The enterovirus D68 (EV D68) is not related to polio. It is no longer a rare respiratory virus and has become a significant cause of acute flaccid myelitis (AFM) and severe respiratory illness in children since 2014.

It is currently under study regarding its molecular evolution, tissue tropism, immunological evasion, and potential vaccine and antiviral targets.
Taxonomy and Classification of Enterovirus D68
Family / Genus / Species
- Family: Picornaviridae; genus: Enterovirus; species: Enterovirus D
- One of five known Enterovirus D members (EV-D68, -D70, -D94, -D111, -D120).
Serotype and clades
- Single serotype, but multiple genetic clades (A–D and subclades, e.g., B3, A2/D1) with distinct epidemiologic behavior and virulence signatures.
Structure and Morphology of Enterovirus D68
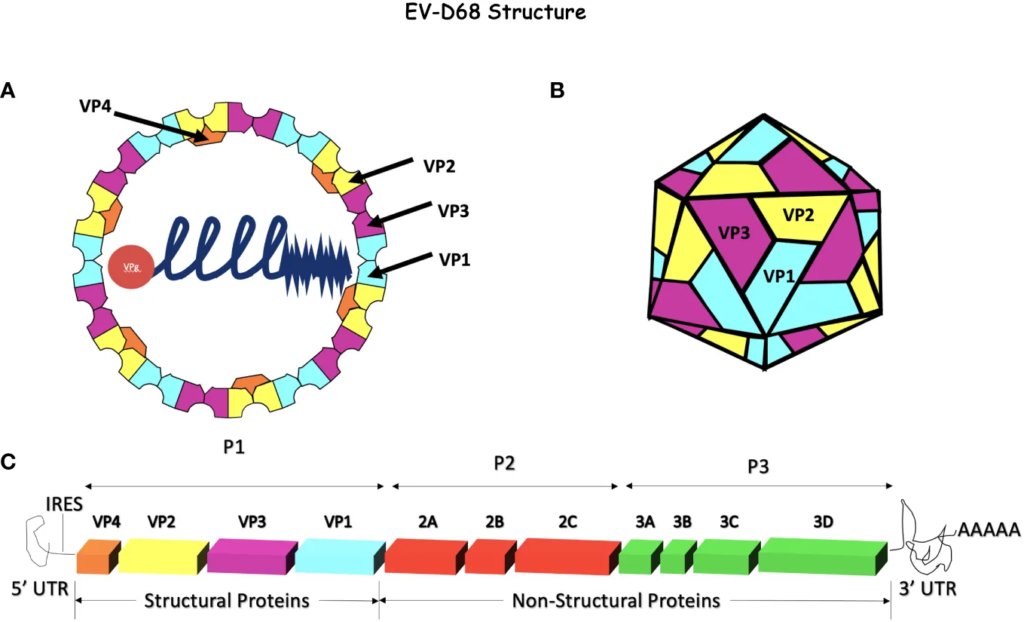
Virion
- Non-enveloped, ~30 nm, icosahedral capsid.
- Capsid proteins: 60 copies each of VP1–VP4; VP1–VP3 exposed, VP4 internal.
Receptor usage & uncoating
- Binds sialylated glycans and neuron-specific receptor ICAM-5; newer data implicate additional receptors (e.g., heparan sulfate, MFSD6).
- Contemporary strains show a broader temperature range, more efficient uncoating, and altered receptor specificity vs. the historic Fermon strain.
Genome Organization and Proteins of Enterovirus D68
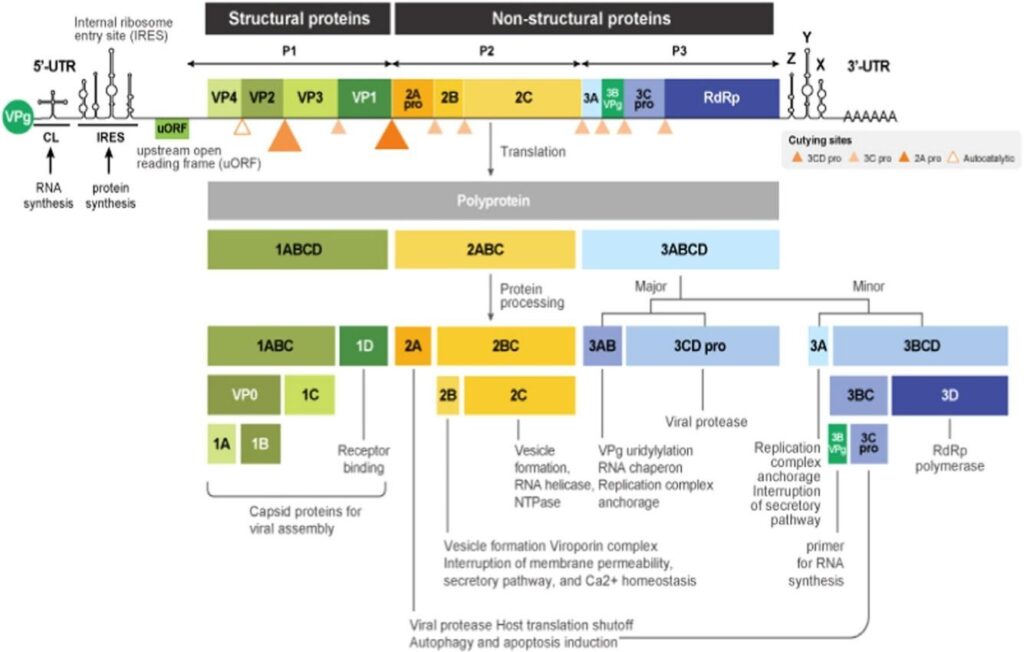
Genome
- ~7.4–7.6 kb, single positive-sense RNA with 5′-UTR (IRES), single ORF, 3′-UTR, poly(A) tail.
