The classical pathway is a chain of events in which complement components react in specific sequences as a cascade resulting in cell lysis. It is activated by antibody bound to antigen (Antigen-Antibody complex) but never by native or free antibody. These complexes may be soluble, or they may be formed when an antibody binds to antigenic determinants, or epitopes, situated on viral, fungal, parasitic, or bacterial cell membranes. Soluble antibody-antigen complexes are often referred to as immune complexes, and only complexes formed by IgM or certain subclasses of IgG antibodies are capable of activating the classical complement pathway.
Activators of the classical pathway
Activators of classical pathways include the following:
- Immunoglobulin IgM and IgG. IgG subclasses vary with regard to their efficiency in activating the complement; IgG3 are the most efficient, followed by IgG1 and IgG2. IgG4 do not activate the classical pathway.
- Staphylococcal protein A,
- C-reactive protein
- DNA
Steps of activation of classical pathway
The classical pathway of complement activation usually begins with the formation of soluble antigen–antibody complexes (immune complexes) or with the binding of antibody to antigen on a suitable target, such as a bacterial cell. Following are the sequential steps in the activation of classical pathway:
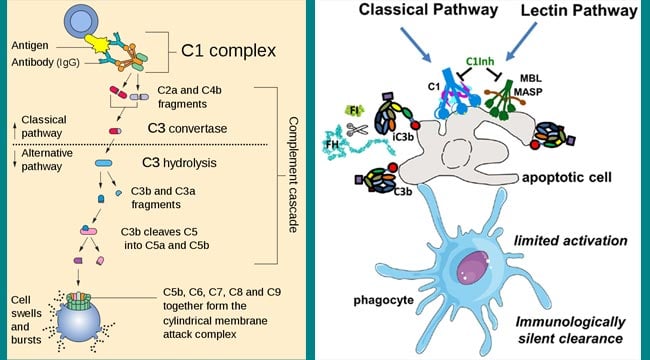
- Activation of C1: The initial stage of activation involves the complement components C1, C2, C3, and C4, which are present in plasma as zymogens. The formation of an antigen-antibody complex induces conformational changes in the non-antigen binding (Fc) portion of the antibody molecule. This conformational change exposes a binding site for the C1 component of complement. This results in the sequential activation of C4, C2, and C3.
In serum, C1 exists as a macromolecular complex consisting of one molecule of C1q and two molecules each of the serine proteases, C1r and C1s, held together in a Ca++ stabilized complex (C1qr2s2). The C1q molecule itself is composed of 18 polypeptide chains that associate to form six collagen-like triple helical arms, the tips of which bind the CH2 domain of the antigen-bound antibody molecule. Each C1 macromolecular complex must bind by its C1q globular heads to at least two Fc sites for a stable C1-antibody interaction to occur
- C1q binding in the presence of calcium ions leads to activation of C1r and C1s. Binding of C1q to the CH2 domains of the Fc regions of the antigen-complexed antibody molecule induces a conformational change in one of the C1r molecules that converts it to an active serine protease enzyme. This C1r molecule then cleaves and activates its partner C1r molecule. The two C1r proteases then cleave and activate the two C1s molecules. Activated C1s is an esterase that splits C4 into two fragments: a small soluble fragment (C4a) and a larger fragment (C4b). C4a has anaphylatoxin activity, and C4b binds to cell membrane along with C1. C4b in the presence of Mg++ splits C2 into C2a and C2b. The smaller fragment (C2b) diffuses away, while the larger fragment (C2a) remains attached to C4b. The resulting C4b2a complex possesses enzymatic activity and is called C3 convertase, which converts C3 into an active form.
- The C3 convertase activate thousands of C3 molecules and splits these molecules into C3a and C3b. A single C3 convertase molecule can generate over 200 molecules of C3b, resulting in tremendous amplification at this step of the sequence. The biological importance of activated C3b as well as C4b is that they are able to bind to C3b/C4b receptors (currently designated as CR1 receptors) present on almost all host cells, most notably phagocytes. The increased affinity of phagocytic cells for C3b (or iC3b)/C4b-coated particles is known as immune adherence. The latter is responsible for a significant enhancement of phagocytosis, which is one of the main defense mechanisms of the body.
- Some of the C3b binds to C4b2a to form a trimolecular complex C4b2a3b called C5 convertase. The C5 convertase splits C5 into C5a and C5b. C5a diffuses away, while C5b attaches to C6 and initiates formation of C5b–9 complex otherwise known as membrane attack complex (MAC).
Note Point:
C4 is activated when C1s hydrolyzes a small fragment (C4a) from the amino terminus of one of its chains. The C4b fragment attaches covalently to the target membrane surface in the vicinity of C1, and then binds C2. C4b binding to the membrane occurs when an unstable, internal thioester on C4b, exposed upon C4 cleavage, reacts with hydroxyl or amino groups of proteins or carbohydrates on the cell membrane.This reaction must occur quickly, otherwise the thioester C4b is further hydrolyzed and can no longer make a covalent bond with the cell surface.
Only antibodies bound to antigens, and not free circulating antibodies, can initiate classical pathway activation. The reason for this is that each C1q molecule must bind to at least two Ig heavy chains to be activated and each Ig Fc region has only a single C1qbinding site. Therefore, two or more Fc regions have to be accessible to C1 in order to initiate classical pathway activation.

Your article Is very educative
I always appreciate your valuable articles, so informative and enreaching.
Good info, thanks
Thank you so much 🙂
I always like reading your article, it is so enriching and educative
Thank you so much 🙂
Your write up is always rich and educating.
Thank you so much 🙂