Continuous culture is a continuous process where nutrients are continually added to the bioreactor and the culture broth (containing cells and metabolites) is removed at the same time.
The volume of the culture broth is constant due to a constant feed-in and feed-out rate (i.e consumed nutrients are replaced and toxic metabolites are removed from the culture).
Some advantages of continuous culture are:
- In continuous culture, keeping the working volume constant simplifies culture scale-up based on a constant- power-to-volume strategy.
- It is possible to set up the optimum conditions for maximum and long-term product synthesis.
- Ability to obtain stable product quality (the steady-state consists of homogeneous cell culture with a constant biomass and metabolite concentration).
- It also results in higher productivity per unit volume, as time-consuming tasks, such as cleaning and sterilization are unnecessary.
- Cultures in a steady-state can last for days, weeks, or even months, thus greatly reducing the downtime and making the process more economically competitive.

Principle of Continuous Culture
- A continuous flow system consists of a reactor into which reactants are pumped at a steady rate and from which products are emitted.
- The factors governing their operation are:
- how material passes through the reactor (which depends upon its design);
- the kinetics of the reaction taking place.
- In continuous culture, growth-limiting nutrients can be maintained at steady-state concentrations, which permits microorganisms to grow at submaximal rates.
- In a steady-state, the cellular growth rate and environmental conditions, like the concentrations of metabolites, stay constant
- Moreover, in continuous culture, parameters such as pH, oxygen tension, the concentration of excretion products, and population densities can easily be monitored and controlled.
Process of Continuous Culture
- In continuous culture, an open system is set up in which one or more feed streams containing the necessary nutrients are fed continuously, while the effluent stream containing the cells, products, and residuals is continuously removed.
- A steady-state is established by maintaining an equal volumetric flow rate for the feed and effluent streams.
- The culture volume is kept constant, and all nutrient concentrations remain at constant steady-state values.
- During this process, the exponential growth phase is prolonged and the formation of byproducts is avoided.
- Continuous fermentation is monitored either by microbial growth activity or by-product formation, and these processes are referred to as-
A. Turbidostat method
- Cell growth is controlled and remains constant in this system, but the flow rate of fresh media varies.
- Cell density is controlled based on the set value for turbidity, which is created by the cell population while fresh media is continuously supplied.
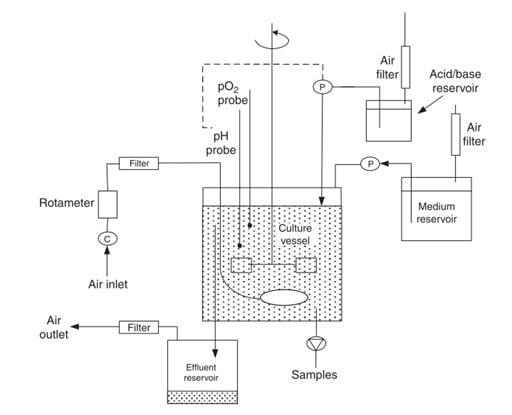
B. Chemostat method
- In a chemostat, the nutrients are continuously supplied at a constant flow rate, and the density of the cells is adjusted according to the supplied essential nutrients for growth.
- In a chemostat, the growth rate is determined by adjusting the concentration of substrates like carbon, nitrogen, and phosphorus.
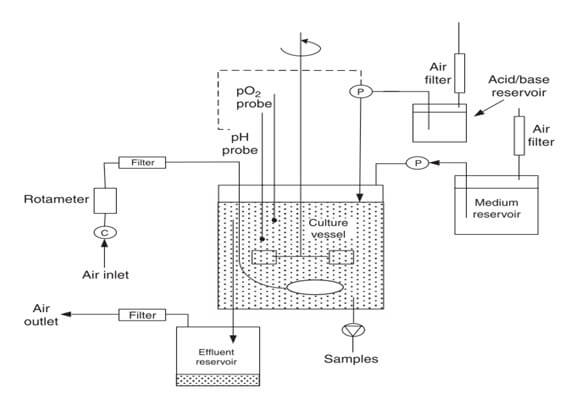
The continuous cultures of chemostat and turbidostat systems have the following criteria:
- Medium and cells are continuously changing
- The cell density is constant
- Steady-state growth
- Open system
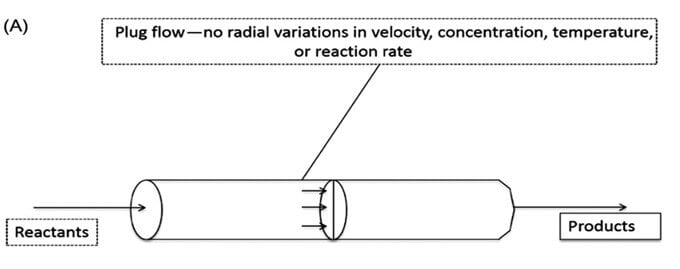
C. Plug-flow reactor
- In this type of continuous culture, the culture solution flows through a tubular reactor without back mixing.
