Hantavirus is a rodent-borne zoonotic virus that causes severe human illnesses, primarily hantavirus cardiopulmonary syndrome (HCPS) and hemorrhagic fever with renal syndrome (HFRS).
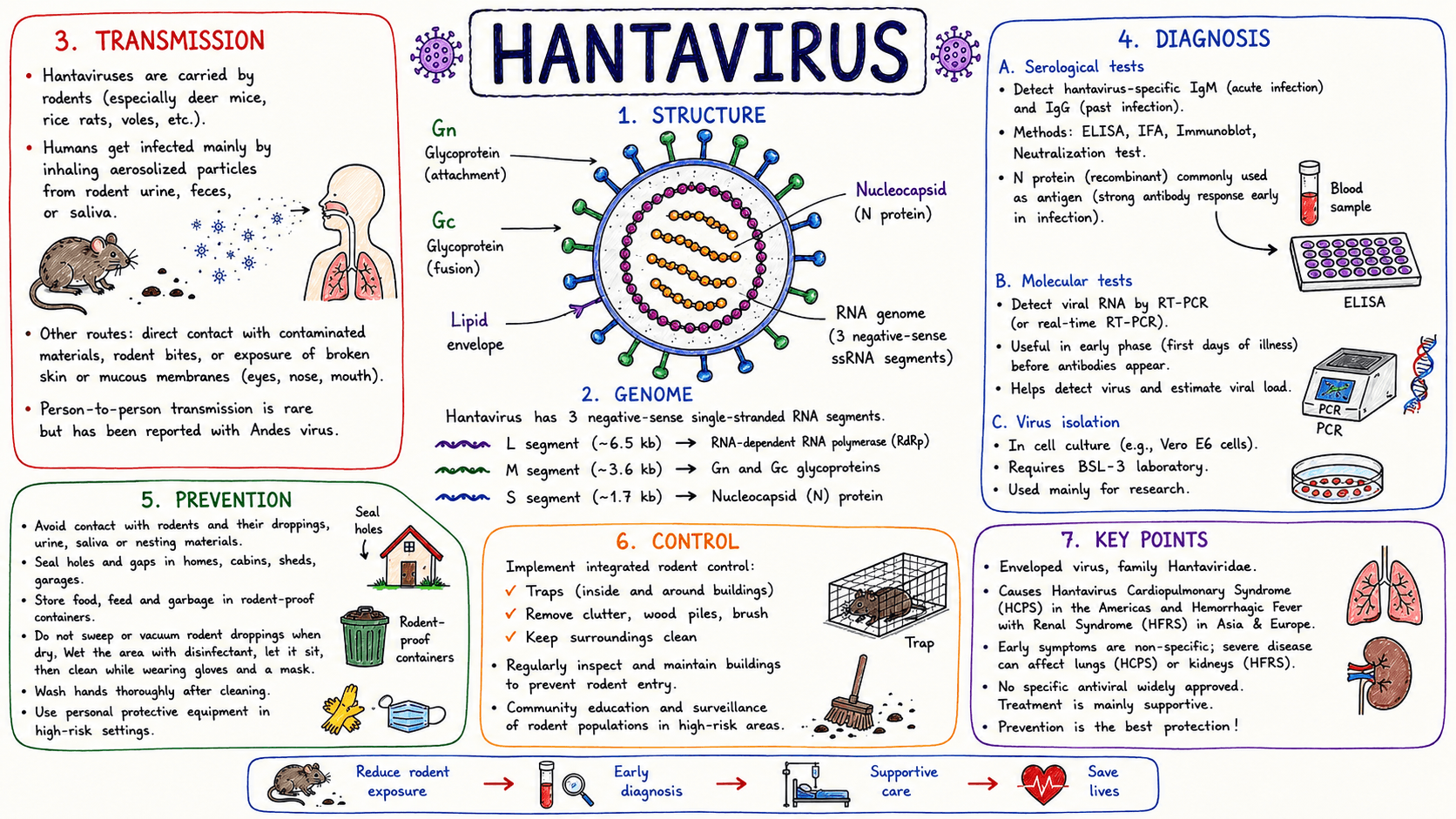
Structure of Hantavirus
- Hantaviruses are currently classified under the family Hantaviridae, order Bunyavirales.
