The process of drug discovery and development is complex, requiring many different skills over a time frame of years. It spans about 10-15 years.

This process visualizes the input and its output at another end. The drug discovery pipeline consists of four stages.
- Research and Discovery: In-vitro and In-vivo testing.
- Preclinical development: Covers the identification of the drug target.
- Clinical Trials: Phase I, Phase II, and Phase III human clinical trials.
- Review/ Marketing: Regulatory review and marketing application.
Each of these stages is designed to ensure that the drug is safe, effective, and of high quality before it reaches patients’ use.
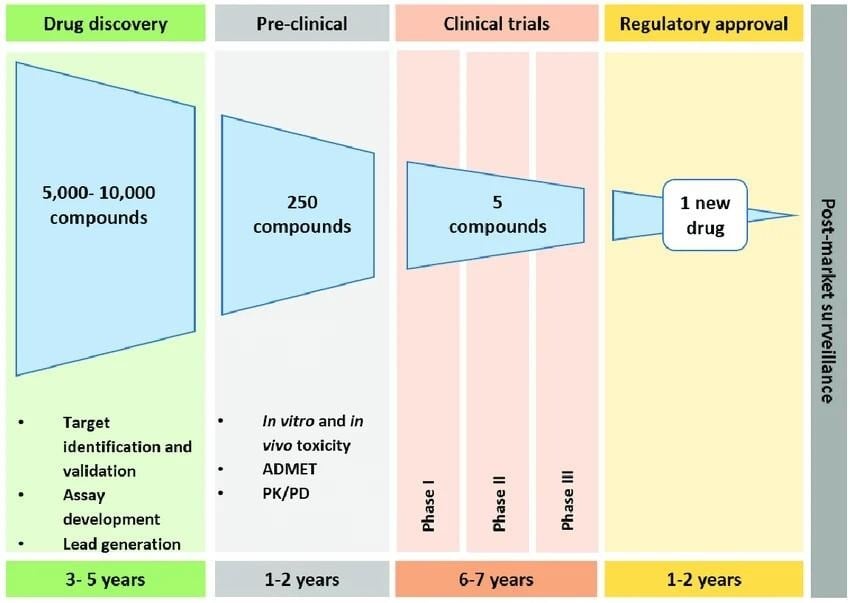
Sources: https://www.researchgate.net/figure/Drug-discovery-and-development-timeline-The-current-drug-approval-pipeline-can-take-15_fig1_308045230
Target Identification and Validation
The target site for drug action plays a significant role in disease management or symptom relief. It is important to find out what the drug will affect. Identification of potential therapeutic targets informs downstream decisions, from compound screening to clinical trial design. Thousands of compounds are screened to identify leads for drug development by determining which genes, proteins, or pathways should be modulated to treat disease effectively. In the modern era, genomics, transcriptomics, proteomics, and AI-driven data integration are key tools for target identification. An ideal target should be effective, safe, and druggable with the properties of:
- The target has a distinct role in the pathophysiology of a disease and/or is a disease-modifying agent.
- Target expression is distributed unevenly throughout the body.
- Availability of 3D- structure of the target assessable for druggability.
- The target is easily ‘assayable’, enabling high-throughput screening.
- Phenotypic data prediction (promising toxicity profile, potential adverse effects) of the target.
- The proposed target has favorable intellectual property.
Target Validation is the process of demonstrating the functional role of the identified target in the disease phenotype. Target Validation should be done experimentally according to the proposed mode of action. The target validation covers discovering a biomolecule of interest, evaluating its potential as a target, designing a bioassay to measure its biological activity, constructing a high-throughput screening platform, and performing screening to identify a lead compound. The multiple steps used in target validation help in increasing confidence and preventing drug failure in later stages. The steps are broken down into two key steps:
- Reproducibility
Once the drug target is identified, the first step is to repeat the experiment to confirm that it can be successfully reproduced.
- Introduce variation to the ligand (drug)-target-environment
Variation may make drug binding less effective, more effective, or not at all. The changes to the drug, the biological environment, or the target itself can all influence how well the drug works. For example, if the properties of the drug are changed, its ability to bind to the target can increase or decrease. The effect of the drug may also vary depending on the type of cell or tissue, or in some cases, it may remain the same.
The validation confirms that the drug turns out to be safe and efficacious in a patient.
High-Throughput Screening (HTS)
This is an advanced technique used in drug discovery that evaluates thousands to millions of chemical compounds quickly for a biological activity against a specific target site (such as cells, receptors, or enzymes). Compounds are tested in microplates, and their activity is measured through a cell-based assay or biochemical assay. It helps to find potential compounds that can be further used in the development of the drug. HTS identifies hit compounds that show activity against the target. The methods used to evaluate hits are:
- Confirmatory Test: Re-test of initial hits using the HTS assay.
- Dose response curve: Test compounds at varying concentrations to determine half- maximal activity.
- Orthogonal testing: Use of a different assay or technology.