- In a plug flow reactor, nutrients (reactants) enter the reactor in the form of “plugs,” which flow in an axial direction through the reactor.
- The culture medium flows steadily through a tube and the cells are recycled from the outlet to the inlet.
Applications of Continuous Culture
- Continuous culture fermentation has been used for the production of single-cell protein, organic solvents, starter cultures, etc.
- It has been used in the production of beer, fodder yeast, vinegar, baker’s yeast, etc.
- In the industrial production of secondary metabolites (such as antibiotics from a Penicillium or a Streptomyces sp.)
- Continuous culture has been tested for L-lysine-producing C. glutamicum mutant B-6
- It has been used in municipal waste treatment processes.
Limitations of Continuous Culture
- In the long-term cultivation process, sterility maintenance can be tricky, and downstream processing can prove challenging.
- Controlling the production of some non-growth-related products is not easy Because of this, continuous culture often requires fed-batch culturing as well as continuous nutrient supply.
- As a result of the viscosity and heterogeneous nature of the mixture, it may be challenging to maintain filamentous organisms.
- If a faster-growing strain overtakes the original product strain, it may be lost over time.
References
- Blaby, I. K. (2011). Modes of Culture / Microbial. In Comprehensive Biotechnology (Second Edi, Vol. 1). Elsevier B.V. https://doi.org/10.1016/B978-0-08-088504-9.00034-9
- Brautaset, T., & Ellingsen, T. E. (2011). 3.47 – Lysine: Industrial Uses and Production. https://doi.org/10.1016/B978-0-08-088504-9.00220-8
- Ghosh, B., Bhattacharya, D., & Mukhopadhyay, M. (2018). Use of Fermentation Technology for Value-Added Industrial Research. Principles and Applications of Fermentation Technology, August, 141–161. https://doi.org/10.1002/9781119460381.ch8
- Herbert, D., Elsworth, R., & Telling, R. C. (1956). The Continuous Culture of Bacteria; a Theoretical and Experimental Study.
- Jaibiba, P., Vignesh, S. N., & Hariharan, S. (2020). Working principle of typical bioreactors. In Bioreactors. INC. https://doi.org/10.1016/B978-0-12-821264-6.00010-3
- Kuenen, J. G., & Johnson, O. J. (2009). Continuous Cultures (Chemostats). Elsevier Inc.
- Kuila A, Sharma V (2018). Principles and Applications of Fermentation Technology. John Wiley & Sons, Inc. https://doi.org/10.1002/9781119460381.
- Najafpour, G. D. (2015). Biochemical engineering and biotechnology. Elsevier. https://doi.org/10.1016/B978-044452845-2/50005-7.
- Paulová, L. (2014). Advanced Fermentation Processes Relationship between butanol efflux and butanol tolerance of Clostridia View project Yeast biotechnology View project. https://doi.org/10.1201/b15426-6
- Saran, S., Malaviya, A., & Chaubey, A. (2019). 1 introduction, Scope, and Significance of Fermentation Technology.Ch1. 1, 1–25.
- Srivastava, A. K. (2011). 2.38 – Fed-Batch Fermentation – Design Strategies. Comprehensive Biotechnology, 1, 515–526. https://doi.org/10.1016/B978-0-08-088504-9.00112-4
- Yang, Y., & Sha, M. (2017). A Beginner’s Guide to Bioprocess Modes-Batch, Fed-Batch, and Continuous Fermentation.

good work
it is really useful. Thank you so much