- The virions are generally spherical to pleomorphic, enveloped particles measuring approximately 80-160 nm in diameter, although size may vary among species.
- The viral envelope consists of a host-derived lipid bilayer embedded with two surface glycoproteins, Gn and Gc, which form characteristic spike-like projections involved in host cell attachment and entry.
- Hantaviruses possess a helical ribonucleoprotein nucleocapsid enclosing a tri-segmented, negative-sense single-stranded RNA genome consisting of:
- L segment encoding RNA-dependent RNA polymerase (RdRp)
- M segment encoding envelope glycoprotein precursor (Gn and Gc)
- S segment encoding nucleocapsid protein
- The ribonucleoprotein complexes are filamentous and arranged within the virion interior.
- Unlike some other bunyaviruses, hantaviruses generally lack a matrix protein, and virion organization depends largely on glycoprotein-capsid interactions.
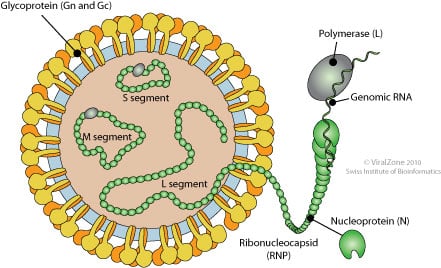
Genome of Hantavirus
- Hantaviruses possess a tri-segmented, single-stranded, negative-sense RNA genome consisting of the small (S), medium (M), and large (L) segments.
- The L segment encodes the RNA-dependent RNA polymerase (RdRp), which is responsible for viral genome replication and transcription.
- The M segment encodes a glycoprotein precursor, which is post-translationally cleaved into the two envelope glycoproteins, Gn and Gc, involved in host cell attachment and membrane fusion.
- The S segment encodes the nucleocapsid (N) protein, which binds viral RNA to form ribonucleoprotein (RNP) complexes.
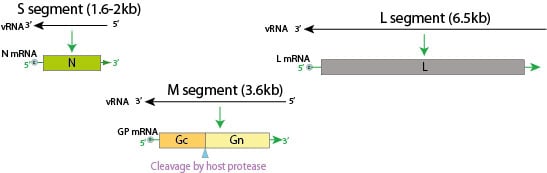
- In some hantavirus species, an additional non-structural protein (NSs) may also be encoded, typically through an overlapping open reading frame in the S segment.
- The genomic RNA segments are encapsidated by the nucleocapsid protein to form helical ribonucleoprotein complexes, which are enclosed within a host-derived lipid envelope studded with Gn and Gc glycoprotein spikes.
Epidemiology and Transmission of Hantavirus
- Hantaviruses associated with HFRS were recognized decades earlier, particularly following outbreaks among United Nations forces during the Korean War. However, the 1993 outbreak in the Four Corners region of the southwestern United States led to the identification of Sin Nombre virus, the major cause of HCPS in North America.
- HCPS occurs mainly in the Americas, including Canada, the United States, Argentina, Brazil, Chile, and Panama. HFRS is reported predominantly in China, Russia, and the Republic of Korea, with additional cases in Scandinavia and parts of Western and Central Europe.
- A notable outbreak occurred in 2012 at Yosemite National Park, where exposure to infected deer mice resulted in multiple HCPS cases.
- Recent infections continue to highlight the public health significance of hantaviruses. Sporadic HCPS cases continue to be reported in the Americas, while HFRS remains endemic in several Asian and European countries.
- In 2026, an outbreak of the Andes virus among expedition cruise passengers prompted international public health monitoring.
Transmission of Hantavirus
- Hantaviruses are primarily transmitted to humans through exposure to infected rodents and their excreta.
- The most common route of transmission is the inhalation of aerosolized particles contaminated with rodent urine, feces, or saliva.
- Transmission may also occur through:
- direct contact with infected rodents or contaminated materials
- rodent bites
- exposure of broken skin to infectious materials
- contact of contaminated materials with the mucous membranes of the mouth, nose, or eyes
- Most hantavirus infections are not transmitted from person to person. However, limited human-to-human transmission has been documented with the Andes virus, particularly in South America.
