Genetic engineering is also referred to as genetic modification or genetic manipulation. It involves the deliberate alteration or modification of an organism’s genetic material using modern technology and tools. The technologies involved are used to modify the cell’s genetic structure either by adding, removing, or altering specific genes.

Additionally, genetic engineering also includes the transfer of genetic material between or across species to generate organisms of improved characteristics. Traditional methods of genetic modification without the use of sophisticated tools involved the selective breeding of plants and animals, with no specification of the character to be altered, to achieve the targeted trait. However, modern genetic engineering enables precise, targeted modifications to the genetic code (DNA).
Source: https://www.pirbright.ac.uk/engage-with-us/science-case-studies/genetic-engineering
History and Development of Genetic Engineering
- The history of genetic engineering is rooted in the earlier practices of selective breeding or artificial selection of different plants and animals by humans.
- However, direct gene manipulation outside these traditional methods was not reported until the 1970s. Gene manipulation was not an independent principle, but it was based upon the principles of conceptual foundations in microbial genetics, heredity, and molecular biology.
- A major turning point was in 1967, with the isolation of DNA ligase, and in 1970 with endonucleases, laying the foundation for Paul Berg to create the first recombinant DNA molecule by combining DNA from a virus and a bacterium in 1972.
- Later, in 1973, Herbert Boyer and Stanley Cohen demonstrated that the recombinant hybrid DNA could be inserted into E. coli, which could be safely inherited by later generations. This marked the beginning of genetic engineering and gene cloning.
- In 1974, Rudolf Jaenisch introduced foreign DNA into a mouse and created the world’s first transgenic animal.
- This breakthrough was soon followed by the formation of Genentech in 1976, the first genetic engineering company, which soon produced human proteins, such as Somatostatin and Insulin, from bacteria.
- In the 1980s, PCR, DNA sequencing, and field trials of genetically engineered plants occurred rapidly.
- In 1992, China commercialized a virus-resistant transgenic tobacco plant, followed by the release of the first genetically modified food: The Flavr Savr tomato, insect-resistant BT cotton, and herbicide-tolerant soybeans.
- The Human Genome Project was also initiated in 1990 to sequence the entire human genetic structure, which has significantly transformed biological research.
- In the 2010s, genetic engineering tools such as TALENs and ZFNs were functionally applied to gene editing.
- In 2010, a fully synthetic genome JCVI-syn1.0 was created by the J. Craig Venter Institute, followed by the application of bacterial CRISPR-Cas9 as a gene-editing tool in 2012. These discoveries and their practical applications transformed the genetic engineering landscape.
- Current aspects of genetic engineering include base and prime editing, CRISPR-based therapies (Casgevy for sickle cell anemia), genetically engineered CAR-T cells for cancer, and many more.
Principles and Techniques Used in Genetic Engineering
The fundamental principle of genetic engineering is the manipulation of DNA, the carrier of genetic information. As DNA is universal and shared among bacteria, plants, animals, and humans, its information can be intentionally modified to achieve specific objectives. Through this intentional modification, genetic engineering influences various traits of an organism, for example, disease resistance, growth and metabolism, recombinant protein production, etc.
Techniques of genetic engineering depend upon the type of organism, the gene of interest, and the intended application. Generally, genetic engineering allows different types of modifications to be made at specific locations in the genome, such as knockouts, knockins, and the replacement of sequences.
Knockouts involve the deletion of DNA sequences for the purpose of stopping gene expression.
Knockins involve the insertion of new DNA sequences into the existing genome to modify the gene function.
Sequence replacement involves the substitution of an existing sequence with a new sequence to simultaneously disable a gene function and introduce a new one.
To achieve these modifications, advanced techniques such as gene editing by CRISPR-Cas9, gene silencing through RNA interference, genome sequencing, construction of synthetic DNA, and computational tools are involved. These techniques allow precise modification of target genes.