- Secondary screening: Evaluation of hits in a functional cell-based assay.
- Synthetic tractability: Assess the feasibility of synthesis, scalability, and cost of production.
- Biophysical testing: Use of different technologies to study binding properties, kinetics, thermodynamics, and to rule out nonspecific interactions.
- Hit ranking and clustering: Grouping under validation test performance.
Hit-to-Lead and Lead Optimization
After the identification of potential compounds “hits”, it is refined to a “lead” compound (potency and selectivity). This process involves the removal of false hits and the confirmation of the activity of a true hit. Structural- Activity studies are performed to understand how chemical modifications affect activity.
The efficacy, potency, and ADME properties of the drug are enhanced by subsequently refining the lead compounds, implementing a process called lead optimization. Dozens of compounds are funneled out by the lead optimization technique, implementing the In-vivo technique using animal models and tools for identifying physicochemical properties, pharmacokinetic properties, and toxicological aspects. The drug discovery process ends when one lead compound is found for a drug candidate, and the process of drug development starts.
Preclinical Safety Testing (In Vitro and In Vivo)
Once a compound or bio-therapeutic molecule has been selected as a lead for further
development, it is ready to leave the discovery phase and move into preclinical development. Preclinical studies are carefully designed to mimic the conditions and variables similar to a human clinical trial. Preclinical studies involve In-vitro and In-vivo testing of the drug. In-vitro studies are performed in controlled laboratory environments using cells or tissues to assess mechanisms of action, toxicity, and pharmacological effects, whereas In-vivo studies are conducted in animal models to evaluate the drug’s pharmacokinetics and pharmacodynamics. The study design involves determining the sample size, duration of study, and endpoints to measure a drug’s efficacy, safety, and potency. The stages of Preclinical safety testing are:
- Drug Discovery
- Formulation Development
- Pharmacology
- Toxicology
The Investigational New Drug (IND) Application
After preclinical studies and before starting a clinical trial, the researchers or sponsors must submit information on the plans of the clinical trial and how the drug will be manufactured to officially consider a new drug to regulatory authorities such as the FDA. The regulatory bodies of the country will review the results from the IND and evaluate the intervention’s potential risks and benefits before determining if the clinical trial in humans can take place. The IND application acts as a bridge between preclinical research and human clinical trials, ensuring the drug is safe, effective, and potent after the approval of the regulatory body.
Clinical Trial Phases (Phase I, II, and III)
If preclinical studies indicate that the drug is potentially safe and effective in humans, clinical development begins with four sequential phases, each phase with specific objectives related to safety, efficacy, and drug performance involving human volunteers.
Phase I Clinical trials: Human Pharmacology
About tens of healthy human volunteers are involved. The purpose of the phase I trial is to determine whether the treatment is safe. The drug is administered to human volunteers to check the maximum dose that can be tolerated without altering the safety. This clinical trial is done to investigate what the drug does to the body and what the body does with the drug, and its safety. This information is later used for the phase II trial.
Phase II Clinical Trial: Exploratory
When a compound is found to be safe and well-tolerated at doses, the phase II trial begins with a study of short-term adverse events. The efficacy of drugs in hundreds of volunteers with the disease for several months to years is tested and monitored. The purpose of this trial is to determine efficacy, safety, and optimal dosing of the treatment plan. The clinical trial in Phase II can be randomized, blinded, and placebo-controlled or active-controlled to ensure the best study of treatment. The result of this test is used to conduct the phase III trial.
Phase III Clinical Trial: Large-scale Confirmatory
This trial is designed to replicate the phase II efficacy data for the drug candidate in thousands of patients over several years, to determine if any safety and efficacy issues arise over the period of the trial phase. The purpose of the study is to evaluate how the new medication works in comparison to existing medications for the same condition. This clinical trial is randomized as well as double blind. It is designed to identify the safety issues that might have been absent in smaller trials. Sometimes a phase III trial is designed to support a marketing approach, or to extend the use of the drug to different types of patients (label expansion).
Regulatory Submission and Review
After a successful clinical trial, a pharmaceutical company must submit a formal application to the regulatory authorities of its own country to obtain approval for marketing of a drug. Different countries have different types of submission, including NDA, BLA, MAA, and NDS.
- From Phase III to Phase IV- US
When a phase III clinical trial shows a new drug is more effective and safer than the current treatment, a new drug application (NDA) is submitted to the FDA for approval. Based on the documented data, the FDA approves or rejects the treatment plan. According to the FDA, about 70 % of drugs move from Phase I to Phase II, 33% from Phase II to Phase III, and 25-30% from Phase III to Phase IV clinical trials.
- From Phase III to Phase IV- Europe
The Marketing Authorization Application (MAA) is submitted to the European Medicines Agency (EMA) after Phase II and Phase III trials meet their objectives, and the benefit-versus-risk ratio of the medicine or targeted treatment is assessed. EMA includes all the relevant preclinical and clinical data that have been gathered over the years since the drug candidate was first identified.