- Polyprotein processing
-ORF encodes polyprotein → cleaved by viral proteases 2Apro and 3Cpro into:
- Structural: VP1–VP4 (capsid).
- Non-structural: 2A, 2B, 2C, 3A, 3B (VPg), 3C, 3D (RNA-dependent RNA polymerase).
- Key functional determinants
VP1–VP3: receptor binding, antigenicity; VP1 BC loop critical for serotype-specific neutralization and virulence evolution.
2Apro / 3Cpro: host shutoff, immune antagonism; virulence-modifying sites include 2Apro–TRAF3 cleavage sequence and 3Cpro immunoregulatory motifs.
2C, 3A, 3Dpol: replication organelle formation and RNA synthesis; 2C and 3A interact with host ACBD3, PI4KB, OSBP.
Selected Virulence-Linked Regions in Enterovirus D68
| Viral region | Functional role | Virulence-relevant changes |
| VP1 RBS | Receptor binding, antigenicity | Altered affinity for sialic acid, heparan sulfate, MFSD6; capsid antigenic drift |
| 2Apro site | Cleaves host TRAF3 | Variants may modulate immune evasion efficiency |
| 3Cpro | Polyprotein, host protein cleavage | Changes in immunoregulatory interfaces and innate immune antagonism |
Replication Cycle and Molecular Features of Enterovirus D68
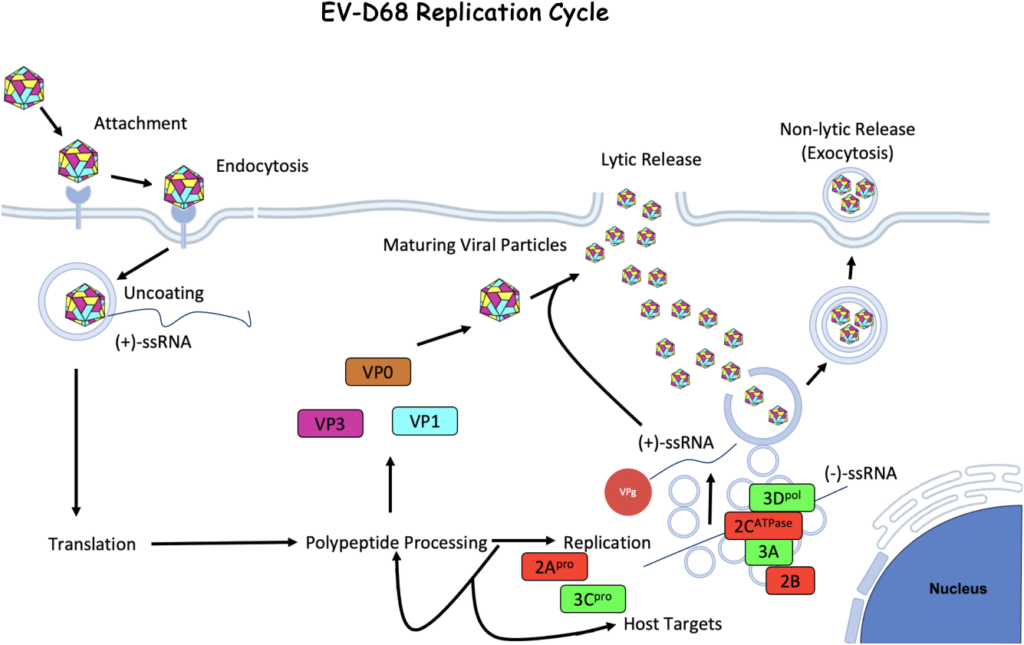
Entry and Uncoating
Receptors and attachment
- EV-D68 uses multiple receptors: MFSD6, a multipass membrane transporter now identified as a bona fide entry receptor, is essential for infection of respiratory and neural cells and directly binds the viral capsid via its extracellular loop L3.
- EV-D68 also recognizes sialic acid–containing glycoconjugates and heparan sulfate (HS); different strains preferentially bind α2,6-linked sialic acids on N-glycans, α2,8-linked sialic acids on gangliosides, or both.
- Ganglioside-type disialoglycolipids function as entry receptors for certain strains; VP1 residue 271 adaptations are linked to HS binding.
Attachment → endocytosis → uncoating
- After receptor engagement, EV-D68 is internalized by endocytosis, followed by transfer of the RNA genome from endosomes into the cytoplasm.
