Colorado tick fever virus (CTFV) is a tick-borne, double-stranded RNA (dsRNA) arbovirus that causes Colorado tick fever (CTF), an acute systemic febrile illness in humans.

- CTFV is endemic in mountainous regions of western North America, especially where its principal vector, the Rocky Mountain wood tick (Dermacentor andersoni), is abundant.
- Typical disease is biphasic fever with headache, myalgia, and marked leukopenia; severe disease involves meningoencephalitis, hemorrhagic manifestations, and rarely death, especially in children.
- CTFV is neglected compared with other tick-borne viruses: case numbers are low, underdiagnosis is common, and molecular/virological tools have only recently improved (reverse genetics, standardized RT-qPCR, genome-wide sequencing).
CTFV offers a model for prolonged viremia, persistence in erythrocytes, and dsRNA arbovirus pathogenesis, with relevance to other reoviruses and orbiviruses.
Taxonomy and Classification of Colorado tick fever virus (CTFV)
- Family: Reoviridae → Subfamily: Spinareovirinae → Genus: Coltivirus.
- Historically placed in genus Orbivirus; reassigned to Coltivirus as genomic and structural features were clarified.
Key taxonomic points:
- Genome: 12-segmented dsRNA, encoding 13 viral proteins (VP1–VP12; VP9/VP9′ by read-through).
- CTFV is the type species of genus Coltivirus; other recognized coltiviruses include Eyach virus (Europe), Salmon River virus, California hare coltivirus (CTFV-Ca), Kundal virus, Tarumizu tick virus, and Tai Forest reovirus.
- Genomic analyses indicate:
-Salmon River virus is a strain within the CTFV species (>90% aa identity in most segments).
-CTFV-Ca is genetically distinct and better classified as a separate Coltivirus species.
Structure and Morphology of Colorado tick fever virus (CTFV)
- Non-enveloped, icosahedral virion with a triple-layered capsid, typical of reoviruses.
-Inner and middle protein shells (core), plus an outer capsid layer; overall diameter ~70–80 nm (inferred from Reoviridae; specific CTFV EM dimensions reported as 60–80 nm).
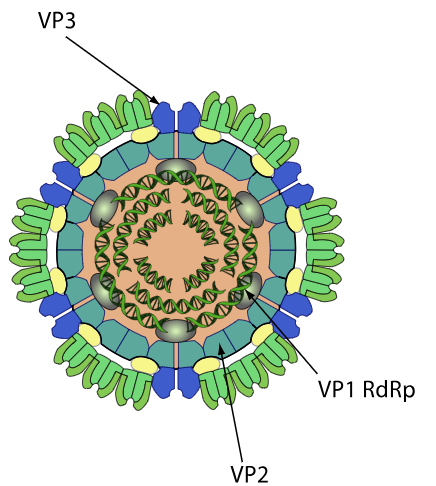
- Triple capsid structure:
-The inner core houses RNA-dependent RNA polymerase and capping enzymes associated with each genome segment.
-Outer capsid proteins mediate cell attachment and entry, analogous to outer capsid proteins of orbiviruses.
Biological implications:
- Non-enveloped, highly stable virions enable environmental persistence, including survival in ticks between blood meals and during overwintering.
- The triple shell protects dsRNA from host pattern recognition receptors (PRRs), limiting exposure of PAMPs during entry and early replication.
Genome Organization and Proteins of Colorado tick fever virus (CTFV)
Figure 2: Genome composition and virion structure of reoviruses based on the genome of CTFV (Atkinson, Barry. (2016). The Characterisation of Arboviral Zoonoses in Central Asia (PhD Thesis)
- Genome architecture
-12 linear dsRNA segments; CTFV is unique among orbivirus-like viruses in having 12, whereas typical orbiviruses have 10.
-Total coding capacity: 13 proteins (VP1–VP12; VP9 and VP9′ from segment 9 via stop-codon read-through).
- Segment/protein overview (based largely on coltivirus comparative genomics; detailed biochemical assignment remains incomplete):
Segment 1 – VP1
- Likely RNA-dependent RNA polymerase (RdRp), as in other Reoviridae.
Segment 2 – VP2
- Putative core structural protein.
