According to the field of biochemistry, numerous metabolic pathways regulate the disintegration and use of different compounds inside living organisms. One of these routes, alpha oxidation, is essential for the metabolism of specific fatty acids.
This note aims to give readers a thorough grasp of alpha oxidation by going through its relevance, location, process, stages, terminology, and related disorders. We may understand the significance of alpha oxidation in cellular metabolism by looking into these factors.
What is Alpha Oxidation?
Alpha-oxidation is the oxidation of fatty acid (methyl group at beta carbon) by removing one carbon unit adjacent to the α-carbon from the carboxylic end.
- The carbon unit is removed in the form of CO2.
- Alpha oxidation occurs in those fatty acids that have a methyl group (-CH3) at the beta-carbon, which blocks beta-oxidation.
- There is no production of ATP.
- A metabolic process known as “alpha oxidation” is in charge of breaking down certain fatty acids that include a methyl group at the alpha carbon position.
- Peroxisomes, subcellular organelles in charge of several metabolic functions, are where it is most commonly found.
- In contrast to beta-oxidation, which occurs in mitochondria, alpha oxidation offers a different pathway for the oxidation of fatty acids with certain structural characteristics.
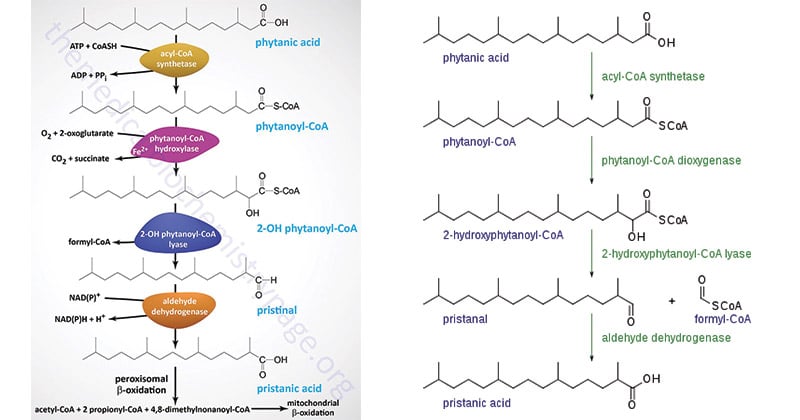
Location of Alpha Oxidation
Peroxisomes are where alpha oxidation takes place.
Peroxisomes are membrane-bound organelles that are present in eukaryotic cells and are essential for the metabolism of lipids. Enzymes for the degradation of different substances, including fatty acids, are present in these organelles. The importance of alpha oxidation in these organs is highlighted by the presence of peroxisomes in certain tissues, such as the liver and kidney.
The Pathway of Alpha Oxidation
The alpha oxidation pathway involves a series of enzymatic reactions that ultimately lead to the breakdown of fatty acids. The main steps of this pathway include activation, hydration, oxidation, and thiolysis. These sequential reactions work synergistically to convert fatty acids into smaller molecules, which can be utilized for energy production or other metabolic processes.
1. Activation
- The fatty acid molecule is activated and prepared for further degradation. The fatty acid first undergoes activation by the enzyme acyl-CoA synthetase.
- The reaction involves the attachment of Coenzyme A (CoA) to the fatty acid, resulting in the formation of fatty acyl-CoA.
- This process requires the input of ATP (adenosine triphosphate), which is hydrolyzed to AMP (adenosine monophosphate) and inorganic pyrophosphate (PPi).
- The fatty acyl-CoA is now primed for the subsequent steps of the alpha oxidation pathway.
2. Hydration
- After activation, the fatty acyl-CoA undergoes hydration, which introduces a hydroxyl group (-OH) at the alpha carbon position of the molecule.
- This reaction is catalyzed by the enzyme enoyl-CoA hydratase.
- The enzyme adds a water molecule across the double bond in the fatty acyl-CoA, resulting in the formation of a hydroxyl group.
- The product of this step is known as 2-hydroxy acyl-CoA.
3. Oxidation
- In the oxidation step, the hydroxyl group introduced in the previous step is oxidized, leading to the formation of a keto group (=O) at the alpha carbon position.
- This reaction is facilitated by the enzyme 2-hydroxy acyl-CoA lyase.