Hierarchy of Genetic Engineering
Generally, the concept of genetic engineering is limited to changes at the genome or gene-specific level. However, genetic engineering can be viewed more broadly at the cellular, organismal, and population levels.
In the gene-specific region, a target gene of interest could be inserted, deleted, or replaced. Changes to the cellular level can thus follow based on this concept. These cellular-level changes can be applied to diverse biological systems. For instance, gene editing tools, such as CRISPR and TALEN, enable genetic modification from single cells to complex multicellular organisms.
Similarly, population-level genetic engineering refers to the modification of an entire population rather than individual organisms. One of the most important tools for this is the Gene Drive. Gene drives alter the normal inheritance pattern of a gene, and, hence, allow a gene to spread rapidly through successive generations. For example, Gene drives are used to spread genes in the population of mosquitoes that make them resistant to transmitting malaria or reduce their population by introducing genes that harm their fertility.
Population-level engineering could lead to disruption of the ecosystem and biodiversity loss. Therefore, it requires strict regulation and ethical consideration before application.
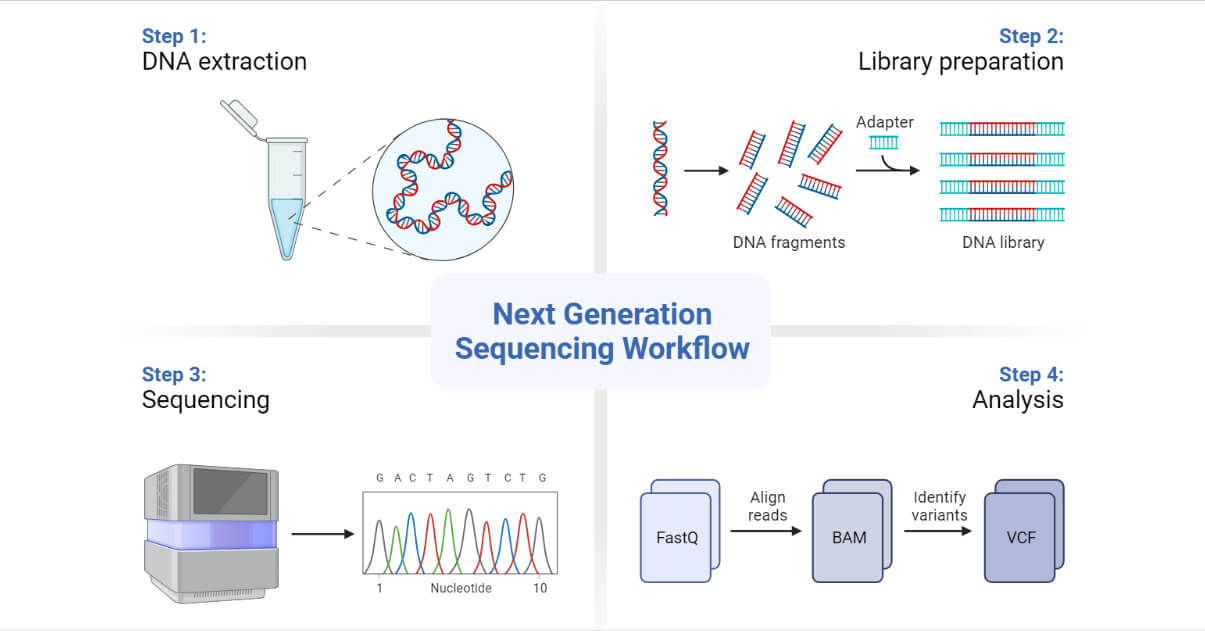
Tools and Techniques in Genetic Engineering
Genetic engineering involves a wide range of molecular tools, techniques, and platforms for gene alteration. These involve enzymes, vectors, gene editing tools, and analysis platforms. Some of the most relevant tools are noted:
Enzymes
Restriction endonuclease: to cleave nucleic acids (DNA) at specific sequences
DNA Ligase: to join cleaved DNA fragments together
DNA polymerase: to synthesize new DNA strands, essential for DNA replication, PCR, and gene cloning
Reverse transcriptase: to synthesize complementary DNA (cDNA) from mRNA
Alkaline Phosphatase: to insert or remove phosphate groups from the 5’ end of DNA to inhibit self-ligation
Vectors
Vectors are essential to carry foreign genetic elements into a host cell for multiplication, storage, or expression of genes. Plasmids, Bacteriophages, Cosmids, and BACs/YACs are some of the most commonly used vectors in genetic engineering.
