy