Acree-Rosenheim test definition
Acree-Rosenheim Test is a biochemical test used for the detection of tryptophan molecules in a protein sample. Acree-Rosenheim test is one of the essential tests for the confirmation of formaldehyde in the milk as milk vendors use formaldehyde as a preservative. This test thus helps to understand the condition of the milk sample so as to determine if the surplus milk is not spoiled. A more simplified method of determining the presence of formaldehyde in milk is also available. For the simpler method, the milk can just be heated with a few drops of concentrated HCl to test the detection of formaldehyde. It is also called the aldehyde test as the test is based on the presence of aldehyde molecules in the sample. The test is named after two biochemists; Solomon Farley Acree and Sigmund Otto Rosenheim.
Objectives of Acree-Rosenheim test
- To detect the presence of tryptophan in a protein sample.
- To detect the presence of formaldehyde in a sample.
Principle of Acree-Rosenheim test
The reagent for the Acree-Rosenheim test is formaldehyde. The test is used to detect tryptophan in a protein solution. The formaldehyde added to the sample results in a condensation reaction with two tryptophan molecules. The reaction is carried out in acidic conditions by the addition of sulfuric acid (H2SO4), which separates two distinct layers in the test tube. The condensation product of the reaction between tryptophan and formaldehyde is violet colored, which indicates the presence of tryptophan molecules.
Reaction
Tryptophan + Formaldehyde + Tryptophan → Coloured condensation product + H2O
Requirements
Reagents
- Protein sample (1% tryptophan or other amino acids)
- Dilute Formaldehyde (CH2O)
- Sulfuric acid (H2SO4)
Materials Required
- Test tubes
- Test tube stand
- Pipettes
Procedure of Acree-Rosenheim test
- About 2-3ml of protein solution is added in a test tube. If egg albumin is to be used, 5-10% solution is to be prepared.
- To the test tube, a few drops of dilute formaldehyde are added.
- Thereafter, few drops of concentrated sulfuric acid are added from the side of the test tubes.
- The shaking of the test tube should be avoided to prevent the mixing of the two layers.
- The formation of the violet-colored ring at the junction of two layers is observed.
For milk samples with preservatives
- In the case of milk samples, a few drops of concentrated acid (H2SO4) are added to the milk sample.
- The mixture is then heated to obtain a purple-colored solution.
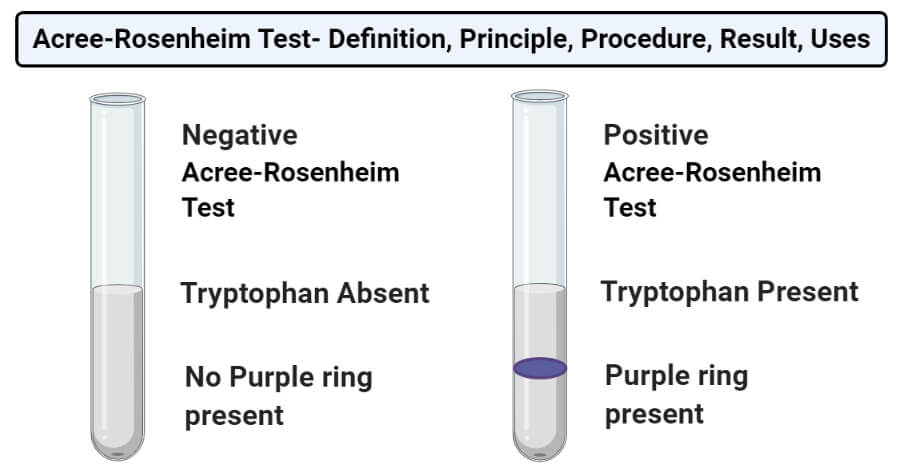
Result and Interpretation of Acree-Rosenheim test
- Positive result: Positive result of the Acree-Rosenheim test is represented by the appearance of a purple ring at the junction of two layers, indicating the presence of tryptophan.
- Negative result: Negative result of the Acree-Rosenheim test is represented by the absence of the purple ring at the junction of two layers.
Uses of Acree-Rosenheim test
- The test is used to detect the presence of distinct proteins and amino acids in a sample.
- This test is the standard test for the detection of tryptophan in a sample.
- A modified version of this test is also used for the detection of formaldehyde in a milk sample.
Limitations of Acree-Rosenheim test
- The test can only be used for the detection of tryptophan, so a negative result doesn’t rule out the presence of other amino acids.
- Gelatin might give a negative Acree-Rosenheim test as it is deficient in tryptophan.
- The shaking of the test tube might result in the mixing of the two layers, which might result in false-negative results.
References and Sources
- Nigam S. C. and Omkar (2003). Experimental Animal Physiology and Biochemistry. New Age International Pvt. Limited. New Delhi.
- D (2012). Biochemistry. Fourteenth Edition. Academic Publishers. Kolkata.
- 1% – https://www.webmd.com/dvt/what-is-the-d-dimer-test
- 1% – https://www.ncbi.nlm.nih.gov/pubmed/2448534
- 1% – https://www.epa.gov/sites/production/files/2016-09/documents/formaldehyde.pdf
- 1% – https://www.chegg.com/homework-help/questions-and-answers/1-meniscus-multiple-choice-1none-answer-choices-correct-2curvature-water-level-3-highest-m-q51401563
- 1% – https://vlab.amrita.edu/?sub=3&brch=63&sim=1094&cnt=1
- 1% – https://uomustansiriyah.edu.iq/media/lectures/6/6_2019_02_23!11_13_25_AM.pdf
- 1% – https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Map%3A_Organic_Chemistry_(McMurry)/19%3A_Aldehydes_and_Ketones-_Nucleophilic_Addition_Reactions/19.03%3A_Oxidation_of_Aldehydes_and_Ketones
- 1% – http://www.ukm.my/mjas/v7_n1/06-khan29.pdf
- 1% – http://www.jbc.org/content/235/4/999.full.pdf
- 1% – http://www.jbc.org/content/2/4/289.full.pdf
- 1% – http://vlab.amrita.edu/?sub=3&brch=63&sim=1094&cnt=1
- 1% – http://amrita.olabs.edu.in/?sub=73&brch=8&sim=140&cnt=2
- <1% – https://www.reference.com/science/make-1-n-sulfuric-acid-solution-234e6a25aa465b62