Gene editing tools
CRISPR-Cas9 system: uses the concept of bacterial adaptive immunity in gene editing, special accessories include the nuclease Cas9 and its associated proteins and RNAs to create double-stranded breaks
Transcription Activator-Like Effector Nucleases (TALENs): Nucleases that are designed to bind to specific sequences and create breaks. They are highly precise and complex systems.
Zinc Finger Nucleases (ZFNs): These are hybrid proteins that combine the zinc finger DNA-binding domains with a nuclease to cause DNA cleavage.
Multiplex Automated Genome Engineering (MAGE): This system introduces simultaneous multiple genetic modifications to modify and engineer an organism, essential for large-scale edits.
Analysis Platforms
Various detection and selection platforms are used to find or detect changes in the genes, such as:
Polymerase Chain Reaction (PCR): a technique that produces multiple copies of a specific gene segment or a target DNA
Gel Electrophoresis: a technique that separates fragments of DNA based on their size
Gene probe: a ss-DNA or RNA tagged with fluorescent dyes, used to detect complementary nucleic acid sequences
DNA microarray: a technique used to quantify multiple gene expression, mutations, or the detection of genes based on complementary base-pairing
Sequencing: a tool to determine the exact nucleotide sequence of a gene, applicable in disease diagnosis, mutation analysis, and determining genetic variations.
Bioinformatics tools: Databases, web-based programs, and software comprise of bioinformatics. Some of them are: NCBI, Ensembl, UCSC for databases of genomic data; BLAST for sequence homology, ClustalW and MUSCLE for sequence alignment; Galaxy for complex bioinformatics workflows, such as sequencing and microarray data analysis; PDB for protein structure; DESeq for gene expression, etc.
Source: https://blast.ncbi.nlm.nih.gov/Blast.cgi
Applications of Genetic Engineering in Medicine, Agriculture, and Industry
Medicine and Healthcare
- Biopharmaceuticals: By using relevant human genes in microbial or mammalian hosts, genetic engineering allows the large-scale manufacture of medically important components, such as hormones and enzymes.
- Diagnostics: Advanced techniques of genetic engineering allow early detection of diseases by the identification of genetic markers of diseases or mutations, or the detection of a pathogen genome.
- Gene-therapies: Genetic engineering techniques are applied to introduce, replace, or regulate gene expression in diseased humans to treat hereditary disorders or acquired diseases.
- Vaccine development: Genetic engineering, in recent times, utilizes sophisticated techniques to produce more effective and safer vaccines.
Agriculture
- Transgenic crops: Genetically modified crops are incorporated with several beneficial traits, such as improved adaptability to climate conditions, resistance to pests, tolerance against herbicides, etc.
- Nutritional quality: Genetically engineered crops are also applied to improve their nutritional composition, being richer in essential macronutrients such as vitamins, minerals, amino acids, etc.
- Yield enhancement: Genetic engineering techniques are used to enhance crop productivity, for example, the use of CRISPR-Cas9 in rice and wheat to increase their grain size. Similarly, transgenic animals in farms are introduced with genes for enhanced production of milk, meat, wool, etc.
- Shelf life: Genetic engineering allows modifications in genes and proteins that are involved in the ripening of fruits and vegetables, which can help them stay fresh longer.
Industry
- Enzymes: Genetically engineered microbes are used to produce enzymes applicable in food, textile, detergents, and pharmaceutical industries, effective in increasing industrial efficiency and cost management.