Segment 3 – VP3
- Likely capping enzyme or core protein (guanylyltransferase/methyltransferase activity predicted).
Segment 4 – VP4
- Highly variable among strains; evidence of reassortment in segment 4 indicates its role in antigenicity or host interaction.
Segments 5–8 – VP5–VP8
- Structural/non-structural proteins with roles in core stabilization, morphogenesis, and possibly cell-to-cell spread (orthology to orbivirus VP5/VP7/NS1 not fully resolved).
Segment 9 – VP9, VP9′
- Encodes two proteins via stop-codon read-through; inferred outer capsid or cell-attachment proteins.
Segments 10–12 – VP10–VP12
- Likely outer capsid, non-structural, and accessory proteins; VP12 has been used as an insertion site for reporter genes in reverse genetics systems, indicating tolerance for C-terminal fusions.
- Genomic diversity
-CTFV strains (e.g., Florio prototype, Salmon River strains) exhibit high amino-acid identity (>90%) across most segments, except segment 4, where divergence suggests selection and reassortment.
-Evidence for natural reassortment among CTFV strains in the field (Bitterroot Valley) based on loci from segments 9–12.
Replication Cycle of Colorado tick fever virus (CTFV)
Direct experimental detail is limited; most understanding is extrapolated from Reoviridae/Coltivirus biology, now accessible through reverse genetics for CTFV.
- Attachment and entry
-Mediated by outer capsid proteins (likely VP9/VP12 analogs) binding to receptors on hematopoietic progenitors, endothelial cells, and possibly erythrocyte precursors.
-Virions enter via receptor-mediated endocytosis and undergo conformational changes in endosomes.
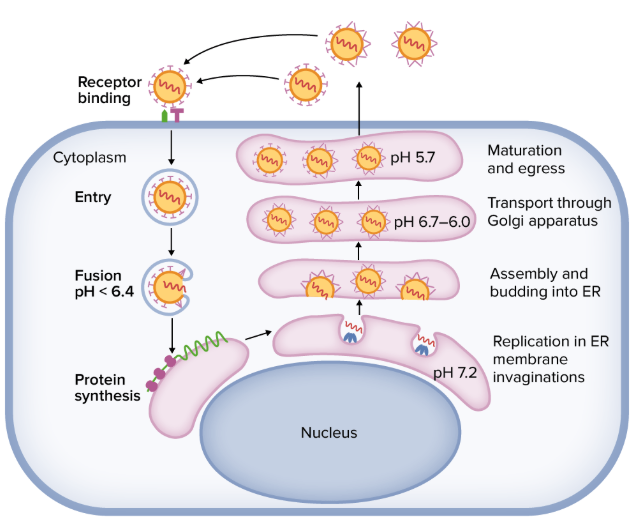
- Uncoating and core transcription
-Outer capsid is removed in endosomes; core particle is released into the cytoplasm.
-dsRNA remains enclosed; viral polymerase transcribes positive-sense mRNAs from each genome segment within the core, minimizing PRR exposure.
- Translation and replication
-Viral mRNAs are exported to the cytoplasm and translated into structural and non-structural proteins.
-Newly synthesized polymerase and core proteins assemble with positive-strand RNAs, which are then copied into dsRNA, forming progeny genome segments inside assembling cores.
- Assembly and egress
-Assembly likely occurs in cytoplasmic viral inclusion bodies, as described for related orbiviruses.
-CTFV can cause cytopathic effects and apoptotic cell death, especially in endothelial cells, facilitating virus release.
-In hematopoietic progenitor cells and erythroid precursors, infection is non-lytic and persistent, leading to prolonged viremia.
- Reverse genetics
A plasmid-based reverse genetics system now enables:
- Rescue of recombinant CTFV from cDNA in T7 polymerase-expressing cells.
- Generation of monoreassortants and reporter viruses (e.g., VP12-2A-NanoLuc fusions).
-These tools will clarify segment functions, replication steps, and virulence determinants.
Pathogenesis and Host Immune Response to Colorado tick fever virus (CTFV)
Cellular targets and persistence
- CTFV infects hematopoietic progenitor cells and subsequently erythrocytes, resulting in prolonged cell-associated viremia for 3–4 months despite the development of neutralizing antibodies.