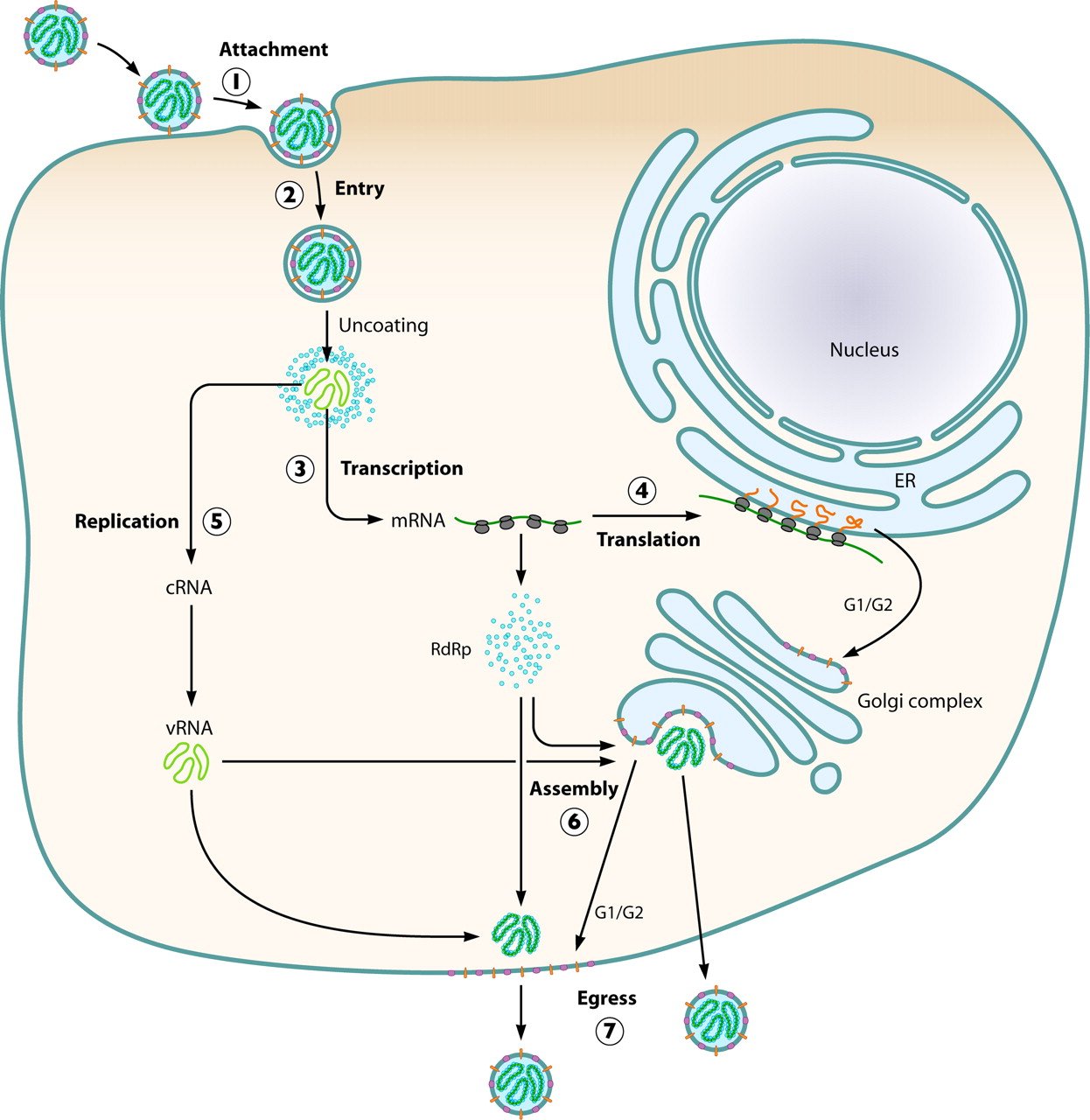
Replication of Hantavirus
- Hantavirus replication occurs primarily in the cytoplasm of the infected host cell.
- The virus attaches to host cell surface receptors through its envelope glycoproteins, Gn and Gc, which mediate viral attachment and entry.