- From Phase III to Phase IV- Australia
In Australia, the TGA (Therapeutic Goods Administration) is responsible for checking the phases involved with registration by evaluating the safety, effectiveness, and benefits versus risk ratio. It also includes side effects, toxicities, long-term sequels, and the nature of the medication. If the TGA is satisfied that the benefits of the product outweigh associated risks, then the drug will be approved and registered.
- From Phase III to Phase IV- Canada
The New Drug Submission (NDS) is submitted to Health Canada for drug approval in Canada. It contains similar documents and information as the NDA to ensure the drug meets regulatory standards.
- From Phase III to Phase IV- UK
The Marketing Authorization Application (MAA) for marketing approval is submitted to the Medicines and Healthcare Products Regulatory Agency (MHRA). MAA includes similar documents to NDA.
Manufacturing and Scale-Up
Manufacturing and scale-up in drug discovery represent the transition from laboratory research to large-scale production of a drug for clinical use and commercialization. Once a lead compound has been identified, its production must be scaled up from grams to kilograms in pilot plants and eventually to industrial-scale manufacturing. This stage includes process optimization, quality control, and adherence to Good Manufacturing Practices (GMP). Scale-up and manufacturing are essential to support the supply of sufficient materials for clinical trials, regulatory approval, and eventual market supply.
Post-Market Surveillance (Pharmacovigilance)
Post-Marketing Surveillance is also known as phase IV of a clinical trial. The purpose of this surveillance is to get more information about long-term safety, efficacy, and to determine the long-term benefits and risks of the drug after the regulatory body of the country approves a drug for marketing. This is an observational study that collects information from the drug on the market and patients who are using it. Even though the drug may have been approved for sale, the manufacturer is required to submit a strategy for monitoring and dealing with side effects; this is a Risk Evaluation and Mitigation Strategy (REMS) or Risk Management Plan (RMP).
Pharmacovigilance/ Post-marketing surveillance is undertaken by passive or active surveillance of the patient population taking the drug. Spontaneous reporting of an adverse event to the regulators by patients and doctors is a common example of passive reporting. During preclinical and clinical phases, safety data are analyzed to identify potential risks and to find out whether they are related to the drug. Pharmacovigilance also includes active reporting of safety data, long-term effects, rare adverse reactions, drug interactions, etc., from the drug development and discovery phase to the drug in the market. It is an ongoing commitment to patient safety, continuously balancing benefits and risks.
Conclusion
The drug discovery and development, clinical trials, and approval processes are a multi-process and time-consuming pathway designed to ensure that new medications are safe, effective, potent, and reliable. From identifying and validating a suitable target to screening compounds to hit and lead identification and optimization.
Each process plays a vital role in promoting patient health by minimizing risk and maximizing therapeutic benefits. Even after approval of the drug, monitoring is endless, and the pharmacovigilance safeguards patient health.
The consistent manufacturing and regulation of the medication ensure the wide availability of medications by minimizing uncertainty and maximizing therapeutic values. Hence, every step of drug development and discovery acts as a filter and safeguard to provide patient compliance.
References
- DRUG DEVELOPMENT www.acuteleuk.org. (n.d.). https://acuteleuk.org/wp-content/uploads/2021/01/ALAN-Webinar_Drug-Development.pdf
- FDA. (2018). Step 2: Preclinical Research. U.S. Food and Drug Administration. https://www.fda.gov/patients/drug-development-process/step-2-preclinical-research
- Gashaw, I., Ellinghaus, P., Sommer, A., & Asadullah, K. (2011). What makes a good drug target? Drug Discovery Today, 16(23-24), 1037–1043. https://doi.org/10.1016/j.drudis.2011.09.007
- Hughes, J., Rees, S., Kalindjian, S., & Philpott, K. (2011). Principles of Early Drug Discovery. British Journal of Pharmacology, 162(6), 1239–1249. https://doi.org/10.1111/j.1476-5381.2010.01127.x
- Laurent, A. (n.d.). Drug Development Pipeline: A Complete Guide to All Phases Drug Development Pipeline: A Complete Guide to All Phases. https://intuitionlabs.ai/pdfs/drug-development-pipeline-a-complete-guide-to-all-phases.pdf
- SCALE-UP & MANUFACTURING – Smart Formulation, Processing & Engineering Solutions to Solve Drug Product Scale-Up & Manufacturing Challenges With Minimum to No Regulatory Impact. (2018, August 13). Drug Development and Delivery. https://drug-dev.com/scale-up-manufacturing-smart-formulation-processing-engineering-solutions-to-solve-drug-product-scale-up-manufacturing-challenges-with-minimum-to-no-regulatory-impact/
- Szymański, P., Markowicz, M., & Mikiciuk-Olasik, E. (2011). Adaptation of High-Throughput Screening in Drug Discovery—Toxicological Screening Tests. International Journal of Molecular Sciences, 13(1), 427–452. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3269696/
