Actin Definition
Actin is a group of globular proteins that are the most abundant proteins in most eukaryotic cells and help in providing shape, structure, and mobility to the body.
- Actin is present in all eukaryotes except for the nematode sperm.
- The actin proteins are highly conserved and participate in more protein-protein interactions than any known protein.
- Actin is different from other proteins in their ability to transition between two states; monomeric (G-actin) and filamentous (F-actin) state.
- The transition is possible under the control of nucleotide hydrolysis, ions, and a large number of actin-binding proteins.
- Actin is the monomeric unit of two different cytoskeleton filaments; microfilaments and contractile apparatus.
- Actin proteins play essential roles in cell division, cell motility, and cell signaling.
- Cell’s ability to form an intensive network of microfilaments allows the cell to remodel itself in response to environmental stimuli like increasing cell adhesion to form tissues.
- Vertebrates express three main actin isoforms, including three α-isoforms of skeletal, cardiac, and smooth muscles, and the β- and γ-isoforms are expressed in nonmuscle and muscle cells.
- Actin filaments in the muscles are short in length ranging from 2-2.6 µm and thin with a diameter of about 0.005 µm.
- The actin filaments in muscles are separated by actin-binding proteins, α-actinin that binds two actin filaments while leaving space for myosin. The α-actin is the significant component of the contractile apparatus of muscles.
- Each actin filament in a muscle fiber is composed of two strands of actin protein.
Myosin Definition
Myosin is a superfamily of motor proteins that, together with actin proteins, form the basis for the contraction of muscle fibers.
- Myosin is termed a motor protein as it is a type of enzyme that converts chemical energy into mechanical energy.
- Myosin is an ATPase that moves along actin filaments by connecting the hydrolysis of ATP to conformational changes.
- All myosins are composed of one or two heavy chains and several light chains.
- Genomic analysis has revealed 13 different myosins.
- All consist of a highly conserved head domain, which is actin activated ATPase responsible for generating movement; a neck domain, which is associated with several regulatory light-chain subunits; and an effector tail domain, which is characteristic to each type of myosin and is responsible for its specific functions in cells.
- Myosin filaments are longer in size of about 4-5 µm and are also thicker with a diameter of 0.01 µm.
- Myosin in muscle is Myosin II that is a polymeric molecule with a long rod-like tail domain, assembled into thick bipolar filaments.
- In the presence of ATP, the dimeric head domain of myosin II alone can generate movement.
- Myosins have functions other than muscle contraction depending on the type of myosin and the organism.
- The structure and function of myosin are conserved across various species with the myosin of mammals compatible with the actin of unicellular organisms.
Key differences (Actin vs Myosin)
Basis for comparison |
Actin |
Myosin |
| Definition | Actin is a group of globular proteins that are the most abundant proteins in most eukaryotic cells and help in providing shape, structure, and mobility to the body. | Myosin is a family of motor proteins that, together with actin proteins, form the basis for the contraction of muscle fibers. |
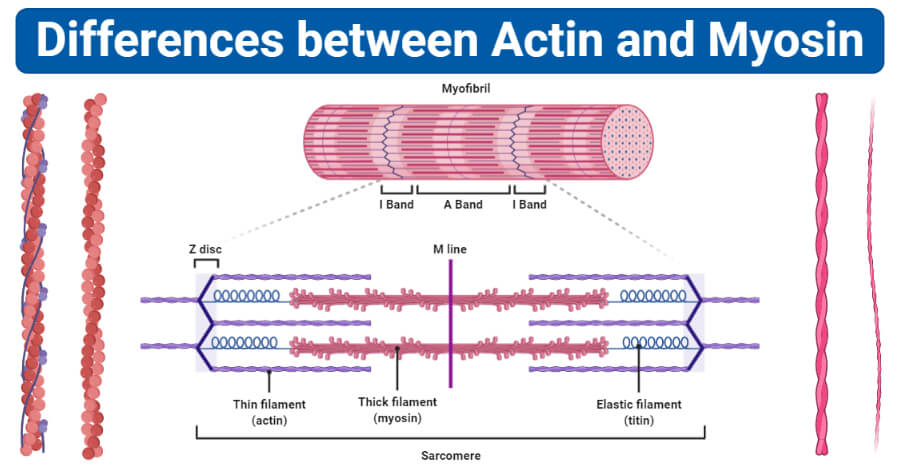
| Found in | Actin proteins are found in both A and I bands of the sarcomere. | Myosin proteins are found only in the A bands of the sarcomere. |
| Size
|
These are shorter (2-2.6 µm in length) and thinner (0.005 µm in diameter). | These are longer (4-5 µm in length) and thicker (0.01 µm in diameter). |
| Nature | Actin proteins are globular proteins. | Myosin proteins are motor proteins. |
| Molecular weight | The molecular weight of the actin proteins is relatively less. | The molecular weight of myosin proteins is relatively more. |
| Abundance in muscle cells | Actin filaments are more abundant than myosin. | Myosin is less abundant compared to actin. There is one myosin for each actin filament. |