- Following attachment, the virus enters the host cell through receptor-mediated endocytosis, which may involve clathrin-dependent, caveolin-dependent, or alternative endocytic pathways depending on the hantavirus species and host cell type.
- Within endocytic vesicles, acidification triggers conformational changes in the viral glycoproteins, leading to fusion of the viral envelope with the endosomal membrane and release of the viral ribonucleoprotein complexes into the cytoplasm.
- The viral RNA-dependent RNA polymerase (RdRp) initiates transcription of the negative-sense RNA genome to produce viral mRNAs using a cap-snatching mechanism, in which short capped primers are derived from host mRNAs.
- The viral mRNAs are translated by host ribosomes to produce viral proteins, including nucleocapsid protein, glycoproteins, and RNA polymerase.
- Genome replication begins when sufficient nucleocapsid protein is available to encapsidate newly synthesized complementary positive-sense RNA intermediates (antigenomes), which then serve as templates for the synthesis of new negative-sense genomic RNA.
- The viral glycoproteins are processed in the endoplasmic reticulum and Golgi apparatus, where assembly of progeny virions occurs with newly synthesized ribonucleoprotein complexes.
- Mature virions are transported in vesicles and released from the host cell through exocytosis.
Pathogenesis of Hantavirus
- Humans acquire hantavirus infection primarily through inhalation of aerosolized particles contaminated with infected rodent urine, feces, or saliva. After entry into the body, the virus initially infects host cells in the respiratory tract and subsequently disseminates through the bloodstream to target organs.
- The incubation period of hantavirus infection is typically 1-8 weeks, depending on the viral species and host factors.
- Hantaviruses primarily infect endothelial cells, which line the interior surface of blood vessels, without causing marked direct cytopathic damage. Instead, disease manifestations result largely from increased vascular permeability, platelet dysfunction, and dysregulated immune responses.
- A key feature of hantavirus infection is the interaction of viral glycoproteins with host cell receptors, particularly β3 integrins, which are expressed on endothelial cells and platelets. This interaction contributes to altered endothelial barrier integrity and impaired platelet function.