-Virus in erythrocytes is protected from circulating antibody and innate defenses, promoting maintenance in nature and possibly immune modulation.
- CTFV also infects endothelial cells:
-Human dermal microvascular endothelial cells (HMEC-1) are susceptible and permissive.
Infection causes:
- Dose-dependent decrease in transendothelial electrical resistance (increased vascular permeability).
- Morphological cytopathic effects.
- Caspase-3/7-dependent apoptosis; pan-caspase inhibitor Z-VAD-FMK reduces cell death and lowers viral titers, indicating apoptosis facilitates replication.
Immunopathology
Hematologic Abnormalities and Bone Marrow Involvement
- Leukopenia and thrombocytopenia as immunopathologic signatures
-Profound leukopenia (low WBC) and thrombocytopenia (low platelets) are hallmark laboratory findings.
-Mechanistic contributors:
- Direct infection of hematopoietic progenitors may impair proliferation and differentiation of myeloid and megakaryocytic lineages.
- Cytokine-mediated suppression of bone marrow during systemic inflammation (e.g., high IFNs, TNF-α) can transiently reduce hematopoiesis.
- Enhanced peripheral destruction or sequestration of platelets and leukocytes in the spleen and liver during immune activation.
- Immunopathologic consequences
-Leukopenia predisposes to secondary bacterial infections, especially in hospitalized patients.
-Thrombocytopenia exacerbates any endothelial injury, facilitating petechiae, mucosal bleeding, and, rarely, more severe hemorrhagic manifestations.
Vascular Injury, Permeability, and Hemorrhagic Features
- Endothelial apoptosis as a central lesion
-CTFV induces caspase-3/7–dependent apoptosis in human endothelial cells, and blocking caspases both reduces apoptosis and decreases viral titers, proving that host cell death is intertwined with viral replication.
-Apoptotic endothelial cell loss leads to:
- Disruption of tight junctions and adherens junctions,
- Increased paracellular leak of plasma,
- Exposure of subendothelial collagen and tissue factor initiates coagulation cascades.
- From cell death to clinical hemorrhage
-Microvascular barrier failure manifests clinically as:
- Flushing, petechial rash, epistaxis, and mild mucosal bleeding in some patients.
-When combined with thrombocytopenia, even modest endothelial injury can result in clinically apparent hemorrhagic diathesis, particularly in pediatric cases with immature homeostatic and immune systems.
- Role of cytokines and systemic inflammation
-Although detailed cytokine profiles for CTFV are limited, analogy with other tick-borne viral hemorrhagic syndromes suggests:
- Elevated IL-6, IL-8, and TNF-α may promote endothelial activation, up-regulation of adhesion molecules, and secondary leukocyte-mediated damage.
- Excessive TNF-α and other mediators can enhance coagulopathy by stimulating tissue factor expression and impairing natural anticoagulant pathways.
Fever, Biphasic Course and Systemic Symptoms
- Biphasic fever as a reflection of host–virus dynamics
-Initial febrile peak corresponds to primary viremia and innate immune activation, driven by:
- Pyrogenic cytokines (IL-1β, IL-6, TNF-α),
- Prostaglandin-mediated hypothalamic set-point changes.
-Temporary defervescence may represent partial containment of the virus in specific compartments and transient damping of systemic cytokines.
-Second febrile phase coincides with:
- Continued or recrudescent viremia from bone marrow/erythroid reservoirs,
- Renewed innate and emerging adaptive responses.
- Myalgia, headache, and malaise
-Likely result from:
- Cytokine-mediated effects on muscle metabolism and nociceptor sensitization,
- Release of lactate, prostaglandins, and kinins during systemic inflammation and vascular leakage.
Organ Involvement and Neurologic Complications
- Central nervous system involvement
-In a minority of patients, virus or immune complexes affect the meninges or brain parenchyma, leading to meningitis or meningoencephalitis.
-Possible mechanisms:
- Direct infection of CNS endothelial cells and choroid plexus, increasing blood–brain barrier permeability.
- Immune-mediated injury from infiltrating T cells and activated microglia responding to viral antigen or damaged endothelium.
- Other organ systems
-Mild hepatocellular injury with elevated transaminases can result from:
- Direct infection of liver sinusoidal or Kupffer cells,
- Or “bystander” hepatotoxicity from high cytokine levels.