- Biofuels: Genetic engineering and its tools have induced improvements in traits of microorganisms regarding the conversion of organic and inorganic waste into the production of biofuels, such as ethanol and biodiesel.
- Bioremediation: Engineered microorganisms and plants can be applied to degrade chemically complex and potentially non-biodegradable compounds, and hence contribute to environmental cleanup.
Advantages of Genetic Engineering
- Precision: Genetic engineering allows the direct change of specific genes responsible for a specific trait, making the overall process more targeted, which improves accuracy and reduces time as compared to conventional methods.
- Improved therapeutics: Genetic engineering has significantly enhanced diagnostics, prevention, and treatment of diseases through the production of recombinant vaccines, therapeutic proteins, and gene therapies.
- Productivity in agriculture: Genetically engineered crops can be designed for better yield, better nutritional quality, resistance against pesticides, herbicides, and tolerance against abiotic stress. All these improvements contribute directly to food security and sustainable agriculture.
- Industrial efficiency: Genetically engineered microorganisms are used for the large-scale synthesis of enzymes, biofuels, and molecules of pharmaceutical and industrial relevance in a more economical, efficient, and controlled manner.
Limitations of Genetic Engineering
- Unintended effects: Off-target changes or undesired consequences due to genetic modification can affect the safety and stability of the target organism.
- Environmental issues: Release of GMOs into the environment may lead to ecological disruption due to undesired gene flow.
- Costly and complex: Techniques of genetic engineering require sophisticated equipment, skilled personnel, and advanced laboratories. This could limit accessibility and functionality in resource-limited settings.
- Regulation: Due to ethical concerns, safety debates, and regulatory policies across the world, genetic engineering faces resistance, leading to a slowing of progress in research and its application.
Ethical, Legal, and Social Issues in Genetic Engineering
- Welfare of engineered organisms: In animals, gene modifications raise questions about welfare, suffering, and the acceptability of altering the natural biological processes for human benefit.
- Human genetic modification: Germline editing in humans raises serious ethical questions about consent, safety, and the long-term effects of genetic engineering on future generations. Similarly, the possibility of modifying genes for enhanced abilities creates concerns about the commercialization of human traits, leading to potential misuse and inequality.
- Biosafety and policy: Governments and regulatory agencies require strict safety testing before the release or use of genetically engineered products to prevent harm to the environment. Additionally, international differences in policies regarding GMOs and their biosafety practices vary, creating more challenges.
- Cultural views and acceptance: Based upon cultural beliefs and religious values of different societies, genetic modifications leading to genetically engineered products could be hard to accept.
Future Trends and Advances in Genetic Engineering
- Updated gene editing platforms: CRISPR-Cas-based newer technologies, such as base and prime editing, epigenome engineering, and retron-based gene editing, are constantly improving precision in gene modifications, with higher efficiency and reduced off-target effects.
- Personalized medicines: Personalized medicines, using a patient-specific genetic makeup, are used to create targeted therapies. For example, personalized medicine in cancer treatment or severe genetic diseases can reduce side effects and improve treatment success rates.
- Synthetic biology: Synthetic biology uses bioinformatics tools and computerized modelling to generate new, novel DNA sequences, applicable for newer functions in medicine, healthcare, agriculture, etc.
- Integration with AI: Traditional genetic engineering is increasingly being conjugated with AI and bioinformatics to analyze genes, predict gene functions, discover newer proteins and drugs, and many more, for effective interventions in microorganisms, plants, and human health.
Conclusion
In this modern era, genetic engineering has developed as a highly powerful tool in biotechnology and modern science, enabling precise and efficient genetic manipulation. This has been applied diversely in the fields of medicine, agriculture, and industry. However, its ethical, social, and environmental challenges and limitations must be carefully addressed.
Genetic engineering and its tools, in summary, hold great potential to address complex biological challenges provided that they are applied while holding scientific integrity, ethical responsibility, and social constraints.