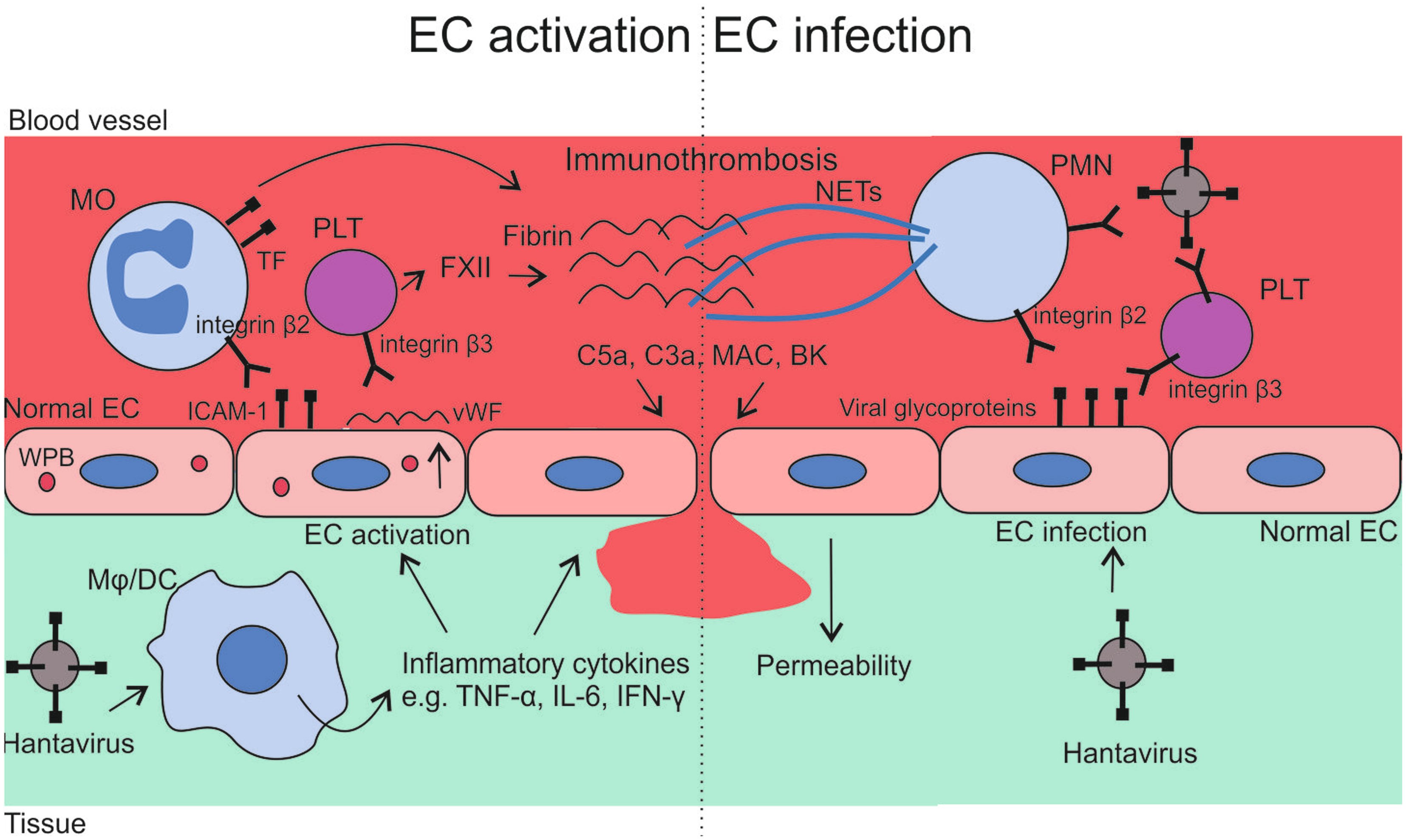
- Following infection, the virus spreads to different organs depending on the hantavirus species. Viruses causing hantavirus cardiopulmonary syndrome (HCPS) primarily affect the lungs, leading to pulmonary capillary leakage, pulmonary edema, hypotension, and respiratory failure. Viruses causing hemorrhagic fever with renal syndrome (HFRS) show greater tropism for the kidneys, resulting in renal vascular leakage, thrombocytopenia, and acute kidney injury.
- The host immune response plays a major role in disease severity. Infection of endothelial cells stimulates the release of inflammatory mediators and chemokines, which recruit immune cells such as CD8+ T lymphocytes, macrophages, and monocytes to infected tissues.
- These immune cells release pro-inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interferon-gamma (IFN-γ), which further increase vascular permeability and tissue inflammation. Excessive immune activation may contribute to severe disease manifestations, sometimes described as an immunopathogenic cytokine-mediated response.
- Overall, the hallmark of hantavirus pathogenesis is capillary leakage syndrome caused primarily by immune-mediated endothelial dysfunction rather than direct viral destruction of infected cells.
Clinical Manifestations of Hantavirus
The clinical manifestations of hantavirus infection vary depending on the viral species and the syndrome caused, primarily hantavirus cardiopulmonary syndrome (HCPS) and hemorrhagic fever with renal syndrome (HFRS).
Hantavirus Cardiopulmonary Syndrome (HCPS)
HCPS typically begins with a prodromal phase lasting several days, characterized by non-specific flu-like symptoms, including:
- fever
- fatigue
- myalgia, especially in the thighs, hips, and back
- headache
- chills
- dizziness
- nausea
- vomiting
- diarrhea
- abdominal pain
As the disease progresses to the cardiopulmonary phase, patients develop:
- cough
- shortness of breath (dyspnea)
- rapid breathing (tachypnea)
- pulmonary edema due to capillary leakage
- hypotension
- tachycardia
- respiratory failure
- cardiogenic shock in severe cases
The convalescent phase occurs in recovering patients and may last for weeks to months, with gradual improvement in respiratory and cardiovascular function.
Hemorrhagic Fever with Renal Syndrome (HFRS)
HFRS typically develops after an incubation period of 1-8 weeks and classically progresses through several clinical phases.