- The enzyme catalyzes the removal of water from the 2-hydroxy acyl-CoA, resulting in the formation of an alpha-ketoacyl-CoA.
4. Thiolysis
- The final step of the alpha oxidation pathway is thiolysis, where the alpha-ketoacyl-CoA is cleaved into acetyl-CoA and a shortened acyl-CoA molecule.
- This reaction is mediated by the enzyme thiolase. The enzyme catalyzes the cleavage of the alpha-ketoacyl-CoA using Coenzyme A-SH (CoA-SH) as a nucleophile.
- As a result, acetyl-CoA is released, while the remaining acyl-CoA molecule is shortened by one carbon atom.
Reactions in Alpha Oxidation
The reactions in the alpha oxidation pathway play a crucial role in breaking down specific fatty acids. These reactions involve the activation of fatty acids, followed by the introduction of a hydroxyl group, oxidation, and subsequent cleavage. The overall result is the generation of acetyl-CoA, which can be utilized for energy production through the citric acid cycle or other metabolic pathways.
Step 1: Activation
Reaction: Fatty acid + CoA + ATP → Fatty acyl-CoA + AMP + PPi
Enzyme: Acyl-CoA synthetase
Step 2: Hydration
Reaction: Fatty acyl-CoA + H2O → 2-Hydroxyacyl-CoA
Enzyme: Enoyl-CoA hydratase
Step 3: Oxidation
Reaction: 2-Hydroxyacyl-CoA → α-Ketoacyl-CoA
Enzyme: 2-Hydroxyacyl-CoA lyase
Step 4: Thiolysis
Reaction: α-Ketoacyl-CoA + CoA-SH → Acetyl-CoA + Shortened acyl-CoA
Enzyme: Thiolase
These reactions occur sequentially and are catalyzed by specific enzymes, leading to the degradation of fatty acids and the generation of acetyl-CoA, which can be further utilized in various metabolic pathways.
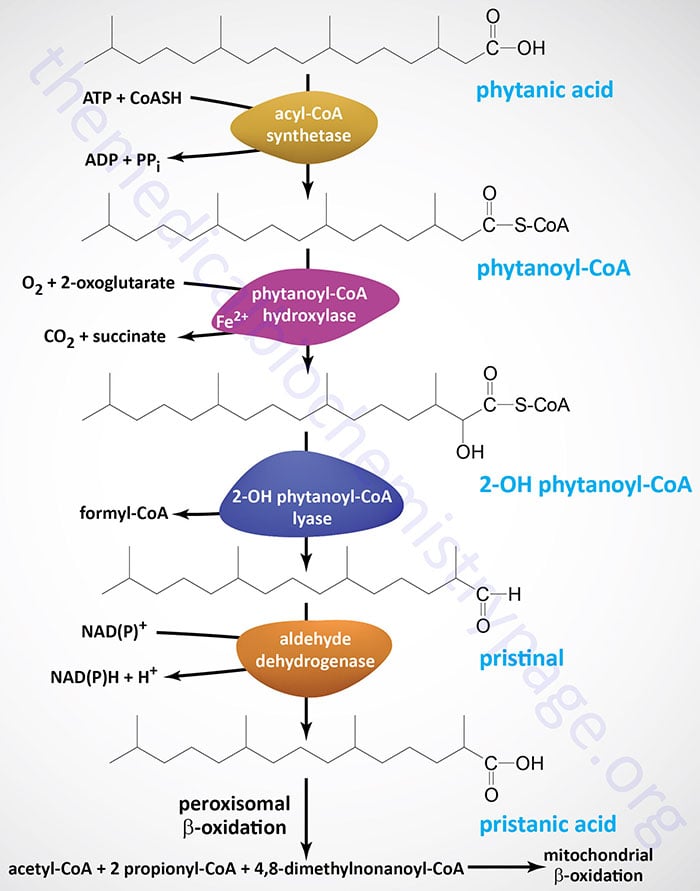
Significance of Alpha Oxidation
- For a number of reasons, alpha oxidation is crucial to cellular metabolism. First off, it gives fatty acids containing methyl groups at the alpha carbon position, which are ineligible for beta-oxidation, another route to breakdown.