-Rare reports of myocarditis or pericarditis suggest that immune-mediated inflammation can extend to cardiac tissues in severe disease.
Epidemiology and Transmission of Colorado tick fever virus (CTFV)
- Geographic distribution
-Endemic in western North America, particularly the Rocky Mountain region of the USA and adjacent Canada, typically at elevations 1,200–3,000 m (4,000–10,000 ft).
-Documented risk states in the USA: Colorado, Wyoming, Montana, Utah, Idaho, Oregon; sporadic cases in additional western states via travel.
- Natural cycle
-Vector: primarily Rocky Mountain wood tick, Dermacentor andersoni; in some areas, Dermacentor occidentalis and sympatric ticks may also carry the virus.
-Hosts:
- Small mammals (e.g., golden-mantled ground squirrel, chipmunks) act as reservoir hosts for immature ticks.
-Transmission:
- Transstadial (larva → nymph → adult) but not transovarial.
- Virus present in tick salivary glands and can be transmitted soon after attachment.
- Prevalence in ticks
-Historical and recent data show stable, non-declining prevalence in endemic areas:
- Bitterroot Valley, Montana: 6.6% of 921 adult D. andersoni PCR-positive (2002–2013).
- Far-West surveillance: CTFV-positive D. andersoni prevalence 58% (CO), 21% (WY); 4–15% in D. occidentalis where sympatric.
- Human epidemiology
-2002–2012: 75 CTF cases identified in the USA; median 5/year, incidence highest in Wyoming (3.4/million) and Montana (1.5/million).
-2013–2022: 148 cases, national average incidence 0.04/million; 96 (65%) male, median age 55 years, 98% infected April–July; hospitalization 16%, no deaths reported.
-In Montana, 2020 saw a large increase (21 cases), attributed mainly to:
- Increased outdoor recreation during the COVID-19 pandemic.
- Switch from IFA serology to more sensitive RT-PCR.
Clinical Manifestations of CFTV
- Incubation period
-Typically, 3–4 days (range 1–14 days) after a tick bite.
- Typical illness
-Sudden onset of:
- High fever (often 39–40 °C).
- Chills, severe headache, retro-orbital pain, myalgia, malaise.
-Characteristic biphasic or “saddleback” fever:
- First febrile phase 2–3 days → brief afebrile period (1–3 days) → recurrence of fever 1–3 days.
- Laboratory abnormalities
-Leukopenia and thrombocytopenia are common; mild elevations in liver enzymes may occur.
- Severe disease (uncommon, <5%)
-Neurologic involvement: meningitis, meningoencephalitis, encephalopathy.
-Hemorrhagic complications: bleeding diathesis, DIC; pediatric fatalities reported historically.
-Other reported complications: hepatitis, pericarditis, myocarditis, atypical pneumonia, epididymo-orchitis.
- Outcomes
-15–30% of symptomatic patients require hospitalization, but the case-fatality rate is <1%, with deaths mostly in children or patients with comorbidities.
-Post-acute fatigue may persist for weeks; there is no evidence for chronic active infection in immunocompetent adults despite prolonged viremia.
Laboratory Diagnosis of Colorado tick fever virus (CTFV)
- Specimens
-Acute phase (first 2 weeks): whole blood or serum.
-Convalescent phase: serum for serology.
- Molecular detection
–RT-PCR targeting S2 (segment 2) or other segments:
- Detects viral RNA from day 1 of symptom onset up to at least 6 weeks.
- Highly specific for CTFV vs related coltiviruses.
–RT-qPCR assays are now standardized for clinical and research use, including quantification in plaque assays and culture systems.
- Serology
-IgM/IgG indirect fluorescence assay (IFA), neutralization tests (PRNT).
-Antibody kinetics:
- Often absent in the first 2 weeks; >50% seronegative at 2 weeks.
- Neutralizing antibodies are detectable in >90% by 4 weeks after onset.
-A fourfold rise in neutralizing or IgG titer between acute and convalescent sera confirms infection.
- Other methods
-Virus isolation in Vero or other permissive cells from blood clots or heparinized blood; used mainly in reference labs and for research.
