Anaerobic Blood Agar is a solid media recommended for use in qualitative procedures for primary isolation and cultivation of anaerobic organisms, including fastidious strains. This medium was formulated by V.R. Dowell and T.M. Hawkins at the Centers for Disease Control and Prevention in Atlanta, Georgia. Anaerobic Blood Agar supports good growth and typical pigmentation of fastidious and slow-growing anaerobes, as well as other anaerobes of significant clinical importance.
Composition of Anaerobic Blood Agar
| Ingredients | Gms/liter |
| Casein enzymic hydrolysate | 15.000 |
| Papaic digest of soybean meal | 5.000 |
| Yeast extract | 5.000 |
| Sodium chloride | 5.000 |
| L-Cysteine | 0.500 |
| Hemin | 0.005 |
| Agar | 13.50 |
Final pH: 7.4±0.2
Principle of Anaerobic Blood Agar
- Anaerobic Blood Agar base serves as a nutritious, nonselective medium allowing the cultivation of not only fastidious anaerobes but also of aerobic and microaerophilic microorganisms.
- It contains peptones which supply nitrogenous substances and amino acids necessary for the growth of anaerobic bacteria.
- Yeast extract provides B-complex vitamins and serves as a growth enhancer.
- Hemin, vitamin K, and sheep blood stimulate the growth of anaerobes like Bacteroides species and gram-positive spore bearers like Clostridium species.
- Sodium chloride is a source of essential electrolytes and maintains osmotic equilibrium.
- Addition of blood provides nutrients and helps to differentiate hemolytic organisms.
Preparation and Method of Use of Anaerobic Blood Agar
- Suspend 44.0 grams in 1000 ml distilled water.
- Heat to boiling to dissolve the medium completely.
- Add the rehydrated contents of 1 vial of Vitamin K1 solution.
- Sterilize by autoclaving at 15 lbs pressure (121°C) for 15 minutes.
- Cool to 45-50°C.
- Aseptically add 5% v/v sterile defibrinated sheep blood.
- Mix well and pour into sterile Petri plates.
- Prior to use, reduce the plates for a minimum of 24 hours by placing them in an anaerobic environment at room temperature.
- Inoculate specimens for anaerobic culture on both selective and non-selective media as soon as possible after receipt in the laboratory; streak plates for isolation.
- Incubate anaerobically at 33-37°C for 48-72 hours.
- Confirm anaerobic growth by subculture to anaerobic blood agar plate.
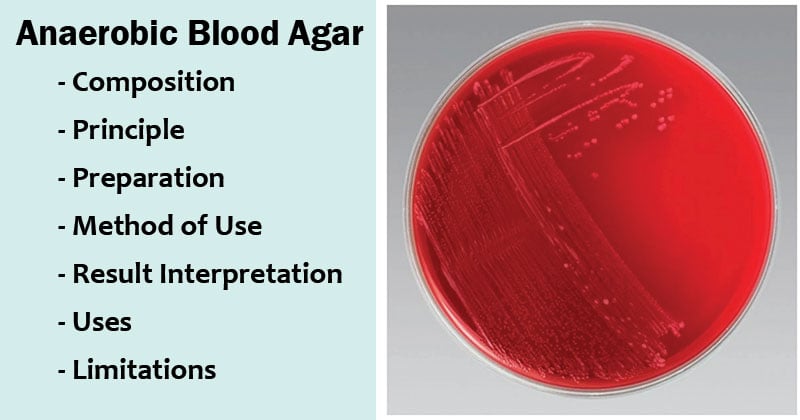
Result Interpretation of Anaerobic Blood Agar
| Organisms | Growth |
| Bacteroides fragilis | Luxuriant Growth |
| Bacteroides melaninogenicus | Luxuriant Growth |
| Peptostreptococcus anaerobius | Luxuriant Growth |
| Clostridium perfringens | Growth, double zone β-hemolysis |
| Fusobacterium nucleatum | Growth |
| Prevotella melaninogenica | Growth; pigment (tan to brown) |
| Escherichia coli | Growth |
| Staphylococcus aureus | Growth |
Uses of Anaerobic Blood Agar
- Anaerobic Blood Agar Base is recommended for the cultivation of anaerobic microorganisms, including very fastidious organisms from clinical specimens.
- It promotes both typical pigment formation in Bacteroides melaningenicus and displays double hemolytic reaction in Clostridium perfringens with added blood to the medium base.
- The media supports typical pigment production by pigmented Prevotella and Porphyromonas
Limitations of Anaerobic Blood Agar
- The media must be prepared, dispensed, and packaged under oxygen-free conditions to prevent the formation of oxidized products prior to use.
- This Agar will not provide complete information for identification of bacterial isolates. Additional test procedures and media are required for complete identification.
- It is recommended that a selective media such as Anaerobic Brucella Laked Blood Agar with Kanamycin and Vancomycin and/or Anaerobic Brucella Blood Agar with Phenylethyl Alcohol also be inoculated with clinical specimens to assure growth of all species present.
References
- http://himedialabs.com/TD/M975A.pdf
- Dowell, V.R., Jr. and T.M. Hawkins. 1974. Laboratory Methods in Anaerobic Bacteriology, CDC Laboratory Manual. U.S. Dept. of H.H.S. and CDC, Atlanta, GA.
- https://assets.thermofisher.com/TFS-Assets/LSG/manuals/IFU1040.pdf
- https://anaerobesystems.com/products/plated-media/anaerobe-blood-agar-cdc/
- https://www.fishersci.ca/

Hi! thanks for this information. I only wonder how you create anaerobic culture conditions. Thanks in advance