- Contemporary strains show more efficient uncoating and reduced receptor-binding stringency, which together may increase infectivity and broaden cell range.
Translation and Polyprotein Processing
- The positive-sense RNA genome encodes a single polyprotein, cleaved into structural (VP1–VP4) and nonstructural (2A–2C, 3A–3D) proteins.
- Translation is directed by an internal ribosome entry site (IRES) in the 5′ UTR; the compound licochalcone A selectively inhibits EV-D68 by blocking IRES-dependent translation without affecting attachment/entry.
- Proteases 2Apro and 3Cpro process the polyprotein and also cleave host factors; sequence variation at the 2Apro/TRAF3 site and within 3Cpro modulates immune evasion and replication efficiency.
RNA Replication and Replication Organelles
- Replication occurs on virus-induced replication organelles (ROs) derived from ER/Golgi membranes, driven by viral proteins 2B, 2C, 3A, and host factors such as ACBD3, PI4KIIIβ, OSBP, and ORP4.
- RNA-dependent RNA polymerase 3Dpol synthesizes negative-strand RNA, which serves as a template for abundant positive-strand genomes.
- Contemporary EV-D68 strains show more efficient RNA transcription and putative changes in replication dynamics, contributing to increased virulence.
Assembly and Release
- Newly synthesized RNA is packaged into capsids formed by VP0 (later cleaved to VP2+VP4), VP1, and VP3.
- Progeny virions are released by cell lysis or via extracellular vesicles, both described for EV-D68 and other enteroviruses.
- Use of vesicle-mediated release may aid immune evasion and dissemination.
Key Molecular Virulence Determinants
| Viral region/function | Effect on biology/virulence |
| VP1 receptor-binding sites | Alter affinity for sialic acid, HS, MFSD6; change tissue tropism and entry efficiency. |
| 2Apro (TRAF3 site) | It modulates the cleavage of TRAF3, affecting innate immune signaling |
| 3Cpro immunoregulatory region | Alters cleavage of host translation/autophagy proteins → replication & immune control |
| 5′ / 3′ UTR elements, IRES | Regulate translation and replication; target of licochalcone A |
Pathogenesis and Host Immune Response to Enterovirus D68
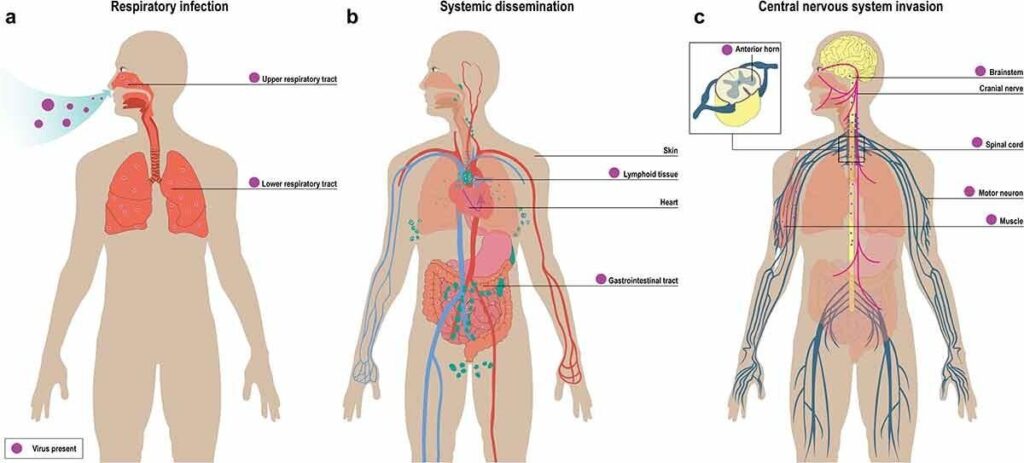
From Respiratory Infection to Systemic Spread and CNS Involvement
- Primary infection site: EV-D68 is primarily a respiratory virus, entering via the upper airway; most infections are mild but can progress to severe lower respiratory disease.
Temperature and acid sensitivity
- Earlier isolates were more acid-sensitive and favored lower temperatures, fitting upper airway replication.
- Some recent strains show reduced sensitivity to acid and higher temperature, enabling replication in a broader range of tissues, including potentially the gut.
Systemic dissemination and neuroinvasion
- Increased virulence and replication efficiency are proposed to raise the likelihood of viremia and CNS invasion rather than a simple gain of neurotropism.
- EV-D68 has been detected in anterior horn cells in a child with AFM, directly linking the virus to spinal cord motor neuron injury.
- Contemporary strains, but not historic ones, infect human spinal cord organoids and maintain replication with minimal direct cytopathic effect, implying that immune-mediated secondary injury likely contributes to neuronal loss in AFM.
- Animal and in vitro models show strain-specific differences in CNS invasion and replication, not restricted to a single clade.
Innate Immune Responses
Recognition and interferons
- Pattern-recognition receptors detect EV-D68 and trigger type I and III interferon responses, activating NK cells and antiviral genes.