1. Febrile phase
Initial symptoms appear suddenly and may include:
- high fever
- severe headache
- back pain
- abdominal pain
- chills
- nausea
- vomiting
- blurred vision
- facial flushing
- conjunctival injection
- petechial rash in some patients
2. Hypotensive phase
As the disease progresses, patients may develop:
- low blood pressure
- vascular leakage
- thrombocytopenia
- shock in severe cases
3. Oliguric phase
Renal involvement becomes prominent, with:
- reduced urine output
- acute kidney injury
- proteinuria
- fluid overload
- electrolyte imbalance
4. Diuretic phase
Patients may experience increased urine output as renal recovery begins.
5. Convalescent phase
Gradual clinical recovery occurs, although renal function may take time to normalize.
Disease severity varies depending on the infecting hantavirus species, host immune response, and timeliness of supportive medical care.
Laboratory Diagnosis of Hantavirus
Laboratory diagnosis of hantavirus infection relies primarily on serological and molecular methods, while virus isolation is used mainly for research purposes.
1. Virus Isolation
Hantavirus can be isolated in cell culture, most commonly using Vero E6 cells (African green monkey kidney cells). However, virus isolation requires specialized laboratory facilities, trained personnel, and biosafety level 3 (BSL-3) containment because of the infectious nature of the virus.
Virus isolation has been performed from infected rodent tissues such as lung and liver, and less commonly from human clinical samples. Due to technical difficulty and biosafety concerns, virus isolation is not routinely used for clinical diagnosis.
2. Serological Diagnosis
Serological testing is the most commonly used approach for diagnosing hantavirus cardiopulmonary syndrome (HCPS) and hemorrhagic fever with renal syndrome (HFRS).
Detection of hantavirus-specific IgM antibodies indicates recent infection, while detection of IgG antibodies or a rising antibody titer in paired sera supports current or past infection.
Common serological methods include:
- enzyme-linked immunosorbent assay (ELISA)
- indirect immunofluorescence assay (IFA)
- immunoblot assays
- neutralization tests
Recombinant viral antigens, particularly the nucleocapsid (N) protein, are commonly used because the N protein is highly immunogenic and induces a strong antibody response early in infection.
3. Molecular Diagnosis
Molecular diagnosis is based on the detection of hantavirus RNA in clinical specimens using reverse transcription polymerase chain reaction (RT-PCR).
Clinical samples may include:
- blood
- serum
- plasma
- tissue specimens in severe or fatal cases
Real-time RT-PCR (qRT-PCR) provides rapid and sensitive detection of viral RNA and may also help estimate viral load in some settings.
Molecular testing is particularly useful during the early acute phase, before antibody responses become fully detectable.
4. Combined Diagnostic Approach
A combination of serological testing (especially IgM ELISA) and RT-PCR provides the most reliable laboratory diagnosis, particularly during acute infection.
Differential Diagnosis
Because hantavirus infection may present with non-specific febrile illness, differential diagnosis should include:
- leptospirosis
- dengue fever
- influenza
- COVID-19
- bacterial sepsis
- other viral hemorrhagic fevers (where geographically relevant)
Treatment of Hantavirus
There is no universally approved specific antiviral therapy for hantavirus infection, and treatment is primarily supportive.
Supportive management includes:
- careful monitoring and correction of fluid and electrolyte balance
- maintenance of adequate oxygenation and respiratory support
- monitoring and stabilization of blood pressure and hemodynamic status
- management of complications such as shock, pulmonary edema, or acute kidney injury
- treatment of secondary bacterial infections when indicated
Patients with severe hantavirus cardiopulmonary syndrome (HCPS) may require:
- admission to an intensive care unit (ICU)
- supplemental oxygen therapy
- Mechanical ventilation for respiratory failure
- vasopressor support for hypotension or shock
- extracorporeal membrane oxygenation (ECMO) in critically ill cases
Patients with severe hemorrhagic fever with renal syndrome (HFRS) may require:
- renal function monitoring
- fluid management
- dialysis or renal replacement therapy in cases of acute kidney injury or severe fluid overload
Intravenous ribavirin has shown benefit in some studies when administered early in certain cases of HFRS, but its effectiveness in HCPS remains unproven, and it is not universally recommended as standard treatment.
Prevention and Control of Hantavirus
Prevention of hantavirus infection focuses primarily on reducing exposure to infected rodents and their excreta.