-Immunofluorescence detection of viral antigen in peripheral blood leukocytes or erythrocytes remains a classical method, reflecting cell-associated viremia.
Diagnostic strategy:
- Acute illness ≤14 days: RT-PCR on blood; negative RT-PCR strongly argues against CTF but may require repeat or serology depending on timing.
- >14 days after onset: serology (IFA or PRNT); paired sera best for confirmation.
Treatment and Antiviral Therapy for Colorado tick fever virus (CTFV)
- No licensed virus-specific antivirals.
- Management is supportive:
-Antipyretics, analgesics, hydration.
-Close monitoring for neurologic or hemorrhagic complications.
- Antibiotics
-Doxycycline is often started empirically for suspected tick-borne illness (e.g., RMSF, ehrlichiosis), but has no activity against CTFV; should be discontinued once CTFV is confirmed and bacterial coinfections excluded.
- Experimental directions
-Reverse genetics and robust cell culture systems enable testing of:
- Broad-spectrum antivirals targeting dsRNA replication (e.g., nucleoside analogues),
- Host-directed therapies (e.g., modulation of apoptosis in endothelial cells).
-However, no clinical data yet support any specific antiviral regimen.
Prevention and Control of Colorado tick fever virus (CTFV)
- No vaccine is currently available for CTFV.
- Personal protective measures (primary prevention):
-Avoid tick-infested habitats or stay on cleared trails when hiking in endemic areas.
-Use EPA-registered tick repellents (e.g., DEET, picaridin), and permethrin-treated clothing.
-Wear long sleeves, tuck pants into socks; perform frequent tick checks and prompt removal.
- Public health strategies
-Education of residents and visitors to endemic regions, especially during spring and early summer when D. andersoni adults are most active.
-Enhancement of provider awareness to consider CTF in febrile illness with leukopenia and recent travel to high-elevation western areas.
-Utilization of RT-PCR in state and reference laboratories to improve case detection and surveillance.
- Vector and reservoir control
-Large-scale tick control is impractical in rugged mountainous terrain.
-Local measures (brush management around recreational sites, targeted acaricide use) may reduce tick densities but have not been systematically evaluated for CTF prevention.
- Surveillance
-CTF is reportable in a limited number of states; not nationally notifiable, complicating burden estimates.
-Molecular surveillance of CTFV in ticks and humans has revealed:
- Stable prevalence over decades in some foci.
- Genetic reassortment among strains and emerging lineages (e.g., Salmon River virus, CTFV-Ca) with implications for diagnostics and taxonomy.
Conclusion
Colorado tick fever virus is a classic member of the Coltivirus family. This virus has 12 segments of double-stranded RNA contained within a strong, non-enveloped package. The virus replicates via a natural cycle of Rocky Mountain wood ticks and small mammals out west. When it replicates in humans, it attacks the blood cells, allowing it to remain within the body for a long time.
This results in a double fever and decreased white blood cells. It can also replicate within the lining of the blood vessels, resulting in an inflammatory response that causes severe disease. This virus causes human illness infrequently, which resolves on its own. However, it remains endemic within ticks in mountainous regions.
New techniques such as PCR and reverse genetics are helping scientists understand at last how it replicates and why it varies. For students, this virus provides an outstanding example of how to study double-stranded RNA viruses, how genetic shuffling results in variation, and how tick-borne viruses persist in our changing world.
References
- Colorado tick fever virus. (2020). Definitions (Qeios). https://doi.org/10.32388/777248
- Beane, S., Burch, M., & Bechelli, J. (2025). Colorado tick fever virus: Quantification, propagation, storage, and detection. Methods in Molecular Biology. https://doi.org/10.1007/978-1-0716-6022-4_6
- Minami, S., Nouda, R., Hirai, K., Chen, Z., Kotaki, T., Kanai, Y., … Kobayashi, T. (2025). Establishment of reverse genetics systems for Colorado tick fever virus. PLOS Pathogens, 21(2), e1012720. https://doi.org/10.1371/journal.ppat.1012720
- Shah, T., Li, Q., Wang, B., Baloch, Z., & Xia, X. (2023). Geographical distribution and pathogenesis of ticks and tick-borne viral diseases. Frontiers in Microbiology, 14, 1080191. https://doi.org/10.3389/fmicb.2023.1080191
- Emmons, R. W. (1985). An overview of Colorado tick fever. Progress in Clinical and Biological Research, 178, 127–133.