- In primary human airway cells, EV-D68 infection elicits a robust type III interferon response that restricts replication; intestinal enteroids mount a weaker response, paralleling tissue-specific susceptibility.
Viral immune evasion
- EV-D68 uses antigenic variation in capsid proteins to escape neutralizing antibodies and alter receptor usage.
- Nonstructural proteins 2Apro and 3Cpro cleave host immune regulators (e.g., TRAF3, translation/autophagy components), dampening antiviral signaling and reshaping cellular homeostasis.
- Shifts in capsid antigenicity and RNA metabolism collectively contribute to increased virulence and immunogenicity changes, facilitating broader spread and more severe disease.
Adaptive Immunity and Immunopathology
Humoral and cellular responses
- B cells generate neutralizing antibodies, and T cells coordinate targeted antiviral responses; however, antigenic drift can reduce pre-existing immunity.
- Immunocompromised hosts appear at higher risk for severe neurologic disease, as illustrated by an adult AFM case with EV-D68 B3 infection.
Role of immune-mediated damage in AFM
- In spinal cord organoids, productive EV-D68 infection alone causes limited cell death. In contrast, another enterovirus causes marked apoptosis, suggesting that host inflammatory responses in vivo may drive much of the neuronal damage seen in AFM.
- Reviews emphasize major gaps in understanding how systemic dissemination, CNS entry routes, and immune responses combine to produce AFM.
Epidemiology and Transmission of Enterovirus D68
Transmission Routes and Tissue Tropism
- EV-D68 is mainly a respiratory pathogen, transmitted via respiratory secretions and droplets; most detections are from respiratory samples.
- Detection in stool and wastewater suggests potential enteric involvement and possibly fecal-oral transmission for some strains, especially acid-tolerant contemporary variants.
- Receptor promiscuity (MFSD6, sialylated glycoproteins, gangliosides, HS) likely underlies its ability to infect respiratory, intestinal, and neural tissues.
Seasonality and Cyclic Outbreak Patterns
- Since 2012, EV-D68 has shown biennial outbreak patterns, with peaks in late summer–fall, especially in even-numbered years (2014, 2016, 2018).
- COVID-19 mitigation measures in 2020 disrupted this pattern, with far fewer detections than expected.
Post-pandemic, sustained and sometimes annual upsurges have been observed:
- Large European upsurge in 2021–2022, with 1004 EV-D68 infections (≈79% in children 0–5 years); 37.9% hospitalized; neurological problems in 6.4% (including 6 AFM).
- Continued B3-lineage evolution and sustained circulation in 2023 Europe, with most detections in respiratory samples and emerging amino acid changes (e.g., VP1 D554E).
- In Maryland, enhanced surveillance documented increased EV-D68 circulation in 2024, restoring the biennial pattern; EV-D68 formed 72.6% of EV-positive samples, peaking in October.
Clades, Lineages, and Virulence Associations
- EV-D68 has evolved into clades A–D (with subclades A1/A2/D1/D2; B1–B3). Post-2014, B3 has predominated, with some D1 circulation.
Genomic and clinical comparisons show:
- 2018 vs 2022 (Maryland): 2018 infections had higher odds of hospitalization and ICU care; a specific amino acid cluster (e.g., at positions 554, 650, 918, 945, 1445, 1943) associated with severe outcomes.
- 2024 B3 genomes acquired additional substitutions in 3C, 3D, and VP2 (e.g., VP2 T145S), underscoring ongoing adaptation.
- A 2026 systematic review identifies VP1 receptor-binding residues, 2Apro/TRAF3 site, and 3Cpro immunoregulatory motifs as recurring virulence hot spots.
Burden, Clinical Spectrum, and AFM Link
- Clinical spectrum ranges from mild upper respiratory illness to severe lower respiratory disease and AFM.
Large European cohort (2021–2022):
- 78.9% of cases in 0–5-year-olds; ~38% hospitalized; frequent acute respiratory distress; AFM rare but present.
U.S. NVSN data (2018–2020):
- 382 pediatric EV-D68 ARI cases in 2018 vs 6 in 2019 and 30 in 2020, with most 2018 patients <5 years old.
- Multimodal surveillance in Colorado (2022), combining clinical, syndromic, and wastewater data, enabled early detection of an EV-D68 wave and close monitoring of associated AFM risk.
- Despite large EV-D68 waves in 2022 and 2024, AFM surges were not always proportional, highlighting that viral genetics, host immunity, and exposure patterns jointly determine AFM incidence.
Therapeutic and Preventive Implications
No licensed vaccines or specific antivirals exist, but several strategies target:
- Capsid–receptor interactions (e.g., MFSD6-based decoy receptor protected mice from lethal EV-D68 challenge).
- Replication machinery and host factors essential for RO formation and RNA synthesis.
- Reviews emphasize the need for ongoing genomic surveillance, immunologic studies, and model systems (organoids, animal models) to guide vaccine and antiviral design.
