Cystine-Lactose-Electrolyte-Deficient (CLED) medium, first described by Sandys and later modified by Mackey and Sandys, is generally used for diagnostic routine urinary bacteriology as a non-selective medium capable of supporting the growth of most urinary pathogens. CLED Agar is a differential medium used for the isolation and enumeration of bacteria from urine. The medium supports the growth of all urinary potential pathogens and provides distinct colony morphology. It supports the growth of urinary pathogens and contaminants and gives good colonial differentiation without the spread of Proteus species due to its lack of electrolytes.
Composition of CLED Agar
| Constituents | gm/Litre |
| Lactose | 10.0 |
| Enzymatic Digest of Gelatin | 4.0 |
| Enzymatic Digest of Casein | 4.0 |
| Beef extract | 3.0 |
| L-Cystine | 0.128 |
| Bromothymol Blue | 0.02 |
| Agar | 15.0 |
pH 7.3 +/- 0.2
Principle of CLED Agar
The CLED comprises of enzymatic digest of Casein, an enzymatic digest of gelatin, and beef extract which provide the nitrogen, vitamins, minerals, and amino acids essential for growth. L-cystine supports the growth of the cysteine-dependent dwarf coliform colony. Lactose is a fermentable carbohydrate that provides carbon and energy. Organisms capable of fermenting lactose will lower the pH and change the color of the medium from green to yellow. The color change is indicated by the presence of the pH indicator Bromthymol Blue. Agar present in the medium is the solidifying agent.
Preparation of CLED Agar
- Suspend 36 gram of the medium in one liter of distilled water.
- Mix well and heat with frequent agitation and boil for one minute until complete to dissolution.
- Autoclave at 121°C for 15 minutes.
- Cool to 50°C, mix well and dispense into plates. When the medium is solidified, invert the plates to avoid excess moisture. The prepared medium should be stored at 8-15°C.
Colony characteristics of various organisms on CLED Agar
Various cultural responses are shown by different microorganisms on CLED Agar at the appropriate atmosphere and temperature after 18 – 24 hours incubation are as follows:
| Organism | Colony Morphology |
| Escherichia coli | Large, elevated, yellow, opaque colonies with a center more intense yellow; yellowish medium |
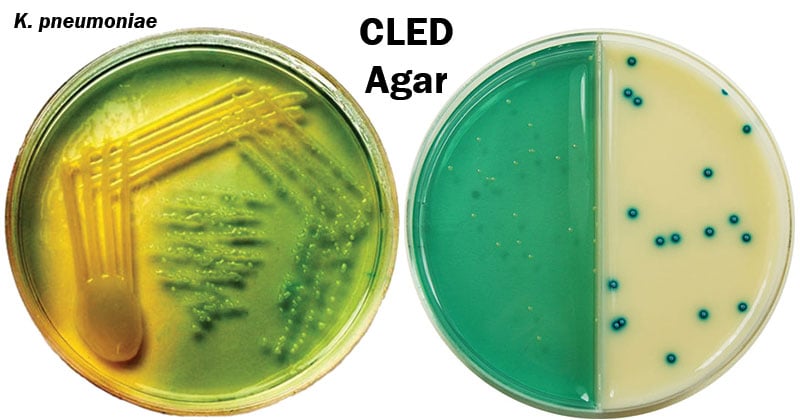
| Klebsiella species | extremely mucoid colonies varying in color from yellow to whitish-blue. yellowish medium. |
| Proteus species | translucent blue colonies are usually smaller than Escherichia coli. blue-green to blue medium. |
| Salmonella species | flat blue colonies |
| Pseudomonas aeruginosa | green colonies with a typical matte surface and rough periphery. “Sweet” odor. Blue-green agar |
| Enterococcus faecalis | yellow colonies about 0.5 mm in diameter. yellow medium |
| Staphylococcus aureus | deep yellow colonies about 0.75 mm diameter, uniform in color. yellow medium |
| Corynebacteria | very small grey colonies |
| Lactobacilli | similar to corynebacteria but with a rougher surface |
Uses of CLED Agar
- It is used for routine urine culture and uses for growth and enumeration of urinary tract organisms.
- It helps in the differentiation of lactose fermenter and lactose non-fermenter.
- It is suitable for the isolation and counting of many aerobically growing microorganisms, such as Enterobacteriaceae, Pseudomonas, and other non-fermenting Gram negative rods, enterococci, staphylococci, Candida species, and many others from urine specimens.
Limitations of CLED Agar
- Streptococci and other organisms requiring blood or serum for growth may only be insufficiently recovered on this medium or may need extended incubation. Therefore, the specimen should also be cultivated onto a blood agar plate if such organisms are expected.
- Genitourinary pathogens such as Neisseria gonorrhoeae, Gardnerella vaginalis, Chlamydia, Ureaplasma, or other fastidious organisms do not grow on this medium.
- Serological tests using pure cultures are necessary for complete identification.
References
- Conda Lab
- Himedia
- https://www.bd.com/resource.aspx?IDX=8967
- Fallon D, Andrews N, Frodsham D, Gee B, Howe S, Iliffe A, Nye KJand R E Warren RE (2002). A comparison of the performance of cystine lactose electrolyte deficient (CLED) agar with Oxoid chromogenic urinary tract infection (CUTI) medium for the isolation and presumptive identification of organisms from urine. Journal of Clinical Pathology 55(7):524-529.
- Cheesbrough M., District Laboratory Practice in Tropical Countries: Part 2:.Cambridge University Press.
- Ronald M. Atlas and James W. Snyder (2014). Handbook of media for clinical and public health microbiology. CRC Press. Taylor & Francis Group, LLC

nice information
Hi, Thanks and very informative articles.
Is it required to use media for pharmaceutical industry where oral liquids or powder are manufactured.
Thanks again.
thanks for good job
Thank you very much. Very useful information for trainees. Wish to add another point “Electrolytes Deficit” stops swarming of Proteus spp on the surface.
beautiful work. very exhaustive.
Thank you so much sir
Kindly pls do more..
So much helpful
THank you for your great information good luck
OK, good luck to you too
Thanks so much for this wonderful explanation. This explanation really help me in my project.
Thank you so much for the notes… Please try to include pictures of each bacterial colonies…