- Owen, S. C., Gaffney, C. J., Grado, L. L., Woodson, M. L., King, J. G., Balaraman, V., & Bechelli, J. (2021). Colorado tick fever virus induces apoptosis in human endothelial cells to facilitate viral replication. Ticks and Tick-borne Diseases, 12(4), 101719. https://doi.org/10.1016/j.ttbdis.2021.101719
- Fagre, A. C., Lehman, J. A., & Hills, S. L. (2024). Colorado tick fever in the United States, 2013–2022. American Journal of Tropical Medicine and Hygiene, 110(2), 339–345. https://doi.org/10.4269/ajtmh.23-0419
- Bowen, G. S. (2019). Colorado tick fever. In The Arboviruses: Epidemiology and Ecology (pp. 85–103). CRC Press. https://doi.org/10.1201/9780429266569-6
- Hughes, H. R., Velez, J., Fitzpatrick, K. A., Davis, E. H., Russell, B. J., Lambert, A. J., … Brault, A. C. (2021). Genomic evaluation of the genus Coltivirus indicates genetic diversity among Colorado tick fever virus strains and demarcation of a new species. Diseases, 9(4), 76. https://doi.org/10.3390/diseases9040076
- Williamson, B. N., Fischer, E. R., Lopez, J. E., Ebihara, H., & Schwan, T. G. (2019). Prevalence and strains of Colorado tick fever virus in Rocky Mountain wood ticks in the Bitterroot Valley, Montana. Vector-Borne and Zoonotic Diseases, 19(11), 786–794. https://doi.org/10.1089/vbz.2019.2462
- McDonald, E., George, D., Rekant, S., Curren, E. J., Debess, E., Hedberg, K., … Gould, C. V. (2019). Notes from the field: Investigation of Colorado tick fever virus disease cases—Oregon, 2018. Morbidity and Mortality Weekly Report, 68(11), 269–270. https://doi.org/10.15585/mmwr.mm6811a6
- Knudson, D. L. (1981). Genome of Colorado tick fever virus. Virology, 112(1), 190–199. https://doi.org/10.1016/0042-6822(81)90612-6
- Marfin, A. A., & Campbell, G. L. (2014). Colorado tick fever and related coltivirus infections. In Tick-borne diseases of humans (2nd ed., pp. 357–370). ASM Press. https://doi.org/10.1128/9781555816490.ch23
- Padgett, K. A., Kjemtrup, A. M., Novak, M. G., Velez, J. O., & Panella, N. A. (2022). Colorado tick fever virus in the Far West: Forgotten, but not gone. Vector-Borne and Zoonotic Diseases, 22(8), 486–496. https://doi.org/10.1089/vbz.2021.0135
- Johnson, A. J., Karabatsos, N., & Lanciotti, R. S. (1997). Detection of Colorado tick fever virus by using reverse transcriptase PCR and application of the technique in laboratory diagnosis. Journal of Clinical Microbiology, 35(5), 1203–1207. https://doi.org/10.1128/jcm.35.5.1203-1207.1997
- Colorado tick fever virus. (2022). CABI Compendium. https://doi.org/10.1079/DIS/329e5d6a785f59eb7c084a1e8ddc398
- Yendell, S. J., Fischer, M., & Staples, J. E. (2015). Colorado tick fever in the United States, 2002–2012. Vector-Borne and Zoonotic Diseases, 15(5), 311–316. https://doi.org/10.1089/vbz.2014.1732
- Emmons, R. W. (1985). An overview of Colorado tick fever. Progress in Clinical and Biological Research, 178, 127–133.
- Soto, R. A., Baldry, E. M., Vahey, G. M., Lehman, J. A., Silver, M. L., Panella, A. J., … Gould, C. V. (2020). Increase in Colorado tick fever virus disease cases and effect of COVID-19 pandemic on behaviors and testing practices, Montana, 2020. Emerging Infectious Diseases, 28(12), 2523–2532. https://doi.org/10.3201/eid2812.220981
- Atkinson, Barry. (2016). The Characterisation of Arboviral Zoonoses in Central Asia (PhD Thesis).