Clinical Manifestations of EV-D68 Infection
Respiratory disease
Typical presentation
- EV-D68 primarily causes acute respiratory illness, especially in children. Symptoms range from mild upper respiratory (rhinorrhea, sore throat, cough, wheeze) to severe lower respiratory tract infection (bronchiolitis, pneumonia, acute hypoxic respiratory failure).
- In a Maryland 2018–2022 series (n=339 with clinical data), 46.9% required supplemental oxygen and 22.4% required ICU care; 55.5% were hospitalized, half with EV-related indications.
Risk groups and severity modifiers
- Children <5 years predominate (median age ~5 years).
- Asthma was the most frequent comorbidity (≈46%); cancer and immunosuppression were also common.
- 2018 infections had higher odds of hospitalization and ICU admission than 2022, despite similar clade distribution, implicating viral genomic differences in severity.
Systemic and extrapulmonary features
- Fever, headache, malaise, and myalgias are variably present.
- Animal models (cotton rats) show systemic inflammation, viremia, and infection of extra-respiratory organs, paralleling occasional systemic signs in humans.
Neurological disease (Acute Flaccid Myelitis and others)
- AFM phenotype
- Poliomyelitis-like AFM generally occurs days to weeks after a febrile respiratory prodrome.
Hallmarks:
- Acute onset of asymmetric flaccid limb weakness, often proximal and with upper-limb predominance.
- Cranial nerve involvement (facial weakness, bulbar symptoms) and in ~⅓ of reported cases, respiratory muscle compromise requiring ventilatory support.
- Swiss case descriptions highlight rapid progression from mild respiratory symptoms to focal limb weakness, with MRI spinal gray-matter lesions; recovery is often incomplete, leaving persistent paresis, though some children fully recover.
Other neurologic presentations
- Less frequent: encephalopathy, meningitis, radiculitis; rarely fatal paralysis.
- Cotton rat intraperitoneal infection models show virus dissemination to the spinal cord and brain, with clinical neurologic signs, strengthening the biological plausibility of EV-D68 neurotropism.
Outcome
- No specific therapy; many AFM patients retain long-term motor deficits despite early immunomodulation and intensive rehabilitation.
Laboratory Diagnosis of Enterovirus D68
Specimen selection and timing
Preferred specimens
- Nasopharyngeal/oropharyngeal swabs or lower respiratory specimens during the acute respiratory phase provide the highest yield.
- Stool and CSF are often collected during AFM assessment, but EV-D68 RNA is infrequently detected in CSF, likely due to timing and low CNS viral loads.
Diagnostic algorithm
Screening for rhinovirus/enterovirus
- Many clinical labs use multiplex respiratory PCR panels detecting a combined “rhinovirus/enterovirus” signal. Positive specimens during the EV-D68 season warrant reflex testing for EV-D68.
Type-specific molecular assays
CDC2022 rRT-PCR assay:
- Targets conserved amino-acid motifs across >60 years of EV-D68 evolution, designed based on phylo-primer-mismatch analysis.
- In 625 respiratory specimens, sensitivity 100% (281/281) and specificity 100% (344/344) vs. VP1 semi-nested PCR/sequencing.
- No cross-reactivity with 104 EV A–D non-D68 isolates, 165 rhinoviruses, or 14 other respiratory viruses.
- LOD: ~361 RNA copies/reaction; detects as little as 0.28 CCID50 per reaction.
- Detects emergent 2024 strains, indicating robustness for evolving clades.
Russian RT-PCR kit with EV-D68 differentiation:
- Simultaneous detection of EV-A/B/C/D and differentiation of EV-D68.
- Analytical sensitivity: 10³ GE/mL for respiratory samples and CSF, 5×10³ GE/mL for stool.
- No cross-reaction with rhinoviruses or other viral/bacterial pathogens.
- In 310 clinical samples, diagnostic sensitivity and specificity were each 100% vs. a reference kit and VP1 sequencing; now approved for in vitro diagnostic use.
VP1 sequencing and genotyping
- VP1 sequencing remains the gold standard for typing and clade/subclade assignment and is essential for outbreak investigations and linking clinical severity to viral genetics.
- Whole-genome sequencing enables identification of non-structural amino acid clusters (e.g., residues 554, 650, 918, 945, 1445, 1943) associated with more severe clinical outcomes (higher odds of hospitalization/ICU).
Other laboratory methods
Virus isolation
- EV-D68 can be cultured in cell lines (e.g., RD, HeLa, respiratory lines), but is not routinely used for clinical diagnosis due to lower sensitivity and slower turnaround; primarily used for research and phenotyping of antiviral susceptibility.
Serology
- Neutralizing antibodies to EV-D68 are detectable in AFM patients at higher titers than in controls, supporting causality, but no standardized serologic assay is in routine diagnostic use.
Surveillance applications
- Type-specific rRT-PCR assays and sequencing are central to national EV-D68 surveillance, allowing real-time correlation of molecular clades with clinical manifestations and AFM incidence.
Treatment and Antiviral Therapy for Enterovirus D68
There is no approved EV-D68-specific antiviral or vaccine. Management is predominantly supportive, but a rapidly expanding pre-clinical pipeline targets multiple viral and host factors.