Key preventive and control measures include:
- eliminating or minimizing contact with rodents in homes, workplaces, farms, storage areas, and campsites
- sealing holes, cracks, and gaps in buildings, homes, and garages to prevent rodent entry
- using rodent traps and implementing rodent control measures around living and working areas
- storing food, animal feed, and waste in rodent-proof containers
- maintaining good environmental sanitation by removing clutter, nesting materials, and potential rodent shelters
- avoiding direct contact with rodent urine, feces, saliva, or nesting materials
When cleaning areas contaminated by rodents:
- Avoid sweeping or vacuuming dry rodent droppings, as this may aerosolize infectious particles
- Wet contaminated areas with disinfectant before cleaning
- Use gloves and appropriate protective equipment when handling contaminated materials
- Wash your hands thoroughly after cleanup
For individuals in high-risk occupational or environmental settings:
- Use appropriate personal protective equipment (PPE)
- Follow infection control and rodent exposure prevention guidelines
At present, no widely available vaccine exists for routine prevention of hantavirus infection in most countries.
References
- CDC. “About Hantavirus.” Hantavirus, 9 May 2026, https://www.cdc.gov/hantavirus/about/index.html. Accessed 10 May 2026.
- “Hantavirus Cluster Linked to Cruise Ship Travel, Multi-Country.” Who.Int, 2024, https://www.who.int/emergencies/disease-outbreak-news/item/2026-DON599. Accessed 10 May 2026.
- CDC. “Hantavirus Outbreak Toolkit.” Hantavirus, 8 May 2026, https://www.cdc.gov/hantavirus/toolkit/index.html. Accessed 10 May 2026.
- CDC. “Clinical Overview of Hantavirus.” Hantavirus, 8 May 2026, https://www.cdc.gov/hantavirus/hcp/clinical-overview/index.html. Accessed 10 May 2026.
- CDC. “Hantavirus Prevention.” Hantavirus, 4 May 2026, https://www.cdc.gov/hantavirus/prevention/index.html. Accessed 10 May 2026.
- CDC. “Hantavirus: Current Situation.” Hantavirus, 9 May 2026, https://www.cdc.gov/hantavirus/situation-summary/index.html. Accessed 10 May 2026.
- https://www.medscape.com/viewarticle/708397_2
- https://diseasesdic.com/hantavirus-pulmonary-syndrome/
- https://www.ncbi.nlm.nih.gov/pubmed/11217408
- https://www.medicinenet.com/pdfguides/booklet/medicinenet-daily0912.pdf
- https://www.symptoma.com/en/info/hemorrhagic-fever-with-renal-syndrome
- https://www.researchgate.net/publication/12114769_Replication_of_Hantaviruses
- https://health.mo.gov/living/healthcondiseases/communicable/communicabledisease/cdmanual/pdf/Hantavirus.pdf
- https://en.wikipedia.org/wiki/Korean_hemorrhagic_fever
- https://elbiruniblogspotcom.blogspot.com/2011/09/cdc-hemorrhagic-fever-with-renal.html
- http://health.mo.gov/living/healthcondiseases/communicable/communicabledisease/cdmanual/pdf/Hantavirus.pdf
- https://www.researchgate.net/publication/259876829_First_Molecular_Evidence_for_Puumala_Hantavirus_in_Poland
- https://www.medscape.com/viewarticle/708397_4
- https://www.medscape.com/viewarticle/708397_3
- https://www.medscape.com/viewarticle/708397
- https://www.medicinenet.com/hantavirus_pulmonary_syndrome/article.htm
- https://www.cdc.gov/hantavirus/hps/prevention.html
- https://www.cdc.gov/hantavirus/hfrs/index.html
- https://viralzone.expasy.org/by_species/213
- https://viralzone.expasy.org/7099
- https://viralzone.expasy.org/4916?outline=all_by_species
- https://viralzone.expasy.org/213
- https://bestpractice.bmj.com/topics/en-us/928/diagnosis-approach
- http://ipm.ifas.ufl.edu/pdfs/Johnson_Viruses_and_Viroids_Important_Plant_Pathogens_pics96pdi.pptx

thank you for this! Such a great resource and primer for those of us who are public facing environmental public health.