- This makes it possible to effectively break down certain fatty acids that cannot be metabolized via the traditional pathway.
- Additionally, the metabolism of branched-chain fatty acids like phytanic acid and pristanic acid depends heavily on alpha oxidation.
- These branched-chain fatty acids are frequently discovered in food sources including dairy goods and specific kinds of meat. The appropriate breakdown of these fatty acids is ensured by alpha oxidation, which also avoids their buildup, which might result in metabolic problems.
- Additionally, alpha oxidation helps in the creation of significant substances like bile acids.
- The breakdown and absorption of dietary lipids depend heavily on bile acids. In order to ensure appropriate fat digestion and absorption, the alpha oxidation pathway contributes to the production of intermediate intermediates that are used in the synthesis of bile acids.
- Additionally, the metabolism of several poisons and xenobiotics involves alpha oxidation. For instance, phytanic acid, a byproduct of alpha oxidation, can be obtained through food or produced internally from chlorophyll breakdown products.
- The buildup of phytanic acid can result in Refsum disease, a syndrome marked by neurological and visual deficits, in people with poor alpha oxidation.
Alpha Oxidation Associated Diseases
- Numerous metabolic diseases can be caused by alpha oxidation-related enzyme shortages or defects.
- One such condition is the previously described Refsum illness, which is brought on by a lack of the alpha oxidation enzyme phytanoyl-CoA hydroxylase.
- This causes phytanic acid to build up and causes neurological and visual problems.
- The uncommon genetic ailment Zellweger syndrome, which is characterized by the lack or malfunction of peroxisomes, is another condition connected to alpha oxidation.
- People with Zellweger syndrome have poor alpha oxidation since peroxisomes are the main location for alpha oxidation, which leads to the buildup of fatty acids and other metabolic problems.
Conclusion
The breakdown of certain fatty acids within peroxisomes is carried out via the essential metabolic route known as alpha oxidation. It gives fatty acids with methyl groups at the alpha carbon position a different pathway for oxidation. The process of alpha oxidation breaks down fatty acids into smaller molecules like acetyl-CoA, which can be used to synthesize vital chemicals or provide energy.
The importance of alpha oxidation can be attributed to its capacity to metabolize branched-chain fatty acids, support the production of bile acids, and participate in the breakdown of toxins and xenobiotics.
However, metabolic illnesses like Refsum disease and Zellweger syndrome can result from flaws or deficits in the enzymes involved in alpha oxidation. Understanding the intricacies of alpha oxidation enhances our knowledge of cellular metabolism and the role of peroxisomes in lipid metabolism.
Further research in this field can shed light on potential therapeutic strategies for managing metabolic disorders associated with alpha oxidation.
References
- Casteels, Minne. “alpha-Oxidation of 3-methyl-branched fatty acids: unraveling of a pathway.” Verhandelingen-Koninklijke Academie Voor Geneeskunde van Belgie 68.3 (2006): 199-221.
- Alpha Oxidation of Fatty Acids – https://byjus.com/neet/alpha-oxidation-of-fatty-acids/
- Wanders, Ronald JA, and Hans R. Waterham. “Peroxisomal disorders: the single peroxisomal enzyme deficiencies.” Biochimica et Biophysica Acta (BBA)-Molecular Cell Research 1763.12 (2006): 1707-1720.
- Ferdinandusse, Sacha, and Sander M. Houten. “Peroxisomes and bile acid biosynthesis.” Biochimica et Biophysica Acta (BBA)-Molecular Cell Research 1763.12 (2006): 1427-1440.
- Smith, C. M., Marks, A. D., Lieberman, M. A., Marks, D. B., & Marks, D. B. (2005). Marks’ basic medical biochemistry: A clinical approach. Philadelphia: Lippincott Williams & Wilkins.
- https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/alpha-oxidation
- https://www.slideshare.net/AnupShamsherBudhatho/fatty-acid-oxidation-beta-alpha-omega-and-peroxisomal
- https://en.wikipedia.org/wiki/Alpha_oxidation
- Rodwell, V. W., Botham, K. M., Kennelly, P. J., Weil, P. A., & Bender, D. A. (2015). Harper’s illustrated biochemistry (30th ed.). New York, N.Y.: McGraw-Hill Education LLC.

Awesome!