Current clinical management
Supportive care
- Oxygen, bronchodilators, and ventilation as needed for respiratory disease; ICU management for respiratory failure or shock.
- AFM care centers on early neurological evaluation, MRI, CSF studies, and supportive management, including respiratory support, physical and occupational therapy.
Immunomodulatory therapy in AFM
- High-dose IV methylprednisolone, intravenous immunoglobulin (IVIG), and sometimes plasma exchange are frequently used empirically; Swiss AFM cases received high-dose steroids followed by IVIG with variable recovery.
- No controlled trials demonstrate a clear benefit; use remains case-by-case, based on extrapolation from other neuro-inflammatory syndromes.
Direct-Acting Antiviral Candidates
Capsid-targeting agents (entry/uncoating inhibitors)
Pleconaril analog 11526092 (isoxazole-3-carboxamide)
- Potent in vitro inhibition of EV-D68 (IC50 ~58 nM) and other enteroviruses, including pleconaril-resistant CVB3.
- Cryo-EM of EV-D68–11526092 and –pleconaril complexes show binding within the VP1 hydrophobic pocket and destabilization of the VP1 loop; strain-dependent effects influence resistance profiles.
In a mouse respiratory EV-D68 model, treatment led to:
- ~3-log reduction in viremia and ~1-log reduction in lung titers by day 5.
- More favorable cytokine profile vs. controls.
However, no efficacy in a mouse AFM model (neurological disease endpoint), suggesting limited CNS penetration or inadequate timing for neuroprotection.
Novel VP1-binding quinoline analogues
- Structure-guided screening identified quinoline derivatives with EC50 0.05–0.10 μM across diverse EV-D68 strains.
- Confirmed reduction in viral proteins and infectious titers by Western blot, IF, and plaque assays; mechanism: interaction with VP1 and inhibition of replication.
- Lead compound showed 23.9% oral bioavailability and good metabolic stability in human liver microsomes, strengthening its candidacy for further development.
Geranyl-p-trans-coumaric acid (GCA)
- Natural product identified through high-throughput screening; EC50 20–40 µM against multiple clinical EV-D68 strains.
- Time-of-addition experiments: maximal effect during adsorption/early entry, with direct binding to the virion; prevents uncoating without interfering with initial attachment.
- Capsid stabilization demonstrated by thermal shift (RNA release temperature rise from 51°C to 53°C); selectively inhibits heparan-sulfate engagement but not ICAM-5 interaction, targeting secondary receptor engagement.
- Resistance requires dual mutations in VP1 (T92N) and 2C (K6R), implying dual targeting of capsid and a 2C-linked function; single mutants retain susceptibility.
- In vivo, GCA improved survival in infected mice from 18% to 71% at 12 mg/kg/day and significantly decreased viral burdens in the brain and muscle.
- Synergy with pleconaril (combination index 0.83) suggests potential for combination regimens to limit resistance.
Decoy receptor microbodies (MFSD6-Fc)
- EV-D68 has recently been shown to use MFSD6 as a receptor. A secreted MFSD6-Fc “microbody” acts as a decoy receptor.
- In vitro, secMFSD6-Mb reduced viral attachment >90% in multiple respiratory cell lines and primary HBECs.
- EM and gradient assays reveal conversion to empty capsids and increased free RNA, indicating that the decoy not only blocks attachment but also triggers premature uncoating/genome release from the cell.
- In a neonatal mouse lethal challenge model, survival improved from 11% to 89%, demonstrating strong in vivo efficacy.
2C-targeting inhibitors (replication organelle and RNA synthesis)
A-967079 (2C inhibitor)
- Identified by phenotypic screening, with potent, broad-spectrum activity and a high selectivity index against multiple EV-D68 strains.
- Mechanistic assays (time-of-addition, resistance selection, differential scanning fluorimetry) implicate viral 2C as the direct target.
- Represents a promising lead for EV-D68-specific therapy; however, clinical pharmacology and in vivo efficacy data are still early.
Pyrazolopyridine 2C inhibitors
- A class of small molecules active against EV-D68, EV-A71, and CVB3; resistance mapping and thermal shift assays confirm 2C as the principal target.
- Provide broad-spectrum non-polio enterovirus coverage, valuable for empiric therapy during outbreaks when typing is not yet available.
Rationally designed 2C inhibitor Jun6504
- Designed via structure-based methods; shows potent, broad-spectrum activity across multiple EV-D68 strains and other enteroviruses, with favorable PK.
In a neonatal mouse EV-D68 model:
- Improved paralysis scores and weight gain when given immediately or 24 h post-infection.
- Reduced viral titers in the spinal cord and quadriceps muscle.
- Demonstrates that 2C inhibition can prevent or ameliorate paralysis, directly relevant to AFM-like disease.
General 2C inhibitor platform
- Broader enterovirus work reveals that 2C inhibitors can yield capsid mutations as resistance, implying 2C–capsid interface interruption and relevance for late stages such as assembly/encapsidation.
- These compounds are valuable tools to dissect EV-D68 assembly and may inform combination therapies (e.g., 2C + VP1 inhibitors).
