Batch Culture Definition
- In batch fermentation, all the medium components are placed in the reactor at the start of cultivation except for atmospheric gases, acid or base for pH control, and antifoaming agents.
- There is a continuous change in the nutrient concentrations over time, and the system remains unsteady.
- The microbial metabolites may be produced at a primary or secondary stage of the microbial cultivation period.
- Fermentation is terminated when either all the nutrient is exhausted or the desired concentration of product is achieved.
Batch culture has the following mention advantages:
- Reduced risk of contamination or cell mutation as the growth period is short.
- Lower capital investment when compared to continuous processes for the same bioreactor volume.
- More flexibility with varying product/biological systems.
- Higher raw material conversion levels, resulting from a controlled growth period.
Batch Culture Principle
- A batch fermentation system is a closed system.
- At time t=0, the sterilized nutrient solution in the fermenter is inoculated with microorganisms, and incubation is allowed to proceed at a suitable temperature and gaseous environment for a suitable period.
- In the course of the entire fermentation, nothing is added, except oxygen (in the case of aerobic microorganisms), an antifoam agent, acid, or base to control pH.
- The composition of the medium, the biomass concentration, and the metabolite concentration generally change constantly as a result of the metabolism of the cells.
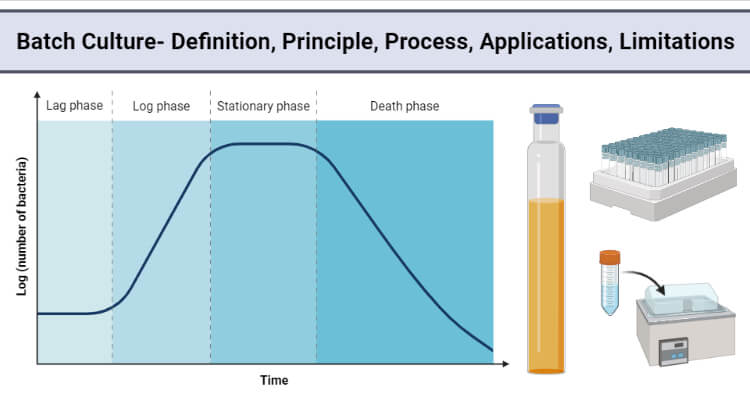
After the inoculation of a sterile nutrient solution with microorganisms and cultivation under physiological conditions, four typical phases of growth are observed.
- Lag phase
- Log phase
- Stationary phase
- Death phase
Batch Culture Process
- A batch culture begins with sterilization, and the sterile culture is then inoculated with microbes (about 2-5 % of the total volume).
- The percentages of nutrients, vitamins, and microbial cells in the reaction mixture and the temperature difference during the reaction cycle.
- The proper mixing keeps them at acceptable concentrations and temperatures.
- The process is carried out in anaerobic conditions by bubbled oxygen in or out, while acidic or alkaline solutions are added to control the pH.
- Antifoaming agents are added when indicated by a foam sensor.
- The microorganism growth is allowed to take place for days, weeks, or months.
- In the lag phase, little or no growth is observed at the beginning of fermentation, depending on a physiochemical equilibrium between the microorganism and the environment.
- Once the cells have adapted to the new conditions of growth, they enter the exponential phase.
- Primary metabolites are produced during the log or exponential phase with their formation decreasing when growth ceases. For example, Saccharomyces cerevisiae produces ethanol as a primary metabolite.
- When the cells enter the stationary phase, secondary metabolites are produced. Most antibiotics are metabolized as secondary metabolites. For example, Penicillium chrysogenum produces Penicillin as a secondary metabolite.
- The fermentation is terminated when one or more of the following has been reached:
- The microbial growth has stopped due to the depletion of the nutrients or the build of toxic compounds;
- After a fixed predetermined period;
- The concentration of desired product has been achieved.
Batch Culture Applications
- It is beneficial for the construction of biomass (Baker’s yeasts) and primary metabolites (lactic acid, citric acid, acetic acid, or ethanol production).
- In food industries, organic acids are used as preservatives or acidifiers(lactic acids, citric acids, and acetic acids), alcoholic beverages (wine, beer, and distilled spirits i.e. brandy, whisky, and rum), and sweeteners (e.g., aspartate) or amino acids used as flavoring agents (e.g., monosodium glutamate)are the various product manufactured by batch cultivation.
Batch Culture Limitations
- Due to nutrient consumption and waste build-up, microbes are exposed to constantly changing environmental conditions in batch culture.
- After reaching an endpoint, batch cultures must be restarted. In large bioreactors, it takes a long time to empty, clean, and refill the reactor.
- In batch culture, the low productivity is a consequence of the high downtime (nonproduction time spent cleaning, sterilizing, and starting up another batch cultivation) during two consecutive batches.
- A buildup of toxic metabolites can sometimes inhibit cell growth and product synthesis.
References
- Blaby, I. K. (2011). Modes of Culture / Microbial. In Comprehensive Biotechnology (Second Edition, Vol. 1). Elsevier B.V. https://doi.org/10.1016/B978-0-08-088504-9.00034-9
- Crueger, W., Crueger, A., & Aneja, K. R. (2017). Crueger’s biotechnology: A textbook of Industrial microbiology. MedTech.
- Kuila A, Sharma V (2018). Principles and Applications of Fermentation Technology. John Wiley & Sons, Inc. https://doi.org/10.1002/9781119460381
- Paulová, L. (2014). Advanced Fermentation Processes Relationship between butanol efflux and butanol tolerance of Clostridia View project Yeast biotechnology View project. https://doi.org/10.1201/b15426-6
- Saran, S., Malaviya, A., & Chaubey, A. (2019). Introduction, Scope, and Significance of Fermentation Technology.Ch1. 1, 1–25.
- Srivastava, A. K. (2011). 2.38 – Fed-Batch Fermentation – Design Strategies. Comprehensive Biotechnology, 1, 515–526. https://doi.org/10.1016/B978-0-08-088504-9.00112-4
- Yang, Y., & Sha, M. (2017). A Beginner’s Guide to Bioprocess Modes-Batch, Fed-Batch, and Continuous Fermentation.