| Surface | The surface of the actin is smooth. | The surface of the myosin is rough. |
| Proteins in filaments | Actin filaments are composed of actin, tropomyosin, and troponin proteins. | Myosin filaments are composed of myosin and meromyosin proteins. |
| Cross bridges | No cross-bridges are present in actin filaments. | Myosin form cross-bridges. |
| Association with ATP | Actin is not associated with ATP molecules. | Myosin remains associated with ATP molecules. |
| End | One end of the actin filament is free while the other is bound to Z lines. | Both the ends of myosin filaments are free. The head domain of myosin, however, remains in association with ATP. |
| Sliding | Actin filaments slide into the H zone during contraction. | Myosin filaments do not slide into the H zone during contraction. |
| Location | Actin is present in muscle fibers, microfilaments, cell membrane, and cell wall. | Myosin is primarily found in muscle cells. |
| Muscle contraction | Actin interacts with myosin to support muscle contraction. | Myosin initiates muscle contraction by generating a force by binding to the ATP molecule. |
Examples of Actin
Microfilaments
- Microfilaments in cells, also termed, actin filaments, are proteins that form a part of the cytoskeleton.
- These are polymers of actin proteins that interact with other proteins in the cell.
- Microfilaments are about 7 nm in diameters and each of the filaments is made up of two strands of actin.
- Microfilaments perform a number of functions ranging from cytokinesis, changing the shape of the cell to cell motility.
- Additionally, actin structures are also present adjacent to the plasma membrane forming a structure that is modified according to the function of the cell, like the middle piece of mammalian sperm.
Examples of Myosin
Myosin in stereocilia
- Myosin III proteins are located at the tip of the stereocilia in the inner ear.
- The stereocilia are about 10-15 µm in length and are similar in structure with the microvilli of the intestinal tract.
- These stereocilia are mechanosensing organelles that respond to the fluid motion in the ear, performing the functions of hearing and balancing.
- It forms a complex with the PDZ domain-containing protein which, together with actin proteins, assists the response of the stereocilia to sound waves.
- The myosin allows the stereocilia to adjust to the change in sound waves and fluid motion by moving the cilia.
- Besides, genes coding for the myosin proteins were also found in some other parts of the ear like the cochlea.
References
- Dominguez, R., & Holmes, K. C. (2011). Actin structure and function. Annual review of biophysics, 40, 169–186. https://doi.org/10.1146/annurev-biophys-042910-155359
- Lodish H, Berk A, Zipursky SL, et al. Molecular Cell Biology. 4th edition. New York: W. H. Freeman; 2000. Section 18.3, Myosin: The Actin Motor Protein.Available from: https://www.ncbi.nlm.nih.gov/books/NBK21724/
Sources
- 4% – https://www.ncbi.nlm.nih.gov/books/br.fcgi?book=mcb&part=A5176
- 2% – https://www.annualreviews.org/doi/full/10.1146/annurev-biophys-042910-155359
- 2% – https://en.wikipedia.org/wiki/Microfilament
- 1% – https://www.wisegeek.com/what-are-myosin-heavy-chains.htm
- 1% – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4022326/
- 1% – https://www.annualreviews.org/doi/10.1146/annurev-biophys-042910-155359
- 1% – https://quizlet.com/37312016/physiology-muscles-flash-cards/
- 1% – https://quizlet.com/29664307/muscle-flash-cards/
- 1% – https://en.wikipedia.org/wiki/Motor_protein
- 1% – https://en.wikipedia.org/wiki/Actin
- 1% – http://www.differencebetween.net/science/difference-between-cilia-and-stereocilia/
- <1% – https://www.sciencedirect.com/topics/neuroscience/actin-filament
- <1% – https://www.researchgate.net/profile/Agnieszka_Rzadzinska/publication/26831330_MyosinVIIa_Interacts_with_Twinfilin-2_at_the_Tips_of_Mechanosensory_Stereocilia_in_the_Inner_Ear/links/09e4150fd4961ca370000000.pdf?origin=publication_list
- <1% – https://study.com/academy/lesson/actin-filaments-function-structure-quiz.html