3C protease inhibitors
Natural-product 3C hits (in silico)
- Docking-based screening highlighted Withaferin A and baicalin as EV-D68 3C protease binders with high predicted affinity (–10.7 and –9.5 kcal/mol) and favorable PK, with stable interactions in 100-ns MD simulations.
- These are computational candidates; wet-lab validation is needed before clinical translation.
- Structural analysis of 3C-inhibitor complexes
High-resolution EV-D68 3C crystal structures with several inhibitors reveal:
- Conserved catalytic site architecture.
- A key structural water molecule is consistently maintained during ligand binding.
- Flexibility of the catalytic His40 side chain, which may influence inhibitor design and resistance.
- Structural data guide resistance-robust 3C inhibitor design, analogous to strategies in HIV protease therapy.
Host-Targeted and Broad Enterovirus Strategies
Broad reviews of enterovirus and EV-D68 antivirals emphasize:
- Capsid binders and monoclonal antibodies (entry/attachment blockade).
- Protease (2A/3C) and polymerase inhibitors (polyprotein processing, RNA replication).
- Host-factor inhibitors (e.g., PI4KB, OSBP, ACBD3, autophagy, vesicular trafficking pathways) disrupt replication organelles.
- Emerging immunotherapeutic and gene-editing approaches (neutralizing antibodies, RNAi, CRISPR-based tools, antiviral peptides).
- Combination therapy is a key principle to enhance efficacy and prevent resistance.
Key Issues for Clinical Translation of Enterovirus D68
Bridging animal models and human disease
- Multiple animal models (neonatal mice, cotton rats, others) now replicate EV-D68 respiratory disease, systemic dissemination, and AFM-like paralysis.
- These models have been instrumental for evaluating 2C inhibitors (Jun6504), capsid-binders (11526092, GCA), and decoy receptors (MFSD6-Fc).
- Nonetheless, heterogeneity in disease phenotypes and differences from human AFM remain major challenges for predicting clinical efficacy.
Gaps and future directions
- No human trial has yet shown a clear benefit of any EV-D68-directed antiviral.
Critical needs include:
- Standardized severity endpoints for respiratory and neurologic disease.
- Optimization of dosing and time-to-treatment windows, especially for AFM (where neurological injury may be established before presentation).
- Combination regimens (e.g., VP1 + 2C ± 3C inhibitors; antivirals plus immunomodulation) to improve outcomes and limit resistance.
Prevention and Control of Enterovirus D68
Public health surveillance
- Biennial peaks and linkage to AFM underscore the need for integrated EV-D68 and AFM surveillance with genomic sequencing to monitor virulence determinants (VP1, 2Apro, 3Cpro, non-structural mutation clusters).
Infection control
- Respiratory hygiene, isolation precautions similar to other respiratory viruses (droplet/contact) in healthcare settings; attention during known peak seasons.
Vaccine development
- No licensed vaccines; candidate approaches include inactivated and subunit vaccines targeting VP1 or cross-reactive epitopes, but work remains preclinical.
- Immunologic reviews highlight the need to define correlates of protection and to account for antigenic evolution in VP1–VP3.
Conclusion
EV D68 has developed into a more observable respiratory virus. This has become a global problem through the intermediation of severe respiratory disease and AFM in children. Genetic changes in capsid and non-structural proteins have augmented the usage of receptors, multiplication capacity, and immune evasion, and host inflammatory and antiviral systems are critical in defining the severity of the disease. No direct-acting or host-directed antiviral strategies are currently available, and those that target innate immunity (TLR7, SOCS3, TREM 1, etc.) and viral proteases/polymerase are the most promising. A combination of genomic surveillance, mechanistic models of pathogenesis (spinal cord organoid and optimized mouse models), and future research on translational antivirals is needed to lower future EV D68 outbreaks and AFM burdens.