References
- J.S. Robert, F. Baylis, Genetic Engineering, Editor(s): Harald Kristian (Kris) Heggenhougen, International Encyclopedia of Public Health, Academic Press, 2008, Pages 35-39, ISBN 9780123739605, https://doi.org/10.1016/B978-012373960-5.00133-7
- Nair, A. J. (2008). Introduction to biotechnology and genetic engineering. Infinity Science Press.
- Nicholl, D. S. T. (2008). An introduction to genetic engineering (3rd ed.). Cambridge University Press.
- https://www.ebsco.com/research-starters/health-and-medicine/historical-development-genetic-engineering#historical-development-of-genetic-engineering
- Sundar Durai, Mala Mani, Karthikeyan Kandavelou, Joy Wu, Matthew H. Porteus, Srinivasan Chandrasegaran, Zinc finger nucleases: custom-designed molecular scissors for genome engineering of plant and mammalian cells, Nucleic Acids Research, Volume 33, Issue 18, 1 October 2005, Pages 5978–5990, https://doi.org/10.1093/nar/gki912
- Sun, N., & Zhao, H. (2013). Transcription activator-like effector nucleases (TALENs): a highly efficient and versatile tool for genome editing. Biotechnology and bioengineering, 110(7), 1811–1821. https://doi.org/10.1002/bit.24890
- Committee on Gene Drive Research in Non-Human Organisms: Recommendations for Responsible Conduct; Board on Life Sciences; Division on Earth and Life Studies; National Academies of Sciences, Engineering, and Medicine. Gene Drives on the Horizon: Advancing Science, Navigating Uncertainty, and Aligning Research with Public Values. Washington (DC): National Academies Press (US); 2016 Jul 28. Available from: https://www.ncbi.nlm.nih.gov/books/NBK379282
- V. Edwin Hillary, S. Antony Ceasar, Chapter Seven – Genome engineering in insects for the control of vector borne diseases, Editor(s): Vijai Singh, Progress in Molecular Biology and Translational Science, Academic Press, Volume 179, 2021, Pages 197-223, ISSN 1877-1173, ISBN 9780323853217, https://doi.org/10.1016/bs.pmbts.2020.12.017.
- https://en.wikipedia.org/wiki/Genetic_engineering
- Shakweer, W. M. E., Krivoruchko, A. Y., Dessouki, S. M., & Khattab, A. A. (2023). A review of transgenic animal techniques and their applications. Journal, genetic engineering & biotechnology, 21(1), 55. https://doi.org/10.1186/s43141-023-00502-z
- Wiley, L., Cheek, M., LaFar, E., Ma, X., Sekowski, J., Tanguturi, N., & Iltis, A. (2025). The Ethics of Human Embryo Editing via CRISPR-Cas9 Technology: A Systematic Review of Ethical Arguments, Reasons, and Concerns. HEC forum : an interdisciplinary journal on hospitals’ ethical and legal issues, 37(2), 267–303. https://doi.org/10.1007/s10730-024-09538-1
- Zebanaz, A., Kareem, A. M., Aher, A. A., & Thitame, S. N. (2025). Personalized Medicine in Treating Rare Genetic Disorders: A Review. Journal of pharmacy & bioallied sciences, 17(Suppl 1), S59–S62. https://doi.org/10.4103/jpbs.jpbs_583_25
- Bradbury, A., Clapp, O., Biacsi, A. S., Kuo, P., Gaju, O., Hayta, S., Zhu, J. K., & Lambing, C. (2025). Integrating genome editing with omics, artificial intelligence, and advanced farming technologies to increase crop productivity. Plant communications, 6(7), 101386. https://doi.org/10.1016/j.xplc.2025.101386
- Buffington, J.D., Kuo, HC., Hu, K. et al. Discovery and engineering of retrons for precise genome editing. Nat Biotechnol (2025). https://doi.org/10.1038/s41587-025-02879-3