References
- Aguglia, G., Coyne, C., Dermody, T., Williams, J., & Freeman, M. (2023). Contemporary enterovirus-D68 isolates infect human spinal cord organoids. mBio, 14. https://doi.org/10.1128/mbio.01058-23
- Ayudhya, S., Laksono, B., & Van Riel, D. (2021). The pathogenesis and virulence of enterovirus-D68 infection. Virulence, 12, 2060 – 2072. https://doi.org/10.1080/21505594.2021.1960106
- Chen, H., Huang, M., Hou, B., Liu, Z., Tan, R., Cui, L., Wang, T., & Wang, Z. (2025). The structural protein VP3 of enterovirus D68 interacts with MAVS to inhibit the NF-κB signaling pathway. Journal of Virology, 99. https://doi.org/10.1128/jvi.00163-25
- Devries, M., Bochkov, Y., Evans, M., Gern, J., & Jackson, D. (2023). Recent Clinical Isolates of Enterovirus D68 Have Increased Replication and Induce Enhanced Epithelial Immune Response Compared to the Prototype Fermon Strain. Viruses, 15. https://doi.org/10.3390/v15061291
- Duan, W., Li, J., Liu, Y., Wang, R., Yang, Q., Li, H., Liang, Y., Shi, Q., Wang, J., Xiao, J., Zhou, J., Sun, Q., & Zhang, Y. (2025). Spinal Cord Injury in Enterovirus D68 Infection: Mechanisms and Pathophysiology in a Mouse Model. Viruses, 17. https://doi.org/10.3390/v17111478
- Elrick, M., Pekosz, A., & Duggal, P. (2021). Enterovirus D68 molecular and cellular biology and pathogenesis. The Journal of Biological Chemistry, 296. https://doi.org/10.1016/j.jbc.2021.100317
- Fall, A., Abdullah, O., Han, L., Norton, J., Gallagher, N., Forman, M., Morris, C., Klein, E., & Mostafa, H. (2024). Enterovirus D68: Genomic and Clinical Comparison of 2 Seasons of Increased Viral Circulation and Discrepant Incidence of Acute Flaccid Myelitis—Maryland, USA. Open Forum Infectious Diseases, 11. https://doi.org/10.1093/ofid/ofae656
- Filipe, I., Guedes, M., Zdobnov, E., & Tapparel, C. (2021). Enterovirus D: A Small but Versatile Species. Microorganisms, 9. https://doi.org/10.3390/microorganisms9081758
- Freeman, M., Wells, A., Ciomperlik-Patton, J., Myerburg, M., Anstadt, J., & Coyne, C. (2021). Respiratory and intestinal epithelial cells exhibit differential susceptibility and innate immune responses to contemporary EV-D68 isolates. eLife, 10. https://doi.org/10.7554/elife.66687
- Grizer, C. S., Messacar, K., & Mattapallil, J. J. (2024). Enterovirus-D68 – a reemerging non-polio enterovirus that causes severe respiratory and neurological disease in children. Frontiers in Virology, 4. https://doi.org/10.3389/fviro.2024.1328457
- Hu, Y., Musharrafieh, R., Zheng, M., & Wang, J. (2020). Enterovirus D68 antivirals: past, present, and future. ACS infectious diseases. https://doi.org/10.1021/acsinfecdis.0c00120
- Li, Y., Qu, L., Tang, Y., Ni, F., Shen, S., Guo, H., Yu, X., & Wei, W. (2025). Cullin 3-mediated ubiquitination restricts enterovirus D68 replication and is counteracted by viral protease 3C. Journal of Virology, 99. https://doi.org/10.1128/jvi.00354-25
- Li, J., Yang, S., Liu, S., Chen, Y., Liu, H., Su, Y., Liu, R., Cui, Y., Song, Y., Teng, Y., & Wang, T. (2021). Transcriptomic Profiling Reveals a Role for TREM-1 Activation in Enterovirus D68 Infection-Induced Proinflammatory Responses. Frontiers in Immunology, 12. https://doi.org/10.3389/fimmu.2021.749618
- Li, H., Huang, Y., Yang, Q., Zhang, Z., Shen, S., Guo, H., & Wei, W. (2024). Pharmacological activation of TLR7 exerts inhibition on the replication of EV-D68 in respiratory cells. Journal of Virology, 98. https://doi.org/10.1128/jvi.00434-24
- Naeem, A., Bello, M., & Bosaeed, M. (2025). Insights Into Enterovirus D68 Immunology: Unraveling the Mysteries of Host‐Pathogen Interactions. Immunity, Inflammation and Disease, 13. https://doi.org/10.1002/iid3.70117
- Rahajamanana, V., Thériault, M., Rabezanahary, H., Sahnoun, Y., Mallet, M., Isabel, S., Trottier, S., & Baz, M. (2025). Advances in the Treatment of Enterovirus-D68 and Rhinovirus Respiratory Infections. Infectious Disease Reports, 17. https://doi.org/10.3390/idr17030061
- Sun, J., Hu, X., & Yu, X. (2019). Current Understanding of Human Enterovirus D68. Viruses, 11. https://doi.org/10.3390/v11060490
- Xiang, Z., Tian, Z., Wang, G., Liu, L., Li, K., Wang, W., Lei, X., Ren, L., & Wang, J. (2023). CD74 Interacts with Proteins of Enterovirus D68 To Inhibit Virus Replication. Microbiology Spectrum, 11. https://doi.org/10.1128/spectrum.00801-23
- Yang, Q., Li, H., Li, Z., Yang, J., Zhang, Z., Zhang, L., Guo, H., & Wei, W. (2023). Pterostilbene, an active constituent of blueberries, enhances innate immune activation and restricts enterovirus D68 infection. Frontiers in Immunology, 14. https://doi.org/10.3389/fimmu.2023.1118933
- Zhang, Y., Xu, L., Zhang, Z., Su, X., Wang, Z., & Wang, T. (2023). Enterovirus D68 infection upregulates SOCS3 expression to inhibit JAK-STAT3 signaling and antagonize the innate interferon response of the host. Virologica Sinica, 38, 755-766. https://doi.org/10.1016/j.virs.2023.08.007
- Zhu, Y., Wang, L., & Shen, J. (2026). Enterovirus D68 Sequence Variations and Pathogenicity: A Review. Viruses. https://doi.org/10.3390/v18010073
